Optimized Industrial Synthesis of Mosapride Intermediate I for Global Pharmaceutical Supply Chains
Introduction to Advanced Mosapride Intermediate Synthesis
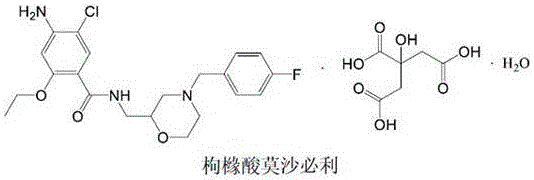
The pharmaceutical landscape for gastrointestinal therapeutics relies heavily on the efficient production of high-purity intermediates for 5-HT4 receptor agonists. Patent CN112225708A introduces a transformative preparation method for Mosapride Intermediate I, specifically 4-(4-fluorobenzyl)-2-aminomethyl morpholine, which serves as a critical building block for Mosapride Citrate. This novel methodology addresses longstanding inefficiencies in traditional synthesis pathways by streamlining the reaction sequence and substituting costly precursors with economically viable alternatives. The technical breakthrough lies in a robust four-step sequence that begins with the alkylation of potassium phthalimide, proceeds through a condensation with 2-aminoethanol, undergoes acid-catalyzed cyclization, and concludes with a highly optimized N-benzylation. By fundamentally restructuring the synthetic logic, this approach not only enhances the chemical purity of the intermediate to levels exceeding 98% but also drastically reduces the impurity profile that often plagues the final API. For R&D directors and procurement specialists, this represents a significant opportunity to optimize both the cost of goods sold (COGS) and the reliability of the supply chain for this essential gastroprokinetic agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Mosapride Intermediate I has been fraught with economic and technical challenges that hinder scalable manufacturing. Prior art methods, such as those disclosed in earlier patents like CN1526700A, typically rely on the synthesis of 2-(4-fluorobenzylamino)ethanol as a key starting material. This precursor is not only expensive to procure on the open market due to its specialized nature but also requires additional synthetic steps to produce, thereby elongating the overall process timeline. Furthermore, conventional routes often involve complex protection and deprotection strategies or harsh reaction conditions that lead to the formation of dark-colored, unstable oily intermediates. These quality issues in the intermediate stage frequently propagate through to the final drug substance, resulting in related substance failures during quality control testing. The reliance on multi-step sequences with lower overall yields increases waste generation and energy consumption, creating a bottleneck for manufacturers aiming to meet the rigorous demands of global regulatory bodies while maintaining competitive pricing structures.
The Novel Approach
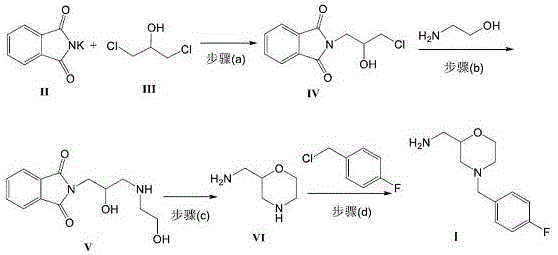
The methodology outlined in CN112225708A offers a decisive break from these traditional constraints by introducing a more direct and atom-economical pathway. Instead of relying on pre-functionalized benzylamines, this novel route utilizes inexpensive and readily available 2-aminoethanol to construct the morpholine core early in the synthesis. The process initiates with the reaction of potassium phthalimide and dichloroisopropanol to form a chloropropyl phthalimide intermediate, which is then condensed with 2-aminoethanol. This strategic inversion of the synthetic order allows for the formation of the morpholine ring via a clean acid-catalyzed cyclization before the introduction of the fluorobenzyl group. The final alkylation step is meticulously optimized using a dual-base system, ensuring high conversion rates and superior product clarity. This approach effectively decouples the complexity of the morpholine ring formation from the benzylation step, resulting in a process that is not only shorter but also produces a solid intermediate with exceptional stability and purity, ready for immediate coupling in the final API synthesis.
Mechanistic Insights into Nucleophilic Substitution and Cyclization
The core chemical innovation of this process revolves around the precise control of nucleophilic substitutions and the thermodynamics of ring closure. In the initial stages, the potassium phthalimide acts as a potent nucleophile, attacking the less hindered carbon of dichloroisopropanol in an SN2 fashion to install the phthalimide protecting group while retaining a reactive chloride handle. Subsequent displacement of this chloride by the primary amine of 2-aminoethanol generates a linear amino-alcohol chain. The critical mechanistic event occurs during the cyclization step, where strong acids such as p-toluenesulfonic acid or concentrated sulfuric acid protonate the hydroxyl groups, facilitating an intramolecular dehydration. This acid-catalyzed cascade drives the formation of the six-membered morpholine ring with high regioselectivity, effectively locking the stereochemistry and preventing the formation of polymeric byproducts. The use of phthalimide as a temporary protecting group is particularly advantageous here, as it prevents over-alkylation of the nitrogen during the chain extension phase and can be cleanly removed or transformed during the cyclization, ensuring that the final morpholine nitrogen is available for the subsequent functionalization without requiring harsh hydrazine cleavage steps typical of Gabriel synthesis.
Furthermore, the final N-benzylation step demonstrates sophisticated base management to maximize yield and minimize side reactions. The patent specifies a mixed base system comprising a tert-butoxide metal salt and triethylamine, preferably in a molar ratio of 3:1. This combination is mechanistically superior to using a single strong base because the bulky tert-butoxide effectively deprotonates the morpholine nitrogen to generate the reactive nucleophile, while the triethylamine acts as a scavenger for the generated hydrogen chloride, preventing the protonation of the product which could lead to salt formation or degradation. The choice of solvent, such as DMF or toluene, further modulates the reactivity by stabilizing the transition state of the SN2 attack on the 4-fluorobenzyl chloride. This fine-tuned reaction environment suppresses the formation of quaternary ammonium salts and other alkylation byproducts, directly contributing to the reported purity levels of over 98% and the characteristic yellow solid appearance of the isolated intermediate, which indicates a lack of oxidative degradation products common in older methods.
How to Synthesize 4-(4-Fluorobenzyl)-2-aminomethyl Morpholine Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and reaction conditions to replicate the high yields reported in the patent examples. The process is designed to be telescoped where possible, particularly between the initial alkylation and the aminoethanol condensation, to minimize solvent swaps and isolation losses. Operators should prioritize the use of anhydrous conditions during the phthalimide alkylation to prevent hydrolysis of the dichloroisopropanol. For the cyclization step, the removal of water via azeotropic distillation is critical to drive the equilibrium towards the morpholine product. Detailed standard operating procedures regarding temperature ramps, addition rates, and workup protocols are essential for maintaining batch-to-batch consistency. The following guide outlines the standardized synthesis steps derived from the patent claims to ensure optimal performance in a GMP environment.
- React potassium phthalimide with dichloroisopropanol in an organic solvent with a base to form N-(2-hydroxy-3-chloropropyl)phthalimide.
- Condense the resulting chloropropyl phthalimide with 2-aminoethanol under alkaline conditions to generate the linear amino-alcohol precursor.
- Perform acid-catalyzed cyclization using strong acids like p-toluenesulfonic acid or concentrated sulfuric acid to form 2-aminomethyl morpholine.
- Alkylate the morpholine intermediate with 4-fluorobenzyl chloride using a mixed base system of tert-butoxide and triethylamine to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound benefits for procurement managers and supply chain directors tasked with optimizing the cost structure of gastrointestinal drug portfolios. The most immediate impact is seen in raw material costs; by replacing the expensive 2-(4-fluorobenzylamino)ethanol with commodity-grade 2-aminoethanol and dichloroisopropanol, the direct material cost of the intermediate is significantly reduced. This substitution eliminates a major cost driver associated with specialized fluorinated building blocks, allowing manufacturers to leverage economies of scale for bulk chemicals. Additionally, the shortened process route reduces the number of unit operations, which translates to lower utility consumption, reduced labor hours, and decreased waste disposal costs. The high purity of the intermediate also means that downstream API manufacturers spend less time and resources on purification, further lowering the total cost of ownership for the supply chain. These factors combine to create a more resilient and cost-effective supply model that can better absorb market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The elimination of expensive specialized precursors and the reduction in process steps lead to substantial operational savings. By utilizing a mixed base system that improves yield efficiency, the process minimizes the loss of valuable intermediates, ensuring that more input material is converted into saleable product. This efficiency gain directly impacts the bottom line, allowing for more competitive pricing strategies in the generic pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals like 2-aminoethanol and potassium phthalimide mitigates the risk of supply disruptions often associated with niche intermediates. The robustness of the reaction conditions, which tolerate standard industrial solvents like DMF and toluene, ensures that production can be scaled across multiple manufacturing sites globally. This diversification potential strengthens the supply chain against geopolitical or logistical shocks, guaranteeing continuous availability of the intermediate for downstream API production schedules.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial operation, avoiding the use of hazardous transition metal catalysts that require complex removal and validation steps. The high purity of the crude product reduces the need for extensive chromatographic purification, thereby lowering solvent usage and waste generation. This aligns with modern green chemistry principles and simplifies regulatory compliance regarding residual solvents and heavy metals, facilitating faster approval times for new drug master files (DMFs) and smoother audits by regulatory agencies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN112225708A, providing a factual basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. The focus is on practical applicability, yield expectations, and quality parameters that define the success of this manufacturing route.
Q: What are the key advantages of this new synthesis route over prior art?
A: The novel route eliminates the need for expensive 2-(4-fluorobenzylamino)ethanol, utilizing cheaper 2-aminoethanol instead. It also simplifies the process by reducing step count and improving overall purity to over 98%, minimizing downstream purification burdens.
Q: How does the mixed base system improve the final alkylation step?
A: Using a combination of tert-butoxide metal salts and triethylamine in a specific molar ratio (preferably 3:1) significantly enhances the yield of the final N-benzylation reaction compared to using single bases, achieving yields upwards of 83% with high purity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial operation. It utilizes commercially available reagents, avoids complex transition metal catalysts, and employs robust solvents like DMF and toluene, ensuring scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Fluorobenzyl)-2-aminomethyl Morpholine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles described in CN112225708A are realized on an industrial scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could affect the safety and efficacy of the final Mosapride Citrate. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of global regulatory bodies, providing our clients with the confidence needed to accelerate their drug development timelines.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this method can reduce your overall manufacturing expenses. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, cost-effective, and high-quality supply of Mosapride Intermediate I, ensuring your production lines run smoothly and efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
