Advanced Synthetic Route for Avanafil: Enhancing Purity and Scalability for Global Pharma Supply Chains
Advanced Synthetic Route for Avanafil: Enhancing Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical landscape for erectile dysfunction treatments continues to evolve, driven by the demand for faster-acting agents with superior safety profiles. At the forefront of this sector is Avanafil, a highly selective phosphodiesterase-5 (PDE5) inhibitor known for its rapid onset of action. A pivotal development in the manufacturing of this active pharmaceutical ingredient is detailed in Chinese patent CN109280050B, which discloses a robust preparation method designed to overcome the limitations of prior art. This technical insight report analyzes the strategic advantages of this novel synthetic pathway, specifically focusing on its potential to serve as a reliable avanafil supplier solution for global markets. By shifting the critical oxidation step to the beginning of the synthesis, the methodology significantly mitigates impurity risks associated with late-stage functional group transformations. For R&D directors and procurement specialists, understanding this mechanistic shift is crucial for evaluating long-term supply chain stability and cost efficiency in pharmaceutical intermediates manufacturing.
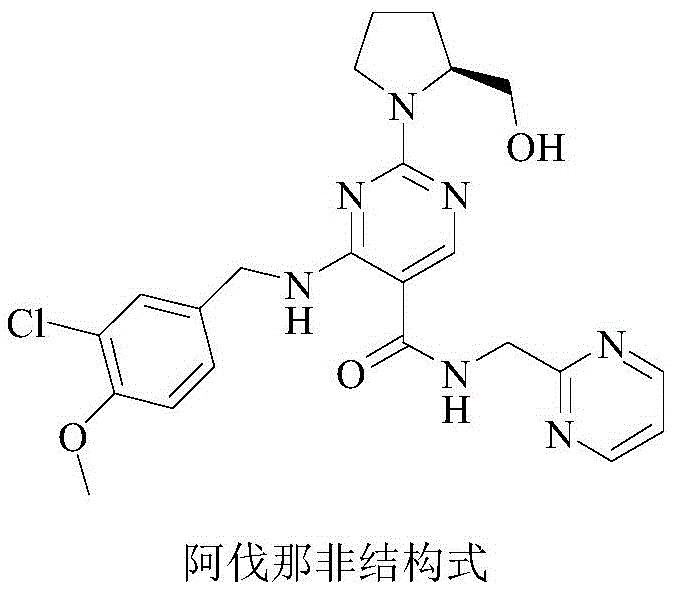
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Avanafil has relied on routes where the oxidation of the sulfur-containing moiety occurs at a later stage of the molecular assembly. In traditional pathways, such as those disclosed in international patent WO 2001019802A1, the methylthio group is oxidized after the initial condensation with the benzylamine fragment. This sequence presents significant chemical challenges, primarily because the intermediate containing the nitrogen heteroatom is susceptible to over-oxidation or incomplete oxidation. Consequently, the reaction mixture often contains a complex array of impurities, including sulfoxides and sulfones, which are structurally similar to the desired product. Separating these oxidation by-products requires rigorous purification protocols, often involving multiple recrystallizations or chromatographic steps, which drastically reduces overall yield and increases production costs. Furthermore, the presence of these impurities can interfere with subsequent coupling reactions, leading to inconsistent batch quality and potential regulatory hurdles during drug substance registration.
The Novel Approach
The methodology outlined in patent CN109280050B introduces a paradigm shift by executing the oxidation of the methylthio group to the sulfone at the very inception of the synthetic route. This strategic reordering of steps ensures that the highly reactive sulfone functionality is established on a simpler precursor before the introduction of complex amine side chains. By utilizing hydrogen peroxide as a green oxidant in a high-pressure reactor, the process achieves high conversion to the sulfone intermediate with minimal by-product formation. This early-stage purification simplifies the downstream processing significantly, as the subsequent coupling reactions proceed with greater selectivity and cleanliness. The result is a streamlined workflow that not only enhances the purity of the final API but also reduces the burden on quality control laboratories. For supply chain heads, this translates to a more predictable manufacturing timeline and reduced risk of batch failures due to impurity spikes.
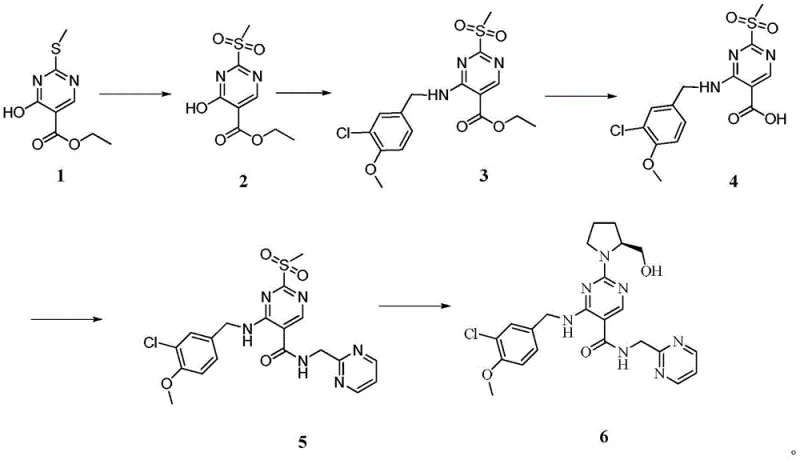
Mechanistic Insights into the Optimized Five-Step Synthesis
The core of this technological advancement lies in the precise control of reaction conditions across five distinct transformation stages. The process initiates with the oxidation of ethyl 4-hydroxy-2-(methylthio)pyrimidine-5-carboxylate (Compound 1) using 30% hydrogen peroxide at 120°C. This step converts the thioether into a sulfone (Compound 2), activating the C2 position of the pyrimidine ring for nucleophilic attack. Following this, the protocol employs phosphorus oxychloride to convert the hydroxyl group into a chloro leaving group, which is immediately displaced by 3-chloro-4-methoxybenzylamine in the presence of diisopropylethylamine. This tandem chlorination-amination sequence is critical, as it builds the core scaffold of the molecule while maintaining the integrity of the newly formed sulfone group. The use of toluene as a solvent in this phase facilitates easy phase separation and removal of inorganic salts, contributing to the high purity observed in Compound 3.
Subsequent steps focus on constructing the amide linkage and installing the chiral pyrrolidine ring. The ester moiety of Compound 3 is hydrolyzed under mild alkaline conditions using sodium hydroxide in DMF, followed by acidification to yield the carboxylic acid (Compound 4). This acid is then activated using thionyl chloride to form an acyl chloride species in situ, which reacts efficiently with 2-aminomethyl pyrimidine to form the amide bond in Compound 5. The final and perhaps most stereochemically sensitive step involves the nucleophilic displacement of the sulfone group by L-prolinol. Conducted in dichloromethane at controlled temperatures between 0°C and 50°C, this substitution installs the chiral hydroxymethyl-pyrrolidine side chain with high fidelity. The mechanistic elegance of this route lies in its modularity; each step generates a stable, isolable intermediate, allowing for rigorous quality checks before proceeding to the next stage, thereby ensuring the final product meets stringent purity specifications.
How to Synthesize Avanafil Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control, particularly during the exothermic oxidation and chlorination phases. The patent provides detailed experimental parameters, including specific molar ratios such as 1:1.1 for the oxidant and 1:1.01-1.05 for the amine coupling partners, which are optimized to minimize waste while maximizing yield. Operators must ensure that the hydrolysis step is kept below 40°C to prevent degradation of the sensitive pyrimidine core. The detailed standardized synthesis steps, including precise reagent quantities and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency process.
- Oxidize the starting pyrimidine ester with hydrogen peroxide in a high-pressure reactor to form the sulfone intermediate.
- Perform chlorination using phosphorus oxychloride followed by condensation with 3-chloro-4-methoxybenzylamine.
- Hydrolyze the ester group using sodium hydroxide, acidify, and isolate the carboxylic acid intermediate.
- Activate the acid with thionyl chloride and couple with 2-aminomethyl pyrimidine to form the amide bond.
- Execute the final nucleophilic substitution with L-prolinol to displace the sulfone group and yield Avanafil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route offers compelling economic and operational benefits that extend beyond simple yield improvements. The primary advantage stems from the elimination of late-stage oxidation, which traditionally acts as a bottleneck in Avanafil production due to the difficulty in purifying oxidation by-products. By moving this step to the beginning, the process removes the need for expensive and time-consuming purification technologies such as preparative HPLC or extensive column chromatography at the final stages. This simplification directly correlates to a significant reduction in manufacturing costs, as it lowers solvent consumption, reduces labor hours, and minimizes the loss of valuable intermediates during cleanup. Furthermore, the use of commodity chemicals like hydrogen peroxide and thionyl chloride ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from volatility associated with specialized reagents.
- Cost Reduction in Manufacturing: The streamlined nature of this five-step sequence inherently lowers the cost of goods sold (COGS) by reducing the total number of unit operations required. Since the oxidation is performed on a low-molecular-weight starting material, the volume of oxidant required is substantially lower compared to oxidizing a heavy, complex intermediate. Additionally, the high yields reported in the patent examples, often exceeding 85-90% per step, mean that less starting material is needed to produce a kilogram of final API. This efficiency gain eliminates the need for oversized reactors to compensate for poor yields, allowing manufacturers to maximize the throughput of existing infrastructure. The cumulative effect is a drastic simplification of the cost structure, making the final product more competitive in price-sensitive generic markets.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that rely on finicky reaction conditions or hard-to-source catalysts. This route utilizes robust, well-understood chemical transformations that are easily scalable from pilot plant to commercial tonnage. The reliance on standard solvents like toluene, DMF, and dichloromethane means that procurement teams can leverage existing vendor relationships without needing to qualify new, exotic supply lines. Moreover, the stability of the intermediates allows for the potential creation of strategic stockpiles at various stages of the synthesis. If demand fluctuates, manufacturers can hold inventory at the stable acid or amide stage and complete the final steps just-in-time, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process demonstrates a commitment to greener chemistry principles. The replacement of hazardous oxidants like m-chloroperoxybenzoic acid (mCPBA) with hydrogen peroxide significantly reduces the generation of chlorinated organic waste and explosive peroxide residues. Hydrogen peroxide decomposes into water and oxygen, presenting a much lower environmental burden. The ability to run the oxidation in water or with minimal organic co-solvents further aligns with modern sustainability goals. For facilities operating under strict environmental regulations, this route offers a clearer path to compliance, reducing the costs associated with waste treatment and disposal. The scalability is further enhanced by the fact that the reaction conditions, such as the 120°C oxidation temperature, are well within the capabilities of standard stainless steel reactors, facilitating a smooth transition from laboratory benchtop to industrial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs.
Q: How does this new route improve impurity profiles compared to conventional methods?
A: By performing the oxidation of the methylthio group to the sulfone at the very beginning of the synthesis (Step 1), rather than on a complex intermediate later in the sequence, the process avoids the formation of difficult-to-remove sulfoxide/sulfone mixtures that typically plague late-stage oxidation steps.
Q: What are the key reagents used in the critical coupling steps?
A: The process utilizes phosphorus oxychloride for activation in the second step and thionyl chloride for amide bond formation in the fourth step, both standard industrial reagents that ensure high conversion rates and manageable waste streams.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the use of common solvents like toluene and dichloromethane, along with mild reaction temperatures and high yields across all five steps, making it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Avanafil Supplier
The technical superiority of the synthesis route described in CN109280050B underscores the importance of partnering with a manufacturer who possesses both the chemical expertise and the infrastructure to execute it flawlessly. NINGBO INNO PHARMCHEM stands ready to leverage this advanced methodology to deliver high-quality Avanafil intermediates and API to the global market. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-volume manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the highest international standards for pharmaceutical ingredients.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient synthesis method. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition to a more reliable and cost-effective source of Avanafil.
