Advanced Synthesis of 6-Trifluoromethyl-1,3-Dihydroxyxanthone for Commercial Pharmaceutical Applications
Advanced Synthesis of 6-Trifluoromethyl-1,3-Dihydroxyxanthone for Commercial Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for novel scaffolds that offer enhanced biological activity and metabolic stability. In this context, patent CN107759558B introduces a significant advancement with the development of 6-trifluoromethyl-1,3-dihydroxyxanthone, a new xanthone derivative demonstrating potent antitumor and tyrosinase inhibitory properties. This compound represents a critical building block for the next generation of oncology therapeutics and cosmetic active ingredients, addressing the growing demand for high-purity fluorinated heterocycles. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the synthetic efficiency and biological potential of this molecule is paramount for strategic sourcing and pipeline development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for substituted xanthones often rely on multi-step sequences involving harsh Lewis acids, elevated temperatures, or expensive transition metal catalysts that complicate downstream processing. These conventional methodologies frequently suffer from poor atom economy and generate significant quantities of hazardous waste, posing challenges for environmental compliance and cost reduction in API manufacturing. Furthermore, the introduction of specific substituents, such as the trifluoromethyl group at the 6-position, can be notoriously difficult to control regioselectively using older techniques, leading to complex impurity profiles that require extensive purification efforts. Such inefficiencies not only inflate production costs but also extend lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates needed for clinical trials.
The Novel Approach
In contrast, the methodology disclosed in the patent utilizes a streamlined one-pot cyclization strategy employing phosphorus pentoxide dissolved in methanesulfonic acid as the reaction medium. This innovative approach allows for the direct condensation of 4-trifluoromethyl-2-hydroxybenzoic acid and phloroglucinol under remarkably mild conditions, specifically maintaining reaction temperatures between 88-92°C. By leveraging the dual functionality of the P2O5/MeSO3H system as both a dehydrating agent and a solvent, the process achieves high conversion rates with simplified operational parameters. 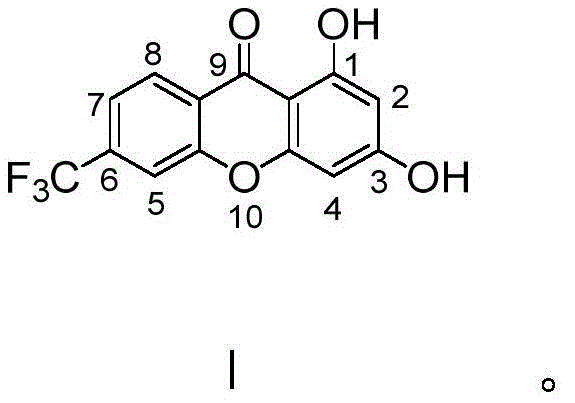
Mechanistic Insights into P2O5/MeSO3H Catalyzed Cyclization
The core of this synthetic breakthrough lies in the activation of the carboxylic acid moiety by the phosphorus pentoxide-methanesulfonic acid complex, which generates a highly reactive acylium ion intermediate in situ. This electrophilic species subsequently undergoes an intramolecular Friedel-Crafts acylation with the electron-rich phloroglucinol ring, facilitated by the strong acidic environment that enhances the nucleophilicity of the phenolic substrate. The presence of the trifluoromethyl group on the benzoic acid starting material exerts a strong electron-withdrawing effect, which necessitates precise control over the reaction kinetics to ensure successful ring closure without decomposition. The patent data indicates that temperature control is crucial; exceeding the optimal range can lead to carbonization of raw materials, while insufficient heat impedes the reaction rate, highlighting the delicate balance required for optimal yield.
From an impurity control perspective, the simplicity of the reagent system significantly reduces the risk of metal contamination, a common issue with transition-metal catalyzed cross-couplings often used in alternative xanthone syntheses. The use of methanesulfonic acid, an organic sulfonic acid, ensures that the reaction mixture remains homogeneous, promoting uniform heat transfer and minimizing localized hot spots that could generate side products. Following the reaction, the product can be easily precipitated by pouring the mixture into water, allowing for straightforward isolation via suction filtration. Subsequent purification via column chromatography using standard silica gel media effectively removes unreacted starting materials and minor byproducts, ensuring the final compound meets stringent purity specifications required for biological testing and potential therapeutic application.
How to Synthesize 6-Trifluoromethyl-1,3-Dihydroxyxanthone Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of sequential addition and thermal management. To achieve the reported yields, operators must first prepare the activating solution by dissolving phosphorus pentoxide in methanesulfonic acid at controlled temperatures before introducing the substrates. The detailed standardized synthesis steps below outline the precise molar ratios and temporal parameters necessary to replicate the high efficiency observed in the patent examples, ensuring reproducibility for process chemists.
- Dissolve phosphorus pentoxide in methanesulfonic acid at 105-115°C to create the activated reaction medium.
- Add 4-trifluoromethyl-2-hydroxybenzoic acid and phloroglucinol in a 1: 1 molar ratio and maintain temperature at 88-92°C for cyclization.
- Precipitate the product into water, followed by filtration and column chromatography purification to isolate the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost structure and logistical reliability. The reliance on commercially available starting materials, such as phloroglucinol and trifluoromethyl-substituted benzoic acids, mitigates the risk of raw material shortages that often plague specialized fine chemical supply chains. Moreover, the elimination of expensive noble metal catalysts and the reduction of reaction steps directly contribute to substantial cost savings in the overall manufacturing process, making the final intermediate more economically viable for large-scale drug development programs.
- Cost Reduction in Manufacturing: The process eliminates the need for costly transition metal catalysts and complex ligand systems, which traditionally drive up the bill of materials for heterocyclic synthesis. By utilizing inexpensive inorganic dehydrating agents and organic acids, the direct material costs are significantly lowered, while the simplified workup procedure reduces labor and utility consumption associated with prolonged purification cycles. This economic efficiency allows for more competitive pricing models when sourcing this key pharmaceutical intermediate for downstream API production.
- Enhanced Supply Chain Reliability: Since the synthesis relies on commodity chemicals rather than bespoke reagents with long lead times, the risk of supply disruption is drastically minimized. The robustness of the reaction conditions means that production can be scaled up rapidly to meet fluctuating demand without requiring specialized equipment or exotic infrastructure. This flexibility ensures a continuous supply of high-quality intermediates, supporting uninterrupted clinical trial timelines and commercial manufacturing schedules for partner organizations.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of volatile organic solvents during the cyclization step align well with green chemistry principles, reducing the environmental footprint of the manufacturing process. The ability to precipitate the product directly from the reaction mixture simplifies waste treatment and solvent recovery, facilitating easier compliance with increasingly stringent environmental regulations. This scalability ensures that the transition from gram-scale laboratory synthesis to multi-ton commercial production can be achieved with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-trifluoromethyl-1,3-dihydroxyxanthone. These insights are derived directly from the technical disclosures in patent CN107759558B, providing clarity on the compound's capabilities and the efficiency of its manufacturing process for potential industry partners.
Q: What are the primary biological activities of 6-trifluoromethyl-1,3-dihydroxyxanthone?
A: According to patent CN107759558B, this compound exhibits significant antitumor activity against various cell lines including HL-60 and A-549, alongside tyrosinase inhibitory properties useful for cosmetic and metabolic applications.
Q: Why is the methanesulfonic acid/P2O5 system preferred for this synthesis?
A: This system acts as both a potent dehydrating agent and a solvent, facilitating efficient cyclization under mild temperatures (around 90°C) while avoiding the harsh conditions often required in traditional xanthone synthesis.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly states the method features simple operation and mild conditions suitable for large-scale preparation, making it viable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Trifluoromethyl-1,3-Dihydroxyxanthone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-trifluoromethyl-1,3-dihydroxyxanthone delivered meets the highest international standards for pharmaceutical applications.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our optimized synthesis routes can support your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into value for your organization. Reach out today to obtain specific COA data and route feasibility assessments tailored to your project's unique demands.
