Advanced Synthetic Route for 10 10-Disubstituted-2-Bromoanthrone: Commercial Scalability and Technical Insights
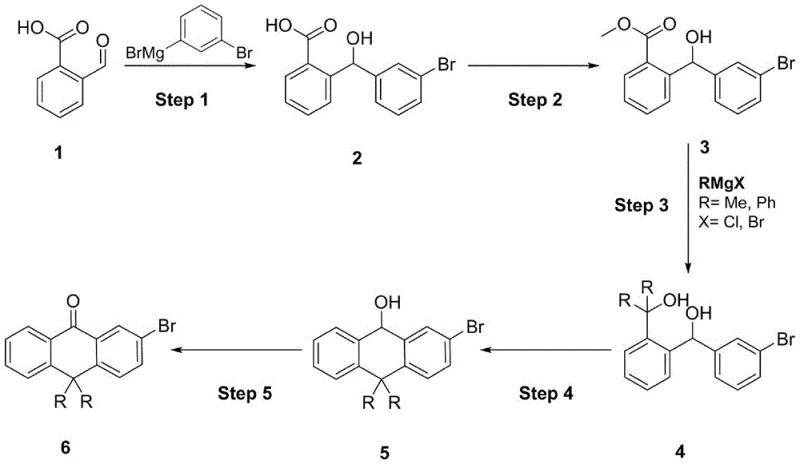
The chemical landscape for organic luminescent materials is constantly evolving, driven by the demand for higher efficiency and thermal stability in display technologies. Patent CN107793302B, published in late 2020, introduces a groundbreaking methodology for the preparation of 10,10-disubstituted-2-bromoanthrone, a critical intermediate in the synthesis of advanced organic light-emitting compounds. This specific chemical architecture is pivotal for constructing molecules that exhibit superior current efficiency and robust industrialization prospects, addressing the limitations of earlier synthetic routes. The invention leverages a five-step continuous reaction sequence starting from o-carboxybenzaldehyde and m-dibromobenzene Grignard reagent, meticulously designed to overcome the incompatibility issues often encountered with bromine-containing functional groups. By integrating esterification, tertiary alcoholation, cyclization, and oxidation into a cohesive workflow, this patent provides a new, reliable pathway for synthesizing complex anthrone derivatives that were previously difficult to access with high purity. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-performance electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 10,10-dimethylanthrone and its derivatives has been plagued by significant technical hurdles that compromise both yield and structural integrity. Traditional routes often rely on the substitution reaction of o-chlorobenzyl chloride with benzene, followed by a Grignard reaction with magnesium strips and subsequent addition to acetone. These multi-step processes are not only labor-intensive but also suffer from poor repeatability due to the formation of two-phase systems that require expensive crown ether phase transfer catalysts. Furthermore, the presence of strong basic conditions in methylation reactions using methyl iodide frequently leads to the generation of substantial methoxylation impurities, which are notoriously difficult to remove during purification. Most critically, these conventional methods are fundamentally unsuitable for preparing 10,10-disubstituted derivatives containing bromo substituents, as the harsh reaction conditions are incompatible with the bromine functional group, often resulting in dehalogenation and the destruction of the desired molecular architecture. This lack of functional group tolerance severely limits the applicability of older methods in the context of modern organic luminescent material synthesis, where specific halogenated patterns are required for fine-tuning electronic properties.
The Novel Approach
In stark contrast to the limitations of prior art, the novel approach disclosed in CN107793302B offers a robust and tolerant synthetic strategy that preserves sensitive functional groups throughout the transformation. The method initiates with a controlled Grignard addition reaction between o-carboxybenzaldehyde and a m-dibromobenzene Grignard reagent, facilitated by lithium bromide at low temperatures to ensure high selectivity. This is followed by a streamlined esterification step using sulfuric acid and methanol, which prepares the intermediate for the crucial introduction of the 10,10-substituents via a second Grignard reaction with methyl or phenyl reagents. The core innovation lies in the subsequent cyclization reaction utilizing boron tribromide, which proceeds under mild conditions to form the anthracene backbone without compromising the bromine atom at the 2-position. Finally, a gentle oxidation step using manganese dioxide and trifluoroacetic acid completes the synthesis, yielding the target 10,10-disubstituted-2-bromoanthrone with exceptional purity. This route not only simplifies the operational complexity but also significantly enhances the tolerance to bromine-containing functional groups, making it an ideal candidate for the reliable production of specialized organic intermediates.
Mechanistic Insights into Grignard Addition and Boron Tribromide Cyclization
The mechanistic elegance of this synthesis lies in the precise control of reactivity during the Grignard addition and the subsequent cyclization phases. In the initial step, the addition of lithium bromide acts as a promoter, stabilizing the Grignard reagent and facilitating the nucleophilic attack on the aldehyde carbonyl of o-carboxybenzaldehyde at 0-5°C. This low-temperature control is critical for minimizing side reactions and ensuring the formation of the 2-((3-bromophenyl)hydroxymethyl)benzoic acid intermediate with high fidelity. The subsequent esterification converts the carboxylic acid into a methyl ester, which is more amenable to the next Grignard addition where methyl or phenyl groups are introduced at the benzylic position. The use of tetrahydrofuran and n-hexane as a mixed solvent system in this step optimizes the solubility of the intermediates and the reactivity of the Grignard reagent, ensuring complete conversion to the tertiary alcohol precursor. This careful orchestration of reaction conditions prevents the formation of unwanted byproducts and sets the stage for the ring-closing event.
The cyclization mechanism mediated by boron tribromide represents a significant departure from traditional Friedel-Crafts methodologies, offering superior regioselectivity for the 10,10-disubstituted architecture. By dropwise adding the tertiary alcohol intermediate and boron tribromide simultaneously at temperatures below 10°C, the reaction promotes an intramolecular electrophilic aromatic substitution that closes the central ring of the anthrone system. This mild Lewis acid condition is specifically chosen to avoid the harsh acidic environments that typically lead to the cleavage of the carbon-bromine bond, thereby preserving the essential bromine substituent for downstream functionalization. The final oxidation step utilizes manganese dioxide in the presence of trifluoroacetic acid to convert the dihydroanthracene-9-ol intermediate into the corresponding anthrone. This oxidation is highly selective for the benzylic alcohol, avoiding over-oxidation of the aromatic rings or the bromine substituent, which is crucial for maintaining the impurity profile within stringent specifications required for electronic material applications. The cumulative effect of these mechanistic choices is a process that delivers high-purity products with minimal impurity burden.
How to Synthesize 10,10-Disubstituted-2-Bromoanthrone Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires strict adherence to the temperature controls and reagent ratios specified in the patent to ensure reproducibility and safety. The process begins with the preparation of the Grignard reagent under an inert nitrogen atmosphere, followed by the controlled addition of the aldehyde substrate to manage the exothermic nature of the reaction. Operators must pay close attention to the quenching steps, particularly the acidification of the aqueous layers to precipitate the intermediate solids, which is vital for isolating the product from magnesium salts and other inorganic byproducts. The detailed standardized synthesis steps, including specific solvent volumes, dropping times, and workup procedures, are critical for achieving the reported yields and purity levels. For a comprehensive guide on the exact operational parameters and safety precautions required for each of the five steps, please refer to the standardized protocol outlined below.
- Perform Grignard addition of o-carboxybenzaldehyde with m-dibromobenzene Grignard reagent at 0-5°C to form the hydroxymethyl benzoic acid intermediate.
- Execute esterification using sulfuric acid and methanol, followed by a second Grignard reaction with methyl or phenyl reagents to introduce the 10,10-substituents.
- Complete the synthesis via boron tribromide-mediated cyclization at low temperature and final oxidation using manganese dioxide and trifluoroacetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemical industry. The reliance on cheap and easily obtainable raw materials, such as o-carboxybenzaldehyde and m-dibromobenzene, significantly reduces the raw material cost base compared to routes requiring specialized or scarce precursors. Furthermore, the simplification of reaction conditions eliminates the need for expensive additives like crown ether phase transfer catalysts, which are often a significant cost driver in traditional anthrone synthesis. The robustness of the process, characterized by its tolerance to functional groups and simple operational requirements, translates into higher process reliability and reduced risk of batch failures. This reliability is paramount for maintaining a consistent supply of high-purity intermediates, ensuring that downstream production of organic luminescent materials is not disrupted by raw material shortages or quality inconsistencies. By adopting this route, manufacturers can achieve significant cost savings and enhance their supply chain resilience against market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive phase transfer catalysts and the use of common solvents like tetrahydrofuran and dichloromethane drastically simplify the cost structure of the manufacturing process. The high yields reported in the patent examples, particularly in the final oxidation step, indicate a highly efficient use of raw materials, minimizing waste and maximizing output per unit of input. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further contributing to overall operational cost reductions. These factors combine to create a economically viable production model that allows for competitive pricing in the market for organic luminescent material intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials ensures that the supply chain is not dependent on single-source suppliers or exotic chemicals that may face availability issues. The robustness of the synthetic route means that production can be scaled up with confidence, knowing that the process is tolerant to minor variations in reaction conditions. This stability is crucial for long-term supply agreements, as it reduces the likelihood of production delays caused by technical difficulties or purification bottlenecks. Consequently, partners can rely on a steady flow of high-quality intermediates, supporting just-in-time manufacturing strategies and reducing the need for excessive inventory buffers.
- Scalability and Environmental Compliance: The five-step sequence is designed with scalability in mind, utilizing standard unit operations such as extraction, filtration, and distillation that are easily implemented in large-scale reactors. The avoidance of heavy metal catalysts and the use of manganese dioxide, which can be managed through standard waste treatment protocols, aligns with increasingly stringent environmental regulations. The process generates manageable waste streams, and the high selectivity of the reactions reduces the burden on downstream purification, leading to a lower environmental footprint. This compliance with environmental standards not only mitigates regulatory risk but also enhances the sustainability profile of the supply chain, a key consideration for modern corporate procurement policies.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical specifications and beneficial effects detailed in patent CN107793302B, addressing common concerns regarding the feasibility and advantages of this synthesis. These insights are intended to clarify the technical merits of the route for stakeholders evaluating its potential for commercial adoption. By understanding the specific improvements over prior art, decision-makers can better assess the value proposition of this technology for their specific application needs. The answers provided reflect the objective data and logical deductions available within the patent documentation, ensuring accuracy and relevance for technical and commercial evaluation.
Q: Why is this method superior for bromine-containing anthrone derivatives?
A: Conventional methods often utilize strong bases or harsh conditions that are incompatible with bromine substituents, leading to dehalogenation. This patent's route employs mild Grignard conditions and specific cyclization agents that preserve the bromine functionality, ensuring high structural integrity for downstream luminescent applications.
Q: What are the key cost drivers in this synthesis?
A: The primary cost advantages stem from the use of cheap and easily obtainable raw materials like o-carboxybenzaldehyde and m-dibromobenzene. Furthermore, the elimination of expensive phase transfer catalysts and the simplification of purification steps significantly reduce the overall manufacturing expenditure compared to prior art.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are simple and tolerant, with temperatures controlled between 0°C and reflux, making it highly adaptable for commercial scale-up. The use of standard solvents like tetrahydrofuran and dichloromethane ensures that the process can be readily transferred to large-scale reactors without specialized equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10,10-Disubstituted-2-Bromoanthrone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex intermediates like 10,10-disubstituted-2-bromoanthrone. Our technical team is adept at translating patent-protected methodologies into robust industrial processes, ensuring that the stringent purity specifications required for organic luminescent materials are consistently met. With rigorous QC labs and a commitment to quality, we guarantee that every batch delivered adheres to the highest standards of chemical integrity, supporting your R&D and production goals with unwavering reliability. We understand the critical nature of supply continuity in the electronic materials sector and have optimized our operations to minimize lead times while maintaining flexibility.
We invite you to collaborate with us to optimize your supply chain for high-purity organic intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By engaging with us, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of our manufacturing capabilities. Let us help you secure a stable and cost-effective source of 10,10-disubstituted-2-bromoanthrone, enabling you to focus on innovation and market expansion with confidence in your raw material supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
