Advanced Manufacturing of Nepyrrolol Hydrochloride via Efficient Chiral Pool Synthesis
Advanced Manufacturing of Nepyrrolol Hydrochloride via Efficient Chiral Pool Synthesis
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the demand for higher purity intermediates and more sustainable manufacturing processes. Patent CN1978442A introduces a groundbreaking synthetic methodology for (R,R,R,S)-2,2'-[iminobis(methylene)]bis-(6-fluoro-3,4-dihydro-2H-1-benzopyran-2-methanol) nepyrrolol hydrochloride, a potent β1-receptor blocker used in the management of hypertension. This technical disclosure represents a significant departure from traditional racemic syntheses, leveraging the inherent chirality of natural D-mannitol to establish stereocenters with high fidelity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this route offers a compelling alternative to legacy methods that rely on cumbersome resolution steps. By shifting the paradigm from resolution-based synthesis to chiral pool utilization, the technology addresses critical pain points regarding optical purity, process complexity, and overall cost efficiency in the production of high-value antihypertensive agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Nepyrrolol hydrochloride has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Prior art methodologies, such as those initiated from chromanic acid, necessitated the resolution of racemic mixtures to obtain the desired enantiomeric excess, a process that inherently caps the maximum theoretical yield at 50% unless dynamic kinetic resolution is employed. Furthermore, the synthesis of key epoxide intermediates in traditional routes often required separation via high-performance liquid chromatography (HPLC), a technique that is notoriously difficult to scale beyond kilogram quantities due to solvent consumption and column capacity limitations. The reliance on expensive coupling agents and the generation of difficult-to-remove impurities during the benzylamine coupling steps further exacerbated the cost profile, making the supply chain vulnerable to fluctuations in reagent pricing and waste disposal regulations. These factors collectively created a bottleneck for manufacturers aiming to reduce lead time for high-purity pharmaceutical intermediates while maintaining competitive pricing structures.
The Novel Approach
In stark contrast to the limitations of the past, the methodology described in CN1978442A utilizes a streamlined, chiral pool strategy that fundamentally simplifies the molecular construction of the target molecule. By employing D-mannitol, an abundant and inexpensive natural sugar, as the starting material, the synthesis bypasses the need for early-stage resolution, effectively locking in the desired stereochemistry from the very first step. The route cleverly integrates a condensation reaction between a chiral glyceraldehyde derivative and 4-fluorophenol derivatives to construct the fluorochroman core, eliminating the need for hazardous epoxide handling and subsequent chromatographic purification. Additionally, the introduction of dry ammonia gas for the amination step replaces costly amine reagents, significantly driving down raw material costs while simplifying the workup procedure. This innovative approach not only enhances the optical purity of the final product but also aligns perfectly with modern green chemistry principles, offering a robust pathway for cost reduction in API manufacturing that is both environmentally conscious and economically viable for large-scale operations.
Mechanistic Insights into Chiral Pool Synthesis and Cyclization
The core mechanistic brilliance of this synthesis lies in the precise transfer of chirality from the carbohydrate backbone to the pharmacologically active chroman ring system. The process initiates with the protection of D-mannitol using acetone under Lewis acid catalysis to form diacetone mannitol, which serves as a stable chiral scaffold. Subsequent oxidative cleavage of the vicinal diol moiety using sodium periodate generates a chiral glyceraldehyde derivative with retained stereochemical integrity. This aldehyde then undergoes a condensation reaction with 2-acetyl-4-fluorophenol, facilitated by amine catalysts such as pyrrolidine or piperidine, to form the critical fluorochroman-4-one intermediates. The stereochemical outcome of this cyclization is governed by the existing chiral centers on the glyceraldehyde fragment, ensuring that the resulting chromanone possesses the correct configuration for downstream biological activity without the need for external chiral auxiliaries or catalysts.
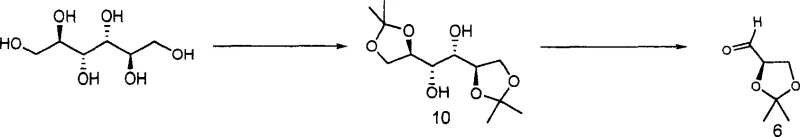
Following the formation of the chromanone core, the mechanism proceeds through a Clemmensen reduction to convert the ketone functionality into a methylene group, simultaneously removing the acetonide protecting groups to reveal the diol side chain. This one-pot deprotection and reduction strategy is particularly elegant as it minimizes unit operations and solvent usage. The resulting diols are then selectively activated via tosylation, converting the primary hydroxyl groups into excellent leaving groups for nucleophilic substitution. The subsequent displacement with ammonia gas proceeds via an SN2 mechanism, inverting the configuration at the reaction center to install the amine functionality with high regioselectivity. Finally, the coupling of the amine fragment with the remaining tosylate precursor closes the molecular architecture, forming the bis-chroman structure characteristic of Nepyrrolol. This sequence demonstrates a masterful control over impurity profiles, as each step is designed to produce crystalline intermediates that can be purified via simple recrystallization rather than complex chromatography.
How to Synthesize Nepyrrolol Hydrochloride Efficiently
The execution of this synthetic route requires careful attention to reaction conditions, particularly during the oxidative cleavage and cyclization stages, to ensure maximum yield and optical purity. The process begins with the preparation of the chiral glyceraldehyde synthon, which must be handled under anhydrous conditions to prevent racemization or degradation. Following the condensation with the fluorophenol derivative, the crude chromanone mixture is subjected to rigorous purification protocols to remove any diastereomeric impurities before proceeding to the reduction step. The use of zinc amalgam in the Clemmensen reduction requires precise control of acid concentration and temperature to avoid over-reduction or side reactions on the aromatic ring. Throughout the synthesis, the emphasis is on telescoping operations where possible, such as the direct use of crude tosylates in the amination step, to minimize material loss. For detailed operational parameters and safety guidelines regarding the handling of reactive intermediates, the detailed standardized synthesis steps are provided in the guide below.
- Protect D-mannitol with acetone using a Lewis acid catalyst to form diacetone mannitol, followed by oxidative cleavage with sodium periodate to generate chiral glyceraldehyde.
- Condense the chiral glyceraldehyde with 2-acetyl-4-fluorophenol to form the fluorochromanone intermediates, establishing the core chroman structure.
- Perform Clemmensen reduction, tosylation, amination with ammonia gas, and final coupling to yield the target Nepyrrolol base, followed by salt formation with HCl.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this mannitol-based synthesis route offers substantial benefits that extend far beyond simple laboratory curiosity. The shift away from resolution-dependent pathways means that the theoretical yield ceiling is significantly raised, allowing for better mass balance and reduced waste generation per kilogram of final product. For supply chain heads concerned with continuity, the reliance on D-mannitol and 4-fluorophenol ensures a stable supply of starting materials, as these are commodity chemicals produced globally in massive quantities, unlike specialized chiral building blocks that may have single-source vulnerabilities. Furthermore, the elimination of HPLC purification steps drastically reduces the solvent footprint and energy consumption associated with the manufacturing process, translating directly into lower operational expenditures and a smaller environmental impact. This alignment with sustainability goals is increasingly becoming a prerequisite for vendor qualification in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of expensive resolution agents and chromatographic media with inexpensive, commodity-grade reagents. By utilizing ammonia gas instead of complex amine coupling partners, the raw material cost is drastically simplified, and the removal of inorganic salts becomes a straightforward aqueous wash rather than a complex extraction. The avoidance of HPLC separation for diastereomeric epoxides removes a major cost center, as preparative chromatography is one of the most expensive unit operations in fine chemical synthesis. Additionally, the ability to purify intermediates via crystallization rather than column chromatography allows for the use of standard stainless steel reactors, reducing capital expenditure requirements for specialized equipment. These factors combine to create a leaner cost structure that provides significant margin protection against raw material price volatility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-proprietary starting materials that are readily available from multiple global vendors. D-mannitol is a fermentation-derived product with a well-established supply chain, ensuring that production schedules are not disrupted by the scarcity of exotic chiral pools. The synthetic route is designed to be modular, allowing different intermediates like the fluorochromanone or the tosylate precursors to be stockpiled strategically to buffer against demand spikes. Moreover, the mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitive reagent degradation, leading to more predictable lead times and higher on-time delivery rates. This reliability is crucial for pharmaceutical customers who require consistent quality and timing to meet their own regulatory filing and production deadlines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial implementation in mind from the outset. The replacement of batch-intensive chromatographic steps with continuous or semi-continuous crystallization processes facilitates the transition from pilot plant to multi-ton commercial production without the need for extensive re-engineering. From an environmental compliance standpoint, the reduction in solvent usage and the elimination of heavy metal catalysts in certain steps simplify wastewater treatment and hazardous waste disposal. The use of ammonia gas, while requiring appropriate scrubbing systems, generates ammonium salts that are easier to manage than complex organic amine waste streams. This cleaner profile not only reduces regulatory burden but also enhances the corporate social responsibility standing of the manufacturing facility, making it a preferred partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer or process validation. The answers reflect the current state of the art as described in the intellectual property, serving as a foundation for further discussion with our technical sales team.
Q: What is the primary advantage of using mannitol in Nepyrrolol synthesis?
A: Using mannitol as a chiral pool starting material eliminates the need for expensive and difficult resolution steps of racemic intermediates, such as the chromatographic separation of diastereomeric epoxides required in older methods.
Q: How does this process improve scalability compared to prior art?
A: The process avoids high-performance liquid chromatography (HPLC) for separating diastereomers and utilizes common reagents like ammonia gas, making the workflow significantly more amenable to large-scale industrial production.
Q: What are the key intermediates in this synthetic route?
A: Key intermediates include diacetone mannitol, chiral glyceraldehyde derivatives, fluorochromanones, and specific tosylate precursors which are coupled to form the final bis-chroman structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nepyrrolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex cardiovascular drugs depends on a partnership grounded in technical excellence and supply chain integrity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of the chiral glyceraldehyde intermediate or full-scale production of the final Nepyrrolol hydrochloride API, our infrastructure is designed to support your regulatory filings and market launch timelines with unwavering consistency and quality assurance.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements and regional logistics. We encourage potential partners to contact us directly to obtain specific COA data for our reference standards and to schedule a comprehensive review of route feasibility assessments tailored to your unique development needs. Let us collaborate to bring this life-saving antihypertensive medication to patients worldwide through a manufacturing process that defines the future of efficiency and quality.
