Advanced Industrial Synthesis of Gabapentin: A High-Yield Route for API Manufacturing
Introduction to Patent CN100398513C
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antiepileptic agents like Gabapentin, where purity and safety profiles are paramount. Patent CN100398513C introduces a transformative preparation method that fundamentally shifts away from cumbersome ion-exchange technologies toward a more direct and efficient acid hydrolysis pathway. This innovation addresses long-standing challenges in the synthesis of 1-(aminomethyl)cyclohexyl acetic acid, specifically targeting the reduction of toxic impurities and the optimization of energy consumption during large-scale production. By utilizing 3,3-pentylidene butyrolactam as the primary starting material, the process achieves a streamlined workflow that enhances both operational safety and economic viability for reliable pharmaceutical intermediates suppliers. The methodology ensures that the final product meets stringent pharmacopeial standards with purity exceeding 99.8% and minimal chloride ion content, making it an ideal candidate for downstream API formulation.
Furthermore, the technical breakthrough lies in the sophisticated management of reaction by-products and mother liquors, which traditionally represented significant waste streams in older synthetic routes. The patent details a closed-loop system where unreacted starting materials are efficiently recovered from various stages of the process, thereby maximizing atom economy and minimizing environmental impact. This approach not only stabilizes the supply chain by reducing dependency on fresh raw material inputs but also simplifies the regulatory burden associated with waste disposal. For procurement teams evaluating cost reduction in API manufacturing, this process offers a compelling value proposition through its inherent efficiency and reduced utility requirements. The stability data confirms that the resulting Gabapentin maintains its integrity under accelerated conditions, validating the robustness of this novel synthetic strategy for global distribution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Gabapentin has relied heavily on ion-exchange resin technologies or complex multi-step sequences involving hazardous reagents like sodium azide or bromine. These conventional pathways often necessitate the use of vast quantities of water as a solvent, creating a massive burden on downstream processing facilities that must distill off this water to isolate the product. The distillation process is particularly problematic because Gabapentin is prone to cyclization back into the toxic precursor, 3,3-pentylidene butyrolactam, if temperatures exceed strict limits such as 45°C during concentration. Additionally, traditional methods frequently employ mixed solvent systems containing ethers and alcohols, which introduce significant safety hazards regarding flammability and peroxide formation, complicating facility compliance and insurance costs. The reliance on ion-exchange resins also introduces variability in product quality, particularly concerning chloride ion content and residual heavy metals, requiring extensive post-treatment validation.
The Novel Approach
In stark contrast, the method disclosed in CN100398513C employs a direct hydrolysis strategy using inorganic acids like hydrochloric acid to open the lactam ring, followed by a precise pH-controlled precipitation sequence. This eliminates the need for ion-exchange columns entirely, thereby removing the associated capital expenditure for resin beds and the operational complexity of resin regeneration cycles. The process cleverly integrates a decolorization and adsorption step using activated carbon and diatomite at a specific acidic pH range, which selectively removes the toxic lactam impurity before the final product crystallizes. By shifting the crystallization environment to an alcoholic system for the final dehydration step, the method avoids the energy-intensive water distillation required by older techniques, significantly lowering the thermal load on the production plant. This results in a safer, more controllable operation that is inherently easier to scale from pilot batches to multi-ton commercial runs without compromising on the critical quality attributes of the active pharmaceutical ingredient.
Mechanistic Insights into Acid Hydrolysis and pH-Controlled Purification
The core chemical transformation in this synthesis involves the acid-catalyzed hydrolysis of the lactam ring in 3,3-pentylidene butyrolactam, which proceeds through a nucleophilic attack by water molecules on the carbonyl carbon under heated conditions. The reaction kinetics are optimized by maintaining a temperature range between 50°C and 120°C, ensuring complete conversion to the Gabapentin salt while minimizing side reactions that could lead to degradation products. The stoichiometry of the mineral acid is carefully controlled, typically using a molar ratio of acid to lactam between 1:2 and 1:15, to drive the equilibrium towards the open-chain amino acid salt form. Following the reaction, the system is cooled to sub-zero temperatures to induce crystallization of the Gabapentin hydrochloride, leveraging the solubility differences to separate the product from the acidic mother liquor efficiently. This initial isolation step is crucial as it sets the stage for the subsequent purification phases by removing the bulk of the reaction medium and soluble impurities.
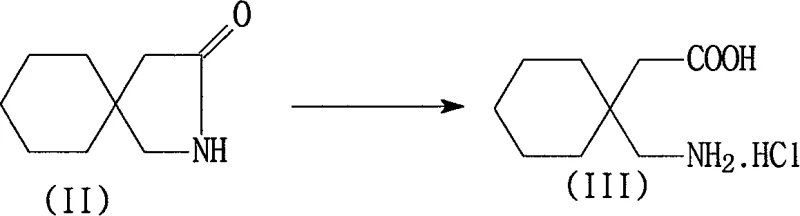
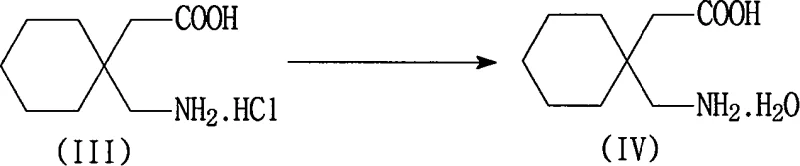
Subsequent purification relies on a sophisticated manipulation of the isoelectric properties of Gabapentin to achieve high purity levels. The Gabapentin salt is redissolved in water, and the pH is adjusted to a range of 3 to 7, a critical window where the addition of activated carbon and diatomite effectively adsorbs residual toxic 3,3-pentylidene butyrolactam and colored impurities. Once filtered, the pH is raised further to between 8 and 8.5, approaching the isoelectric point of the molecule, which triggers the precipitation of Gabapentin hydrate in a highly pure crystalline form. This two-stage pH adjustment is superior to single-step neutralization methods as it decouples the impurity removal phase from the product isolation phase, ensuring that the toxic precursor does not co-crystallize with the final product. The final conversion to anhydrous Gabapentin is achieved by suspending the hydrate in an alcohol solvent, facilitating the removal of crystal water without exposing the molecule to harsh thermal conditions that could induce degradation.

How to Synthesize Gabapentin Efficiently
The synthesis of high-purity Gabapentin via this patented route requires precise control over reaction parameters and sequential unit operations to maximize yield and minimize impurity carryover. Operators must strictly adhere to the temperature profiles during the hydrolysis and crystallization steps to prevent the reformation of the toxic lactam precursor, while also managing the pH transitions during the purification phase to ensure effective adsorption of contaminants. The detailed standardized synthesis steps involve specific ratios of reagents, cooling rates, and filtration protocols that have been optimized to deliver consistent batch-to-batch quality suitable for pharmaceutical applications. For a comprehensive breakdown of the exact operational parameters and equipment specifications required to implement this process, please refer to the technical guide below.
- Hydrolyze 3,3-pentylidene butyrolactam with inorganic acid (e.g., HCl) at 50-120°C to form Gabapentin salt, followed by crystallization at low temperatures.
- Dissolve the salt in water, adjust pH to 3-7 for decolorization with activated carbon to remove toxic impurities, then adjust pH to 8-8.5 to precipitate Gabapentin hydrate.
- Convert Gabapentin hydrate to anhydrous Gabapentin by mixing with alcohol (e.g., ethanol) at 0-50°C, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers substantial strategic benefits primarily driven by the simplification of the manufacturing workflow and the reduction of utility consumption. By eliminating the ion-exchange step, facilities can avoid the recurring costs associated with resin replacement and the extensive water treatment infrastructure needed to handle large aqueous waste streams. The ability to recycle mother liquors to recover unreacted starting material creates a circular economy within the production line, drastically reducing the net consumption of raw materials and insulating the supply chain from volatile market pricing for key precursors. Furthermore, the use of common solvents like ethanol instead of hazardous ether mixtures simplifies logistics, storage, and safety compliance, leading to lower insurance premiums and reduced risk of production stoppages due to safety incidents.
- Cost Reduction in Manufacturing: The elimination of energy-intensive water distillation steps results in significantly lower steam and electricity usage per kilogram of product produced. Since the process avoids the need for specialized ion-exchange equipment and the associated maintenance downtime, the overall capital efficiency of the plant is improved, allowing for better asset utilization. The recovery of 3,3-pentylidene butyrolactam from waste streams further contributes to cost optimization by turning what was previously a waste disposal cost into a valuable feedstock input. These factors combine to create a leaner cost structure that enhances competitiveness in the global generic pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the chemical process, characterized by wide operating windows for temperature and pH, ensures high batch success rates and minimizes the risk of production failures that could disrupt supply continuity. The reliance on readily available commodity chemicals such as hydrochloric acid, sodium hydroxide, and ethanol reduces dependency on specialized or scarce reagents that might face supply bottlenecks. Additionally, the shorter production cycle time compared to traditional resin-based methods allows for faster turnaround on customer orders, enabling manufacturers to respond more agilely to fluctuations in market demand. This operational flexibility is critical for maintaining service levels in a just-in-time delivery environment.
- Scalability and Environmental Compliance: The process is designed with inherent scalability, utilizing standard reactor types and separation equipment that can be easily scaled from pilot plants to multi-ton commercial production lines without complex re-engineering. The reduction in aqueous waste volume and the avoidance of hazardous ether solvents significantly lower the environmental footprint of the manufacturing site, facilitating easier permitting and compliance with increasingly strict environmental regulations. The efficient recovery and reuse of solvents and raw materials align with green chemistry principles, enhancing the corporate sustainability profile of the manufacturer and appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced Gabapentin synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or capacity expansion projects.
Q: How does this process control the toxic impurity 3,3-pentylidene butyrolactam?
A: The process utilizes a specific pH adjustment strategy (pH 3-7) combined with activated carbon and diatomite adsorption during the purification stage. This effectively removes residual toxic lactam before the final precipitation step, ensuring levels remain below 0.03%.
Q: What are the advantages over traditional ion-exchange methods?
A: Unlike ion-exchange methods that require vast amounts of water and energy-intensive distillation to remove solvents, this method uses direct acid hydrolysis and controlled precipitation. It eliminates the need for resin regeneration, reduces energy consumption significantly, and avoids the safety risks associated with ether/alcohol mixed solvents.
Q: Is the mother liquor recyclable in this synthesis route?
A: Yes, the process allows for the recycling of mother liquors from the hydrolysis, purification, and dehydration steps. By adjusting the pH of combined mother liquors to alkaline conditions and heating, unreacted 3,3-pentylidene butyrolactam can be recovered and reused, substantially improving overall yield and reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gabapentin Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to bring complex synthetic routes like the one described in CN100398513C to life on an industrial scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Gabapentin meets the highest international standards for safety and efficacy. Our commitment to process excellence allows us to deliver high-quality pharmaceutical intermediates that support the global supply of essential medications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic advantages of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a successful and profitable collaboration.
