Advanced Manufacturing of 2-Amino-5-Chlorobenzamide for Global Pharmaceutical Supply Chains
The global demand for high-quality heterocyclic intermediates continues to surge, driven by the expanding pipeline of kinase inhibitors and agrochemical active ingredients. At the forefront of this chemical evolution is the efficient synthesis of 2-amino-5-chlorobenzamide, a pivotal building block for quinazoline derivatives. Patent CN101575301A introduces a transformative two-step methodology that fundamentally alters the economic and safety landscape of producing this critical intermediate. By shifting away from hazardous chlorinating agents toward a benign sodium hypochlorite-based system, this technology addresses the longstanding pain points of toxicity and waste generation inherent in legacy processes. For R&D leaders and procurement strategists, understanding this shift is essential for securing a resilient supply chain capable of meeting stringent regulatory standards while maintaining cost competitiveness in a volatile market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of chlorinated aniline derivatives has relied heavily on aggressive reagents such as sulfuryl chloride (SO2Cl2) and sulfur dichloride (SCl2). These traditional pathways are fraught with significant operational hazards, including the release of corrosive gases that necessitate specialized, expensive containment infrastructure. The use of ether-based solvents in older literature further exacerbates safety concerns due to flammability and peroxide formation risks. Moreover, these conventional routes often suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. The cumulative effect of these factors is a process with high variable costs, substantial environmental liability, and inconsistent batch-to-batch quality, making it increasingly untenable for modern, compliance-driven manufacturing facilities seeking long-term viability.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a sophisticated yet operationally simple chlorination strategy employing sodium hypochlorite solution and glacial acetic acid. This system operates under mild conditions, specifically maintaining temperatures below -5°C to drive the reaction kinetics toward the desired 5-position substitution with exceptional precision. The subsequent ammonolysis step leverages high-pressure autoclave technology to convert the ester intermediate directly into the amide, bypassing the need for harsh activation steps. This streamlined approach not only eliminates the handling of toxic sulfur-based reagents but also facilitates solvent recycling, drastically reducing the environmental footprint. The result is a robust, scalable process that delivers yields exceeding 85%, providing a clear competitive advantage in both efficiency and sustainability metrics.
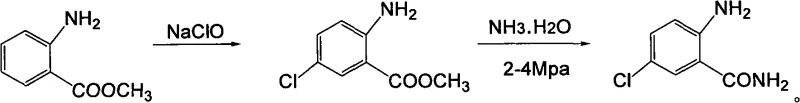
Mechanistic Insights into NaClO-Mediated Electrophilic Substitution
The core innovation of this synthesis lies in the controlled generation of the electrophilic chlorinating species within an acidic aqueous-organic biphasic system. When sodium hypochlorite interacts with glacial acetic acid, it generates hypochlorous acid in situ, which acts as the active chlorinating agent. The presence of the electron-donating amino group on the aromatic ring activates the substrate toward electrophilic aromatic substitution, directing the incoming chlorine primarily to the ortho and para positions. However, steric hindrance at the ortho position adjacent to the ester group, combined with the specific reaction conditions, favors substitution at the 5-position (para to the amino group). Maintaining the reaction temperature strictly below -5°C is mechanistically crucial; it suppresses the formation of polychlorinated byproducts and prevents the oxidation of the sensitive amine functionality, which is a common side reaction in uncontrolled hypochlorite oxidations.
Following the chlorination, the conversion of the methyl ester to the primary amide proceeds via a nucleophilic acyl substitution mechanism under high pressure. In this step, ammonia acts as a potent nucleophile, attacking the carbonyl carbon of the ester to form a tetrahedral intermediate. The application of elevated pressure (2-4 MPa) and temperature (50-200°C) shifts the equilibrium toward the amide product by increasing the effective concentration of ammonia in the liquid phase and overcoming the activation energy barrier. This high-pressure environment ensures complete conversion without the need for excessive molar equivalents of ammonia, which would complicate downstream recovery. The final purification via activated carbon treatment effectively removes trace colored impurities and residual organic byproducts, ensuring the final crystalline product meets the rigorous purity specifications required for pharmaceutical applications.
How to Synthesize 2-Amino-5-Chlorobenzamide Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity profiles. The process begins with the careful preparation of the chlorination mixture, where the ratio of organic solvent to water and the concentration of the hypochlorite solution must be strictly adhered to. Detailed standard operating procedures for temperature ramping, phase separation, and high-pressure reactor management are critical for successful scale-up. Operators must be trained to handle the exothermic nature of the initial mixing and the pressure dynamics of the ammonolysis step. For a comprehensive breakdown of the specific stoichiometric ratios, solvent choices, and workup protocols validated in the patent examples, please refer to the standardized synthesis guide provided below.
- Perform selective 5-position chlorination of methyl anthranilate using sodium hypochlorite and glacial acetic acid at temperatures below -5°C to ensure regioselectivity and minimize side reactions.
- Separate the organic phase containing methyl 2-amino-5-chlorobenzoate, dry thoroughly, and prepare for the subsequent ammonolysis reaction.
- Conduct high-pressure ammonolysis in an autoclave at 2-4 MPa and elevated temperatures, followed by recrystallization and activated carbon treatment to achieve final purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing route offers profound advantages that extend beyond simple yield improvements. The substitution of expensive, specialty chlorinating agents with commodity-grade sodium hypochlorite fundamentally alters the cost structure of the raw material basket. This shift mitigates exposure to price volatility associated with sulfur-based chemicals and reduces the logistical burden of transporting hazardous materials. Furthermore, the simplicity of the workup procedure, which avoids complex distillation or chromatographic separations, translates directly into reduced utility consumption and shorter cycle times. For supply chain managers, this means a more predictable production schedule and a lower risk of disruption due to reagent shortages or regulatory changes regarding hazardous substance transport.
- Cost Reduction in Manufacturing: The elimination of toxic sulfur chlorides and the adoption of inexpensive bleach-based reagents result in a drastic reduction in direct material costs. Additionally, the ability to recycle the organic solvent phase significantly lowers waste disposal fees and fresh solvent purchase requirements. By removing the need for specialized corrosion-resistant alloys often required for handling sulfur chlorides, capital expenditure for reactor maintenance and replacement is also substantially decreased, leading to improved long-term asset utilization and overall profitability.
- Enhanced Supply Chain Reliability: Sodium hypochlorite and glacial acetic acid are globally available commodity chemicals with stable supply chains, unlike niche reagents that may face geopolitical or logistical bottlenecks. This abundance ensures consistent availability of raw materials, reducing the risk of production stoppages. The robustness of the process against minor fluctuations in reaction conditions further enhances reliability, allowing for consistent output even when scaling from pilot plants to full commercial production lines, thereby securing continuity of supply for downstream customers.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, simplifying compliance with increasingly stringent environmental regulations such as REACH or TSCA. The absence of sulfur-containing byproducts reduces the load on wastewater treatment facilities and minimizes the generation of noxious off-gases. This environmental compatibility facilitates easier permitting for capacity expansion and aligns with the sustainability goals of major pharmaceutical clients, making the manufacturer a preferred partner for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-amino-5-chlorobenzamide. These insights are derived directly from the patented technology and practical manufacturing experience, aiming to clarify the operational benefits and quality assurances associated with this advanced synthesis route. Understanding these details is vital for technical teams evaluating vendor capabilities and for procurement officers assessing total cost of ownership.
Q: What are the primary safety advantages of this synthesis route compared to traditional methods?
A: Traditional methods often utilize highly toxic and corrosive reagents such as sulfuryl chloride (SO2Cl2) or sulfur dichloride (SCl2), which pose severe risks of respiratory irritation and equipment damage. This novel protocol replaces those hazardous chemicals with sodium hypochlorite and glacial acetic acid, significantly reducing operator exposure risks and minimizing the potential for severe equipment corrosion, thereby enhancing overall plant safety standards.
Q: How does the low-temperature chlorination step impact product quality?
A: Maintaining the reaction temperature below -5°C during the chlorination phase is critical for controlling regioselectivity. This precise thermal management prevents over-chlorination and oxidative degradation of the sensitive amino group, ensuring that the chlorine atom substitutes exclusively at the 5-position. This results in a much cleaner crude profile with fewer isomeric impurities, simplifying downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for scalability. By utilizing inexpensive, commodity-grade reagents like sodium hypochlorite and avoiding complex catalytic systems, the method reduces raw material costs substantially. Furthermore, the ability to recycle solvents and the robustness of the high-pressure ammonolysis step make it highly adaptable for multi-ton manufacturing campaigns without compromising yield or purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Chlorobenzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is not just a technical upgrade but a strategic imperative for the modern pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-amino-5-chlorobenzamide meets the exacting standards required for API synthesis. Our commitment to quality assurance ensures that your downstream processes remain uninterrupted by impurity-related failures.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized supply chain can become a cornerstone of your production strategy.
