Advanced Metal-Free Manufacturing of 4-(Aryldiazenyl)-2,3-dihydropyrrole Derivatives for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. Patent CN111153847A introduces a groundbreaking methodology for the preparation of 4-(aryldiazenyl)-2,3-dihydropyrrole derivatives, a class of compounds with profound significance in medicinal chemistry and material science. This innovation leverages a novel three-component tandem reaction involving aziridines, aryl terminal alkynes, and aryl tetrafluoroborate diazonium salts to construct the dihydropyrrole core efficiently. Unlike traditional methods that often rely on precious or toxic transition metal catalysts, this disclosed process operates under metal-free conditions, utilizing mild thermal energy to drive the cascade transformation. For R&D directors and process chemists, this represents a pivotal shift towards cleaner synthesis, eliminating the regulatory burdens associated with residual heavy metals in active pharmaceutical ingredients (APIs). The reaction proceeds smoothly in common chlorinated solvents, offering a practical pathway for generating diverse libraries of azo-functionalized pyrrolines, which serve as critical intermediates for photochemical switches, antibacterial agents, and advanced dye precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydropyrrole and pyrroline skeletons has heavily depended on transition metal catalysis, presenting significant challenges for industrial scale-up and regulatory compliance. Prior art, such as the work by the Wender research group, utilized silver hexafluoroantimonate (AgSbF6) to catalyze [3+2] cycloadditions between aziridines and alkynes, while other methodologies employed iron(III) chloride (FeCl3) to achieve similar transformations. These metal-dependent pathways introduce severe complications in downstream processing, necessitating rigorous purification steps to reduce metal residues to parts-per-million (ppm) levels required by global health authorities like the FDA and EMA. Furthermore, the use of stoichiometric or catalytic amounts of expensive metals drastically inflates the raw material costs, impacting the overall economic viability of the manufacturing process. The sensitivity of these metal catalysts to moisture and oxygen often requires stringent anhydrous conditions and inert atmospheres, adding operational complexity and equipment costs. Additionally, the disposal of metal-contaminated waste streams poses environmental hazards, conflicting with the growing industry mandate for green chemistry and sustainable manufacturing practices.
The Novel Approach
The methodology described in CN111153847A circumvents these historical bottlenecks by employing a metal-free, thermal three-component tandem reaction that directly couples aziridines, alkynes, and diazonium salts. This approach not only simplifies the reaction setup by removing the need for specialized catalyst handling but also inherently improves the purity profile of the final product by eliminating metal contamination at the source. The reaction mechanism capitalizes on the intrinsic reactivity of the strained aziridine ring and the electrophilic nature of the diazonium species to drive the cyclization without external metallic promoters. As illustrated in the general reaction scheme below, the process tolerates a wide array of functional groups, allowing for the modular assembly of complex molecular architectures.
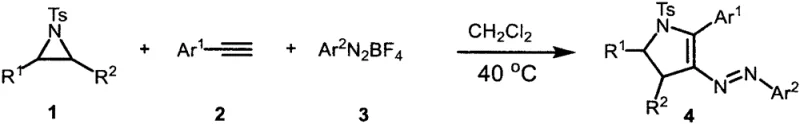
This versatility is crucial for medicinal chemists who require rapid access to structural analogs for structure-activity relationship (SAR) studies. By operating at mild temperatures between 20°C and 60°C, the process reduces energy consumption and minimizes the risk of thermal degradation of sensitive functional groups, such as esters and nitriles, which are often present in bioactive molecules. The use of stable and commercially available aryl tetrafluoroborate diazonium salts further enhances the practicality of this method, providing a reliable source of the azo moiety without the instability issues associated with free diazonium ions. Consequently, this novel approach offers a streamlined, cost-effective, and environmentally friendlier alternative to traditional metal-catalyzed routes, aligning perfectly with the strategic goals of modern pharmaceutical supply chains.
Mechanistic Insights into Metal-Free Three-Component Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated cascade that elegantly merges ring-opening chemistry with azo-coupling, driven entirely by thermal activation and electronic complementarity between the reactants. The reaction initiates with the nucleophilic attack of the electron-rich alkyne on the strained aziridine ring, facilitated by the polar environment of the solvent, leading to a ring-opened zwitterionic or radical intermediate depending on the specific electronic nature of the substituents. This intermediate subsequently undergoes an intramolecular cyclization to form the dihydropyrrole core, a step that is thermodynamically favored due to the relief of ring strain and the formation of a stable conjugated system. Concurrently or sequentially, the aryl tetrafluoroborate diazonium salt acts as an electrophilic partner, engaging with the newly formed pyrroline scaffold to install the aryldiazenyl group at the 4-position. This tandem sequence avoids the isolation of unstable intermediates, thereby maximizing atom economy and minimizing waste generation. The absence of metal catalysts suggests that the reaction relies on the inherent polarization of the carbon-nitrogen bonds within the aziridine and the high reactivity of the diazonium cation to overcome activation barriers.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. In metal-catalyzed reactions, side products often arise from metal-mediated homocoupling of alkynes or decomposition of the catalyst itself, creating complex impurity profiles that are difficult to separate. In contrast, the primary byproducts in this thermal process are likely limited to unreacted starting materials or simple hydrolysis products of the diazonium salt, which are generally easier to remove via standard aqueous workups or silica gel chromatography. The patent data indicates that the reaction tolerates various substituents on the aziridine nitrogen (such as the tosyl group) and the carbon backbone, suggesting that steric hindrance does not significantly impede the cyclization efficiency. This robustness ensures consistent batch-to-bquality, a critical parameter for supply chain reliability. Furthermore, the mild conditions prevent the formation of polymeric tars often seen in high-temperature thermal reactions, resulting in cleaner crude reaction mixtures and higher isolated yields, as evidenced by the successful synthesis of derivatives bearing electron-withdrawing cyano and chloro groups as well as electron-donating methoxy and methyl groups.
How to Synthesize 4-(Aryldiazenyl)-2,3-dihydropyrrole Efficiently
The operational protocol for this synthesis is designed for simplicity and reproducibility, making it highly accessible for both laboratory-scale discovery and pilot-plant production. The procedure typically involves charging the aziridine and the aryl tetrafluoroborate diazonium salt into a reaction vessel, followed by degassing and backfilling with inert gas to maintain an oxygen-free environment, which is crucial for stabilizing the reactive diazonium species. Subsequent addition of the solvent and alkyne initiates the reaction upon heating, with the temperature carefully controlled between 20°C and 60°C depending on the specific reactivity of the substrates. Detailed standardized synthesis steps are provided in the guide below to ensure optimal results.
- Charge aziridine and aryl tetrafluoroborate diazonium salt into a Schlenk tube, evacuate, and backfill with argon three times.
- Add solvent (DCM or DCE) and aryl terminal alkyne under argon atmosphere, seal the system, and heat to 20-60°C.
- Stir for 36-72 hours, dilute with dichloromethane, add triethylamine, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic benefits that extend beyond mere chemical novelty. The elimination of transition metal catalysts fundamentally alters the cost structure of the manufacturing process by removing the line item for expensive noble metals like silver or specialized Lewis acids like iron salts. More importantly, it obviates the need for costly downstream purification technologies, such as scavenger resins or specialized filtration units, which are mandatory for meeting strict heavy metal specifications in API production. This simplification of the workflow leads to a drastic reduction in processing time and utility consumption, directly enhancing the overall throughput of the manufacturing facility. The reliance on stable, shelf-stable solid reagents like aryl tetrafluoroborate diazonium salts mitigates the risks associated with the storage and handling of hazardous liquid reagents, improving workplace safety and reducing insurance and compliance costs. Moreover, the use of common solvents like dichloromethane and 1,2-dichloroethane ensures that sourcing is straightforward and not subject to the volatility of specialized chemical markets.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of metal catalysts, which eliminates the capital expenditure associated with metal recovery systems and the operational expenditure of purchasing high-purity catalytic grades. Without the need for rigorous metal scavenging steps, the number of unit operations in the production line is reduced, leading to lower labor costs and higher equipment utilization rates. The high atom economy of the three-component tandem reaction means that a larger proportion of the raw material mass is converted into the desired product, minimizing waste disposal fees and maximizing yield per kilogram of input. Additionally, the mild reaction conditions reduce energy demands for heating and cooling, contributing to a lower carbon footprint and reduced utility bills. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final pharmaceutical intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly de-risks the supply chain by utilizing widely available and stable starting materials. Aziridines and aryl terminal alkynes are commodity chemicals with established global supply networks, ensuring consistent availability even during market fluctuations. The stability of the aryl tetrafluoroborate diazonium salts allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to maintain strategic stockpiles and buffer against raw material shortages. The simplicity of the reaction conditions, which do not require exotic ligands or sensitive catalysts, reduces the dependency on single-source suppliers for specialized reagents. This diversification of the supply base enhances resilience against geopolitical disruptions or logistics bottlenecks. Furthermore, the high reproducibility of the metal-free process ensures consistent lead times, allowing procurement teams to plan inventory levels with greater confidence and reduce the need for safety stock.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden complexities, particularly with exothermic metal-catalyzed reactions that require precise thermal control. This metal-free thermal process, operating at moderate temperatures, presents a much flatter thermal profile, making it inherently safer and easier to scale to multi-ton production volumes. The absence of heavy metals simplifies the environmental permitting process, as wastewater and solid waste streams do not require specialized treatment for metal removal before discharge. This alignment with green chemistry principles not only reduces regulatory compliance costs but also enhances the corporate sustainability profile of the manufacturer, a key metric for modern ESG-focused investors and partners. The ability to run the reaction in standard glass-lined or stainless steel reactors without corrosion concerns from aggressive metal salts further facilitates rapid technology transfer and commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the primary advantages of this metal-free synthesis route?
A: The primary advantage is the complete elimination of transition metal catalysts such as silver or iron, which removes the need for expensive and complex heavy metal scavenging steps, thereby significantly reducing production costs and ensuring higher purity profiles suitable for pharmaceutical applications.
Q: What is the substrate scope for this three-component reaction?
A: The reaction demonstrates broad substrate tolerance, successfully accommodating aziridines with both aryl and alkyl substituents, as well as aryl terminal alkynes and diazonium salts bearing various electron-donating and electron-withdrawing groups like methoxy, chloro, cyano, and ester functionalities.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes mild reaction temperatures ranging from 20°C to 60°C and common organic solvents like dichloromethane or 1,2-dichloroethane, which facilitates easier thermal management and safer scale-up compared to harsh catalytic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Aryldiazenyl)-2,3-dihydropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in advancing the production of high-value pharmaceutical intermediates. Our team of expert process chemists has extensively evaluated the technology disclosed in CN111153847A and possesses the technical capability to adapt and optimize this three-component tandem reaction for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the absence of heavy metal residues, guaranteeing that every batch meets the highest international standards for API intermediates.
We invite you to collaborate with us to leverage this innovative chemistry for your next drug development program. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free route for your specific molecule. We encourage you to contact us today to discuss your requirements, obtain specific COA data for relevant analogs, and receive comprehensive route feasibility assessments tailored to your supply chain objectives. Let us help you secure a reliable, cost-effective, and sustainable supply of these critical heterocyclic building blocks.
