Revolutionizing SGLT2 Inhibitor Synthesis with Advanced Halogenated Pivaloyl Glucopyranose Technology
Introduction to Next-Generation SGLT2 Inhibitor Synthesis
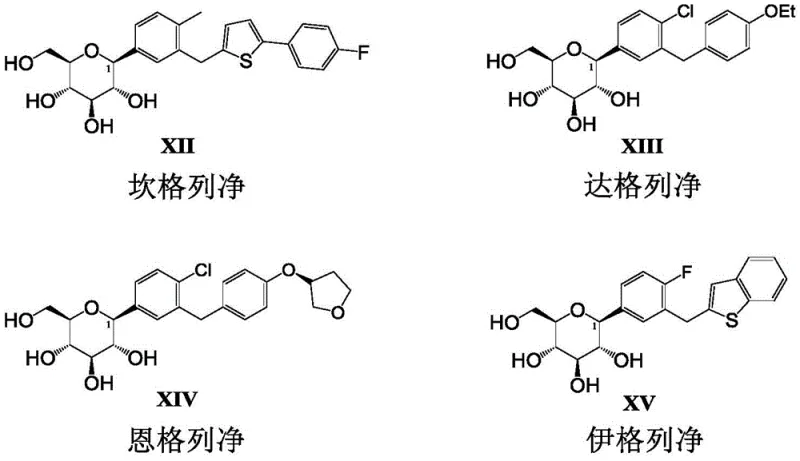
The pharmaceutical landscape for Type 2 diabetes treatment has been significantly shaped by Sodium-Glucose Cotransporter 2 (SGLT2) inhibitors, a class of drugs that includes blockbuster molecules such as Canagliflozin, Dapagliflozin, Empagliflozin, and Ipragliflozin. As demand for these therapeutics grows, the industry faces increasing pressure to optimize synthetic routes for better purity, yield, and cost-efficiency. Patent CN109111490B presents a groundbreaking advancement in this domain by introducing a novel class of intermediates: 1,2,3,4,6-penta-O-halogenated pivaloyl glucopyranose and their derivatives. This technology addresses critical bottlenecks in existing manufacturing processes, offering a robust platform for the production of beta-C-aryl glucosides. By leveraging halogenated pivaloyl protecting groups, this method achieves superior stereoselectivity and operational simplicity, making it an ideal candidate for reliable SGLT2 inhibitor intermediate supplier partnerships aiming for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
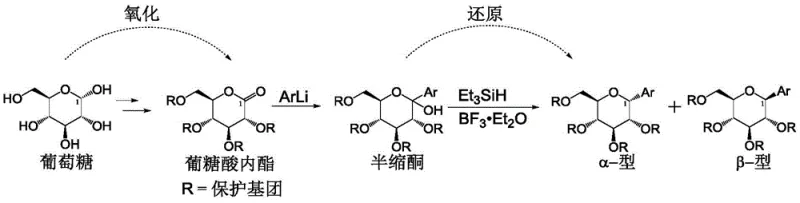
Historically, the synthesis of beta-C-aryl glucosides has relied heavily on the gluconolactone method, a pathway that, while effective, suffers from inherent inefficiencies. This classical approach typically involves the oxidation of protected glucose to gluconolactone, followed by the addition of an aryl lithium derivative to form a hemiketal, and finally, a reduction step using triethylsilane and boron trifluoride etherate. A major drawback of this sequence is its poor redox economy; the carbon atom at the C1 position undergoes unnecessary oxidation and subsequent reduction to reach the final oxidation state, leading to increased reagent costs and waste generation. Furthermore, the lack of stereospecificity in the reduction step often results in a mixture of desired beta-anomers and undesired alpha-anomers, necessitating costly purification processes to meet stringent pharmaceutical purity standards.
The Novel Approach
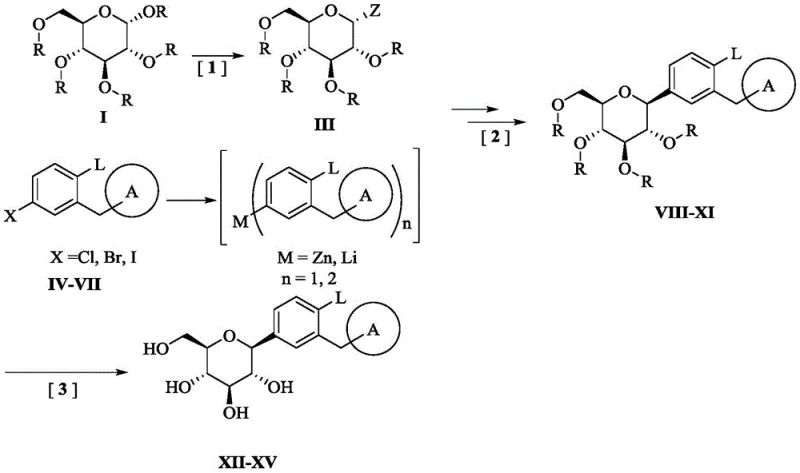
In stark contrast, the methodology disclosed in CN109111490B circumvents these redox inefficiencies by utilizing a direct nucleophilic substitution strategy. The core innovation lies in the use of 2,3,4,6-tetra-O-halogenated pivaloyl-alpha-D-halogenated glucopyranose as a key electrophilic intermediate. By employing bulky halogenated pivaloyl groups as protecting moieties, the new route enhances steric hindrance around the sugar ring. This steric bulk not only stabilizes the intermediate but also critically influences the stereochemical outcome of the glycosylation reaction, favoring the formation of the beta-configuration with high selectivity. This approach simplifies the synthetic sequence by eliminating the oxidation-reduction cycle, thereby streamlining the process flow and significantly improving the overall atom economy for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Stereoselective Nucleophilic Substitution
The mechanistic superiority of this halogenated pivaloyl route is rooted in the interplay between the protecting group sterics and the organometallic nucleophile. The synthesis begins with the preparation of an organozinc or organolithium reagent from a substituted methyl phenyl halide. This metallated species then attacks the anomeric carbon of the halogenated glucose intermediate. The presence of the bulky halogenated pivaloyl groups at the 2, 3, 4, and 6 positions creates a shielded environment that directs the incoming nucleophile to attack from the less hindered face, effectively locking in the beta-stereochemistry. This intrinsic stereocontrol minimizes the formation of alpha-isomer impurities, which are notoriously difficult to separate due to their similar physical properties. Consequently, the process yields intermediates with high diastereomeric purity, reducing the burden on downstream purification units.
Furthermore, the stability of the halogenated pivaloyl protecting groups allows the arylation reaction to be conducted at elevated temperatures, typically ranging from 25°C to 145°C, depending on the specific substrate reactivity. This thermal robustness contrasts sharply with the cryogenic conditions often required for sensitive organolithium additions in traditional methods. The ability to operate at higher temperatures enhances reaction kinetics and conversion rates, leading to shorter reaction times and improved throughput. Additionally, the deprotection step is facilitated by mild basic conditions, where the ester linkages are cleaved efficiently to reveal the free hydroxyl groups of the final SGLT2 inhibitor. This combination of robust intermediate stability and mild deprotection conditions ensures a high-yielding process that is amenable to rigorous quality control protocols.
How to Synthesize High-Purity SGLT2 Inhibitor Intermediates Efficiently
The implementation of this technology involves a streamlined three-stage protocol designed for maximum efficiency and reproducibility. First, the organometallic reagent is generated in situ by reacting a specific aryl halide with a lithium or Grignard reagent in an inert solvent such as toluene or tetrahydrofuran at controlled low temperatures. Second, this reactive species is coupled with the halogenated glucopyranose intermediate under heated conditions to form the protected C-aryl glucoside. Finally, the protecting groups are removed via alkaline hydrolysis to yield the target molecule. This logical progression minimizes unit operations and handling steps, which is crucial for maintaining product integrity. For detailed standard operating procedures and specific reaction parameters, please refer to the comprehensive guide below.
- Prepare an organozinc or organolithium reagent by reacting a substituted methyl phenyl halide with a lithium reagent or Grignard composite at low temperatures (-40 to 20°C).
- Perform nucleophilic substitution by reacting the organometallic species with 2,3,4,6-tetra-O-halogenated pivaloyl-alpha-D-halogenated glucopyranose at elevated temperatures (25-145°C).
- Remove the halogenated pivaloyl protecting groups using an organic or inorganic base in a solvent system to yield the final high-purity SGLT2 inhibitor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this halogenated pivaloyl technology translates into tangible strategic benefits beyond mere chemical elegance. The elimination of redox steps directly correlates to a reduction in raw material consumption and waste disposal costs, addressing the growing imperative for sustainable and cost-effective manufacturing. By simplifying the synthetic route, manufacturers can reduce the number of isolation and purification stages, which in turn shortens the overall production cycle time. This efficiency gain enhances supply chain reliability, ensuring consistent availability of critical intermediates even during periods of high market demand. Moreover, the high stereoselectivity reduces the risk of batch failures due to impurity profiles, safeguarding against costly delays in regulatory filings and commercial launches.
- Cost Reduction in Manufacturing: The removal of oxidation and reduction reagents significantly lowers the bill of materials. Traditional routes require expensive oxidants and silane reducing agents, along with the associated catalysts and scavengers. By bypassing these steps, the new method achieves substantial cost savings through reduced reagent inventory and simplified waste management protocols. The higher yields reported in the patent data further amplify these savings by maximizing the output per kilogram of starting glucose, effectively lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The robustness of the halogenated pivaloyl intermediates allows for more flexible storage and handling compared to sensitive lactols or epoxides used in alternative methods. This stability reduces the risk of degradation during transport and warehousing, ensuring that quality is maintained from the supplier to the manufacturing site. Additionally, the use of common industrial solvents like toluene and methanol facilitates easier sourcing and recycling, mitigating supply risks associated with specialized or hazardous chemicals.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, featuring reaction conditions that are easily scalable from pilot plant to multi-ton commercial batches. The avoidance of hazardous reducing agents that generate flammable hydrogen gas improves plant safety profiles and reduces the need for specialized explosion-proof equipment. Furthermore, the improved atom economy aligns with green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability goals without compromising on production volume.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding the practical application and benefits of the halogenated pivaloyl method. These insights are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making regarding technology transfer and vendor qualification.
Q: How does the halogenated pivaloyl method improve stereoselectivity compared to the gluconolactone route?
A: The use of bulky halogenated pivaloyl protecting groups creates significant steric hindrance, which directs the nucleophilic attack to favor the formation of the desired beta-C-aryl glucoside anomer, minimizing alpha-isomer impurities without requiring complex separation steps.
Q: What are the primary cost drivers eliminated in this new synthetic pathway?
A: This method eliminates the redox economy issues of traditional routes by avoiding the oxidation of glucose to gluconolactone and subsequent reduction steps, thereby reducing reagent consumption, energy usage, and waste generation associated with redox byproducts.
Q: Is this process suitable for large-scale commercial production of Canagliflozin and Dapagliflozin?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to its operational simplicity, high yields (exceeding 90% in deprotection steps), and the use of robust intermediates that facilitate scalable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SGLT2 Inhibitor Intermediate Supplier
As the global demand for diabetes therapeutics continues to surge, securing a stable supply of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic technologies like the halogenated pivaloyl route to deliver exceptional value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Canagliflozin, Dapagliflozin, or Empagliflozin intermediate meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our innovative processes can drive efficiency and reliability in your SGLT2 inhibitor projects.
