Advanced Electrochemical Strategy for Scalable Benzyl Isothiocyanate Production in Fine Chemicals
Advanced Electrochemical Strategy for Scalable Benzyl Isothiocyanate Production in Fine Chemicals
The landscape of organic synthesis is undergoing a transformative shift towards greener, more efficient methodologies, particularly in the production of high-value intermediates for the pharmaceutical and agrochemical sectors. A groundbreaking development detailed in patent CN113881956A introduces a novel electrochemical preparation method for benzyl isothiocyanate compounds, addressing long-standing challenges in efficiency and environmental impact. This technology leverages constant current electrolysis to facilitate the direct functionalization of ethylbenzene derivatives, offering a streamlined alternative to conventional multi-step processes. The core innovation lies in the ability to generate these biologically active scaffolds under mild conditions, utilizing trimethylsilyl isothiocyanate as a key reagent in an undivided cell setup. For R&D directors and procurement specialists seeking reliable sources of complex heteroatom-containing intermediates, this patent represents a significant leap forward in process chemistry.
Benzyl isothiocyanates are not merely academic curiosities; they are pivotal structures found in numerous natural products and serve as critical building blocks for synthesizing thiourea derivatives and various heterocyclic systems with potent biological activities. The patent highlights their relevance in developing agents with sterilization and anticancer properties, underscoring the commercial imperative for robust supply chains. By establishing a new synthetic pathway that bypasses the limitations of prior art, this method ensures a more stable and cost-effective supply of these essential pharmaceutical intermediates. The structural versatility of the products is evident, allowing for diverse substitution patterns that are crucial for medicinal chemistry optimization campaigns.
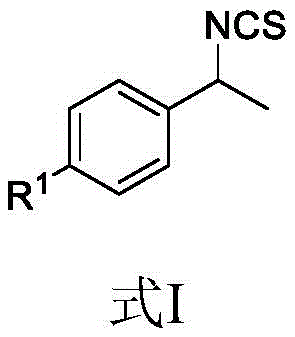
The general structure of the target molecules, as defined in the patent, allows for a broad scope of substituents denoted as R1. This flexibility is paramount for drug discovery teams who require rapid access to analog libraries. The substituents can range from electron-withdrawing halogens like fluorine, chlorine, and bromine to electron-donating groups such as methoxy and tert-butyl. Furthermore, the tolerance for bulky groups like cyclohexyl and functional handles like chloromethyl expands the utility of these intermediates in downstream coupling reactions. This breadth of scope ensures that the methodology is not limited to a single niche application but serves as a platform technology for generating a wide array of high-purity fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzyl isothiocyanate compounds has been plagued by significant operational and safety hurdles that hinder efficient commercial manufacturing. Traditional routes often rely on the reaction of amines with carbon disulfide, a process that typically requires multiple steps and involves the handling of highly toxic and volatile reagents. Alternatively, methods utilizing alcohols and sodium thiocyanate necessitate the addition of strong acids like oxalic acid and heating to elevated temperatures, such as 60°C, to drive the reaction to completion. These severe reaction conditions not only pose safety risks in a plant environment but also limit the functional group compatibility, often leading to decomposition of sensitive substrates. Moreover, the generation of stoichiometric waste and the difficulty in separating byproducts contribute to higher production costs and environmental burdens, making these legacy processes less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a paradigm shift by utilizing electricity as a clean reagent to drive the transformation. The reaction proceeds by mixing substituted ethylbenzene with trimethylsilyl isothiocyanate (TMSNCS) in the presence of a supporting electrolyte and a specific solvent system. This approach eliminates the need for toxic carbon disulfide or harsh acidic promoters, operating instead at room temperature under constant current conditions. The use of a graphite felt anode and a nickel sheet cathode provides a robust and inexpensive electrode setup that facilitates the oxidative coupling efficiently. This mild protocol not only enhances safety but also simplifies the workup procedure, as the reaction mixture can be directly concentrated and purified via standard chromatography, thereby reducing the overall processing time and resource consumption.

Mechanistic Insights into Electrochemical Oxidative Functionalization
The success of this synthesis relies on the precise control of electrochemical parameters to achieve selective C-H functionalization at the benzylic position. The mechanism likely involves the anodic oxidation of the ethylbenzene substrate to generate a reactive benzylic cation or radical cation intermediate, which is subsequently trapped by the nucleophilic sulfur species derived from TMSNCS. The choice of solvent plays a critical role in stabilizing these charged intermediates; the patent specifies a mixture of dichloroethane (DCE) and hexafluoroisopropanol (HFIP) in a 3:1 ratio. HFIP is known for its unique ability to stabilize cationic species through hydrogen bonding and its low nucleophilicity, which prevents unwanted side reactions and promotes the desired substitution pathway. The electrolyte, such as tetrabutylammonium hexafluorophosphate or perchlorate, ensures sufficient conductivity in the organic medium, allowing for efficient electron transfer at the electrode surface without requiring excessive voltage that could lead to substrate degradation.
From an impurity control perspective, the mildness of the room temperature operation is a decisive factor in maintaining high product quality. In traditional thermal methods, elevated temperatures often promote elimination reactions or polymerization of the reactive isothiocyanate species, leading to complex impurity profiles that are difficult to purge. By maintaining the reaction at ambient temperature, the electrochemical method minimizes thermal stress on the product, resulting in cleaner crude mixtures. Furthermore, the use of TMSNCS as the sulfur source avoids the generation of inorganic salts that are typical in reactions using sodium thiocyanate, thus simplifying the purification process. This results in a final product with a superior impurity profile, which is a critical requirement for commercial scale-up of complex pharmaceutical intermediates where regulatory compliance regarding residual solvents and heavy metals is stringent.
How to Synthesize Benzyl Isothiocyanate Efficiently
The practical implementation of this electrochemical protocol is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The procedure involves charging a reactor with the substrate, reagent, electrolyte, and solvent, followed by the application of a constant current for a defined period. The simplicity of the setup, requiring only a power supply and basic electrodes, lowers the barrier to entry for adoption. Detailed operational parameters, including current density and reaction time, have been optimized to ensure consistent reproducibility across different batches. For process chemists looking to implement this route, the following guide outlines the standardized steps derived from the patent examples to achieve optimal yields and purity.
- Prepare the reaction mixture by adding substituted ethylbenzene, trimethylsilyl isothiocyanate (TMSNCS), and an electrolyte such as tetrabutylammonium hexafluorophosphate into a reactor containing a solvent mixture of dichloroethane and hexafluoroisopropanol.
- Set up the electrochemical cell using a graphite felt anode and a nickel sheet cathode, then apply a constant current (e.g., 7 mA) at room temperature for approximately 6 hours.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using silica gel column chromatography to isolate the pure benzyl isothiocyanate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers tangible benefits that extend beyond mere technical novelty. The elimination of hazardous reagents like carbon disulfide reduces the regulatory burden and insurance costs associated with storing and handling toxic materials. Additionally, the avoidance of expensive transition metal catalysts, which are often required in cross-coupling approaches to similar structures, removes the need for costly metal scavenging steps and rigorous testing for residual metals in the final API. This streamlining of the process translates directly into cost reduction in pharmaceutical intermediate manufacturing, as fewer unit operations and lower material costs contribute to a more competitive pricing structure. The reliance on electricity as the primary driver of the reaction also aligns with sustainability goals, potentially qualifying the process for green manufacturing incentives.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the use of commodity chemicals such as substituted ethylbenzenes and trimethylsilyl isothiocyanate, which are readily available in bulk quantities. By removing the need for stoichiometric oxidants or precious metal catalysts, the raw material costs are significantly optimized. Furthermore, the simplified workup procedure, which involves solvent removal and direct chromatography, reduces labor hours and consumable usage compared to multi-step extractions and washes required in traditional methods. These factors collectively drive down the cost of goods sold (COGS), allowing for more aggressive pricing strategies in a competitive market.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of raw materials and the robustness of the synthesis route. The starting materials for this electrochemical process are stable, shelf-stable liquids that do not require special storage conditions like cryogenic temperatures or inert atmospheres, unlike many organometallic reagents. This stability ensures that inventory can be maintained without significant degradation, mitigating the risk of supply disruptions. Moreover, the scalability of electrochemical reactors is well-established, meaning that production capacity can be increased by adding more cells or increasing electrode surface area without fundamental changes to the chemistry, ensuring reducing lead time for high-purity intermediates during periods of high demand.
- Scalability and Environmental Compliance: As the industry moves towards stricter environmental regulations, the waste profile of a chemical process becomes a key decision factor. This electrochemical method generates minimal waste, primarily consisting of the supporting electrolyte and solvent, both of which can potentially be recovered and recycled. The absence of heavy metal contamination simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the supply chain against tightening regulations but also enhances the brand value of the end-product by associating it with sustainable manufacturing practices, a growing priority for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthesis method is essential for making informed sourcing decisions. The following questions address common inquiries regarding the scope, safety, and scalability of this electrochemical process. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of this electrochemical method over traditional synthesis?
A: Unlike traditional methods that require toxic reagents like carbon disulfide or harsh acidic conditions with sodium thiocyanate, this electrochemical approach operates under mild room temperature conditions with easily available starting materials, significantly simplifying the operational workflow and improving safety profiles.
Q: What types of substituents are compatible with this synthetic route?
A: The method demonstrates excellent functional group tolerance, successfully accommodating a wide range of substituents on the benzene ring including halogens (fluorine, chlorine, bromine), alkoxy groups (methoxy, benzyloxy), alkyl chains (ethyl, tert-butyl, isopropyl), and even chloromethyl groups.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes constant current electrolysis which is inherently scalable, avoids the use of expensive transition metal catalysts that require complex removal steps, and employs standard solvents, making it highly viable for commercial scale-up in fine chemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Isothiocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic technologies to maintain a competitive edge in the global fine chemicals market. Our team of expert process chemists has extensively evaluated the electrochemical pathway described in CN113881956A and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with advanced electrochemical synthesis units and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzyl isothiocyanate delivered meets the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can optimize your supply chain and accelerate your time to market.
