Advanced Synthesis of Chiral Oxazolidin-2-Ones for Commercial Scale-Up
Advanced Synthesis of Chiral Oxazolidin-2-Ones for Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing chiral scaffolds, particularly when dealing with high-value active pharmaceutical ingredients (APIs). Patent CN1139581C introduces a transformative approach to synthesizing chiral oxazolin-2-ones, also known as Evans auxiliaries, which are pivotal in asymmetric synthesis. This innovation addresses critical bottlenecks in traditional manufacturing by replacing hazardous reagents with safer, cost-effective alternatives while maintaining exceptional stereochemical integrity. By leveraging a streamlined two-step sequence involving chloroformate acylation followed by base-catalyzed cyclization, this technology offers a reliable pathway for producing high-purity pharmaceutical intermediates. The strategic shift away from complex borane chemistry not only enhances operational safety but also drastically simplifies the supply chain logistics for global manufacturers seeking consistent quality.
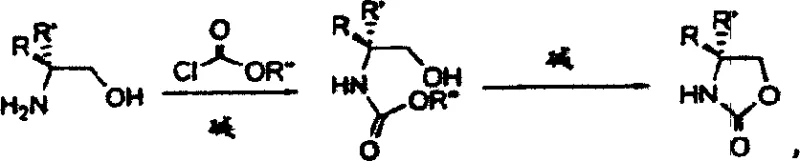
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral oxazolidinones has been dominated by the classical Evans method, which relies heavily on the reduction of chiral amino acids using borane complexes. This legacy approach presents severe logistical and safety challenges for industrial scale-up, primarily due to the requirement for strictly anhydrous and anaerobic operating conditions. The handling of borane reagents poses significant explosion hazards and necessitates specialized equipment that increases capital expenditure and operational complexity. Furthermore, the isolation of the intermediate amino alcohols is notoriously difficult, often leading to emulsification issues during extraction and substantial product loss. The reproducibility of these older methods is frequently compromised by minor variations in moisture content or temperature, resulting in inconsistent yields that are unacceptable for commercial API manufacturing. Consequently, procurement teams have long struggled with the high costs associated with waste disposal and the rigorous safety protocols needed to manage these reactive species.
The Novel Approach
In stark contrast, the methodology disclosed in CN1139581C utilizes readily available chloroformates, such as methyl or ethyl chloroformate, to effect the initial acylation of chiral amino alcohols under mild conditions. This novel route eliminates the need for dangerous borane reductions entirely, allowing the reaction to proceed safely in aqueous or common organic solvent systems at ambient or slightly cooled temperatures. The subsequent cyclization step is achieved simply by heating the intermediate in the presence of inexpensive inorganic bases like potassium carbonate or sodium hydroxide. This streamlined process not only improves the overall atom economy but also facilitates a much cleaner workup procedure, typically involving simple phase separation and solvent evaporation. For a reliable pharmaceutical intermediate supplier, adopting this chemistry means transitioning from a high-risk, low-yield operation to a predictable, high-efficiency manufacturing platform that aligns perfectly with modern green chemistry principles.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this technological advancement lies in the efficient intramolecular nucleophilic attack that forms the oxazolidinone ring system without compromising the chiral center. In the first stage, the amino group of the chiral amino alcohol reacts with the chloroformate to form a stable N-alkoxycarbonyl intermediate, effectively protecting the amine while activating the molecule for cyclization. The use of mild bases during this acylation step ensures that no racemization occurs at the alpha-carbon, preserving the optical purity essential for downstream asymmetric transformations. Following isolation or direct telescoping, the application of heat and base triggers the deprotonation of the hydroxyl group, generating an alkoxide that attacks the adjacent carbonyl carbon of the carbamate moiety. This intramolecular displacement expels the alkoxide leaving group (methoxide or ethoxide), closing the five-membered ring with high fidelity. The mechanistic simplicity of this pathway minimizes the formation of by-products, ensuring that the final impurity profile is exceptionally clean and easy to characterize.
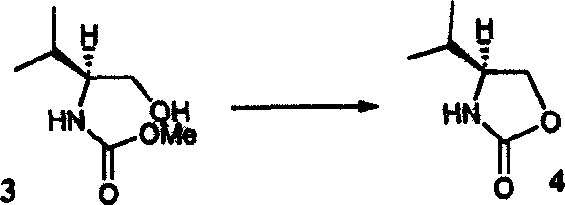
Controlling impurities in chiral synthesis is paramount, and this method excels by avoiding harsh acidic or basic conditions that typically lead to epimerization. The reaction parameters, specifically the temperature range of 60°C to 250°C and the precise molar ratios of base to substrate, are optimized to drive the equilibrium towards the cyclic product while suppressing degradation pathways. Unlike methods employing bulky protecting groups like Boc-anhydride, which can leave stubborn residues, the methyl or ethyl carbamate leaving groups are volatile and easily removed during the concentration phase. This results in a final product that requires minimal purification, often crystallizing directly from the reaction mixture or after a simple wash. For R&D directors, this level of control translates to reduced analytical burden and faster release times for new drug candidates, as the spectral data consistently matches literature standards without the need for extensive recrystallization campaigns.
How to Synthesize Chiral Oxazolidin-2-Ones Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the base and the selection of appropriate solvents to maximize throughput. The process begins with the reaction of the chiral amino alcohol with a slight excess of chloroformate in the presence of an inorganic base such as potassium carbonate, typically in water or a water-miscible organic solvent. Once the N-alkoxycarbonyl intermediate is formed, the mixture is subjected to thermal conditions to induce cyclization, a step that can be performed under reduced pressure to facilitate the removal of the alcohol by-product. Detailed standardized synthetic steps for optimizing this transformation, including specific temperature ramps and workup procedures, are outlined below to ensure reproducible results across different batch sizes.
- React chiral amino alcohol with methyl or ethyl chloroformate in the presence of inorganic or organic base at temperatures between -40°C and +60°C to form the N-alkoxycarbonyl intermediate.
- Heat the resulting N-alkoxycarbonyl-amino alcohol mixture with a catalytic amount of inorganic base at temperatures ranging from 60°C to 250°C to induce cyclization.
- Cool the reaction mixture, extract with organic solvents such as ethyl acetate or toluene, and purify to obtain the final chiral oxazolidin-2-one with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method represents a strategic opportunity to optimize cost structures and mitigate supply risks. By shifting away from proprietary or hazardous reagents towards commodity chemicals, manufacturers can achieve significant cost reduction in API manufacturing without sacrificing quality. The elimination of complex safety infrastructure required for borane handling further reduces overhead costs, while the simplified purification workflow decreases solvent consumption and waste treatment expenses. This efficiency gain allows for more competitive pricing models and improved margins in a highly regulated market environment.
- Cost Reduction in Manufacturing: The substitution of expensive acylating agents like phenyl chloroformate or Boc-anhydride with inexpensive methyl or ethyl chloroformate drastically lowers raw material costs. Additionally, the ability to use water as a solvent in the initial step reduces the reliance on costly anhydrous organic solvents, leading to substantial savings in material procurement. The high yield of the cyclization step, often reaching quantitative levels, ensures that valuable chiral starting materials are not wasted, maximizing the return on investment for every kilogram of input. Furthermore, the simplified workup reduces labor hours and utility consumption associated with distillation and chromatography, contributing to a leaner overall production cost structure.
- Enhanced Supply Chain Reliability: The starting materials for this process, including chiral amino alcohols and simple chloroformates, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor environmental fluctuations or equipment variability. This stability ensures consistent delivery timelines for downstream customers, fostering stronger long-term partnerships. Moreover, the reduced hazard profile of the reagents simplifies logistics and storage requirements, allowing for more flexible inventory management and faster response to market demand surges.
- Scalability and Environmental Compliance: This methodology is inherently scalable, having been demonstrated to work effectively from gram to multi-kilogram scales without loss of efficiency. The use of less toxic reagents and the generation of benign by-products align with increasingly stringent environmental regulations, facilitating easier permitting and compliance reporting. The potential for telescoping the two steps into a one-pot process further enhances scalability by reducing vessel turnover time and intermediate handling. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, ensuring that production capacity can be expanded rapidly to meet clinical or commercial needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, yield, and operational feasibility. Understanding these aspects is crucial for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: How does this method improve safety compared to traditional Evans auxiliary synthesis?
A: Traditional methods often require hazardous borane reductions under strictly anhydrous and anaerobic conditions. This patented process utilizes stable chloroformates and aqueous or alcoholic solvents, significantly reducing operational risks and eliminating the need for specialized air-free equipment.
Q: What are the yield advantages of using chloroformates over other acylating agents?
A: The use of methyl or ethyl chloroformates allows for near-quantitative conversion in the acylation step, avoiding the side reactions often seen with more bulky or expensive reagents like di-tert-butyl dicarbonate. The subsequent cyclization proceeds with total yields reaching 98-100%, minimizing material loss.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the protocol is designed for scalability. It employs inexpensive, commodity chemicals and avoids complex purification steps like column chromatography. The ability to perform the reaction in water or common organic solvents simplifies waste treatment and downstream processing for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazolidin-2-Ones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral auxiliaries play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral oxazolidin-2-ones meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis with precision, delivering materials that empower your R&D efforts.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
