Advanced Synthesis of 13-Hydroxy Cytisine Cinnamates for Anti-Tumor Drug Development
The pharmaceutical landscape is continuously evolving with the discovery of novel hybrid molecules that combine the pharmacophores of distinct bioactive natural products. A significant breakthrough in this domain is detailed in patent CN108840871B, which discloses a series of 13-hydroxy cytisine cinnamate compounds exhibiting potent anti-tumor activity. This technology represents a strategic convergence of alkaloid chemistry and phenylpropanoid biology, creating a new class of therapeutic candidates designed to overcome multi-drug resistance in tumor cells. By esterifying the 13-hydroxyl position of cytisine—a natural alkaloid with a rich history of biological utility—with various cinnamic acid derivatives, researchers have generated a library of compounds that leverage the antioxidant and kinase-modulating properties of cinnamates. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the synthetic accessibility and structural versatility of these molecules is paramount for future drug development pipelines.
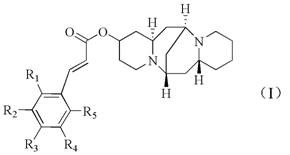
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of bioactive alkaloids like cytisine and its derivatives has relied heavily on direct extraction from plant sources such as Sophora alopecuroides or Laburnum anagyroides. While nature provides a starting point, the isolation of specific monomers like 13-hydroxy cytisine is often plagued by extremely low natural abundance, leading to prohibitive costs and supply chain instability. Furthermore, simple extraction does not allow for the structural diversification necessary to optimize pharmacokinetic profiles or enhance specific biological activities like tyrosinase inhibition or extracellular regulatory protein kinase activation. Conventional chemical modifications of alkaloids often suffer from harsh reaction conditions, poor regioselectivity, or the use of toxic heavy metal catalysts that complicate downstream purification and regulatory approval. These limitations create a bottleneck in the commercial scale-up of complex pharmaceutical intermediates, restricting the ability of drug developers to explore structure-activity relationships (SAR) effectively.
The Novel Approach
The methodology outlined in the patent introduces a streamlined esterification strategy that circumvents these historical challenges by utilizing readily available raw materials and mild reaction conditions. Instead of struggling with low-yield extractions or dangerous reagents, the novel approach employs a condensation reaction between 13-hydroxy cytisine and cinnamic acid derivatives in the presence of organic solvents and standard condensing agents. This route allows for the precise installation of diverse functional groups—such as hydroxyl, methoxy, halogen, or alkyl substituents—on the cinnamate aromatic ring, thereby tuning the electronic and steric properties of the final molecule. The result is a robust synthetic platform that facilitates the rapid generation of a focused library of analogs, significantly accelerating the lead optimization phase. This shift from extraction-dependence to semi-synthetic flexibility is a critical advancement for cost reduction in pharmaceutical intermediate manufacturing, offering a predictable and scalable supply of high-value candidates.
Mechanistic Insights into DCC-Mediated Esterification
The core of this synthetic innovation lies in the efficient formation of the ester bond at the C-13 position of the cytisine scaffold. The reaction mechanism typically involves the activation of the carboxylic acid group of the cinnamic acid derivative by a condensing agent such as Dicyclohexylcarbodiimide (DCC) or 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC). This activation generates a highly reactive O-acylisourea intermediate, which is subsequently attacked by the nucleophilic hydroxyl group at the 13th position of the 13-hydroxy cytisine. To further enhance the reaction rate and yield, a nucleophilic catalyst like 4-Dimethylaminopyridine (4-DMAP) is often employed. 4-DMAP acts as an acyl transfer agent, forming an even more reactive acylpyridinium species that facilitates the attack by the sterically hindered alcohol of the alkaloid. This catalytic cycle ensures high conversion rates under relatively mild thermal conditions, typically ranging from 60°C to 100°C, preserving the integrity of the sensitive alkaloid backbone while driving the equilibrium toward the desired ester product.
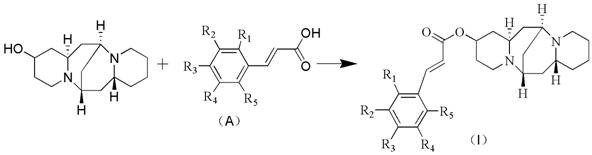
From an impurity control perspective, this mechanism offers distinct advantages over alternative coupling methods. The use of carbodiimide-based coupling avoids the introduction of transition metals, which are notoriously difficult to remove to ppm levels required by ICH Q3D guidelines. The primary byproduct, dicyclohexylurea (DCU) in the case of DCC, is generally insoluble in many organic solvents and can be removed via simple filtration, simplifying the workup procedure. Furthermore, the regioselectivity of the reaction is inherently controlled by the unique reactivity of the C-13 hydroxyl group compared to the tertiary amines present in the cytisine structure, minimizing the formation of quaternary ammonium salts or N-acylated side products. This inherent chemoselectivity reduces the burden on chromatographic purification, leading to higher overall process efficiency and cleaner crude profiles, which is essential for maintaining high-purity pharmaceutical intermediates suitable for preclinical safety studies.
How to Synthesize 13-Hydroxy Cytisine Cinnamate Efficiently
Executing this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize yield and minimize waste. The general protocol involves dissolving the 13-hydroxy cytisine and the chosen cinnamic acid derivative in a suitable organic solvent such as toluene, DMF, or acetonitrile. The molar ratio is typically adjusted to a slight excess of the acid component (1:1 to 1:1.3) to drive the reaction to completion. Following the addition of the catalyst and condensing agent, the mixture is heated under reflux for a period ranging from 4 to 12 hours, depending on the specific electronic nature of the substituents on the cinnamic acid ring. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided in the technical guide below.
- Charge 13-hydroxycytisine and cinnamic acid derivatives into a reactor with a molar ratio of 1: 1 to 1:1.3.
- Add organic solvent such as toluene or DMF, along with catalyst 4-DMAP and condensing agent DCC.
- Reflux the mixture at 60-100°C for 4-12 hours, followed by workup involving washing, drying, and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers tangible logistical and financial benefits that extend beyond simple yield metrics. The reliance on commodity chemicals like cinnamic acid derivatives and established alkaloid precursors ensures a stable supply base that is less susceptible to the seasonal and geopolitical fluctuations often associated with botanical extraction. Moreover, the simplicity of the reaction setup—requiring only standard glass-lined or stainless steel reactors capable of heating and stirring—means that production can be easily transferred to existing multipurpose facilities without significant capital expenditure on specialized equipment. This operational flexibility translates directly into reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond more agilely to clinical trial demands.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts (such as palladium or platinum) removes the need for costly scavenging resins and extensive metal testing, which are significant cost drivers in modern API synthesis. Additionally, the use of recyclable solvents like toluene and the ability to filter off solid urea byproducts simplifies the downstream processing train. By avoiding complex chromatographic separations and reducing the number of unit operations, the overall cost of goods sold (COGS) is significantly optimized, making the commercial production of these anti-tumor candidates economically viable.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, including various substituted cinnamic acids and cytisine scaffolds, are commercially available from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the esterification reaction, which tolerates a wide range of functional groups without requiring protecting group strategies, further streamlines the supply chain by reducing the number of intermediate steps. This simplicity ensures a consistent and reliable flow of material, crucial for maintaining uninterrupted clinical supply chains and meeting strict delivery timelines for partner organizations.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, presenting minimal safety risks related to exotherms or high-pressure containment. From an environmental standpoint, the absence of heavy metals and the potential for solvent recovery align with green chemistry principles, facilitating easier regulatory compliance and waste management. The scalability of the reflux condensation method is well-proven in the fine chemical industry, allowing for seamless translation from laboratory benchtop experiments to multi-ton annual production capacities without loss of efficiency or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel compounds. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, aiming to clarify the feasibility and value proposition of this technology for potential licensees and manufacturing partners.
Q: What represents the primary structural innovation in these compounds?
A: The innovation lies in the ester linkage between the 13-hydroxyl group of cytisine and various cinnamic acid derivatives, creating a synergistic anti-tumor effect.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reflux temperatures (60-100°C) and common organic solvents like toluene, making it highly adaptable for industrial scale-up without specialized high-pressure equipment.
Q: How does this synthesis method improve upon traditional extraction?
A: Unlike extraction which yields low quantities of natural monomers, this semi-synthetic esterification allows for the production of diverse derivatives with enhanced biological activity and consistent purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 13-Hydroxy Cytisine Cinnamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the 13-hydroxy cytisine cinnamate scaffold in the fight against resistant tumors. As a premier CDMO partner, we possess the technical expertise to translate the innovative pathways described in patent CN108840871B into reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from early-stage development to full-scale manufacturing. We maintain stringent purity specifications across all our lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timeline. Our technical team is ready to perform a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments for this promising class of anti-tumor intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
