Advanced Copper-Catalyzed Synthesis of 3-Chloro-4-Amino N-Phenylmaleimide Intermediates
The pharmaceutical industry is constantly seeking more sustainable and atom-economical pathways to construct complex heterocyclic scaffolds, particularly those containing halogenated motifs which are prevalent in bioactive molecules. A groundbreaking approach detailed in patent CN113372254A introduces a highly efficient preparation method for 3-chloro-4-amino N-phenylmaleimide compounds, leveraging a transition metal copper-catalyzed radical tandem reaction. This innovative strategy fundamentally shifts the paradigm of alkenyl chloride synthesis by utilizing secondary amine drug molecule hydrochlorides not merely as aminating reagents, but as bifunctional sources of both nitrogen and chlorine. By operating under an oxygen atmosphere in common organic solvents, this method achieves high yields and exceptional purity while circumventing the traditional reliance on stoichiometric amounts of hazardous chlorinating agents or expensive noble metal catalysts. The technical breakthrough lies in the ability to recycle the hydrochloric acid typically released as waste during amination reactions, transforming it into a valuable 'green' chlorine source for building critical chloride functionalities. This development represents a significant leap forward for process chemists aiming to streamline the synthesis of beta-chloroalkenylamine structures, which are essential intermediates in the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alkenyl chlorides has relied heavily on transition metal-catalyzed hydrochlorination of alkynes or cross-coupling reactions that often necessitate the use of pre-functionalized substrates and stoichiometric amounts of strong acids. These conventional routes frequently suffer from poor atom economy, as the hydrochloric acid generated during the neutralization of amine salts is discarded as chemical waste rather than being utilized in the synthetic transformation. Furthermore, many existing methods require the presence of auxiliary directing groups or expensive noble metal catalysts to achieve acceptable levels of regioselectivity and conversion, which drastically inflates the cost of goods and complicates the purification process. The reliance on harsh reaction conditions and specialized reagents often limits the scope of these reactions, making them unsuitable for late-stage functionalization of complex, sensitive drug molecules. Additionally, the separation of heavy metal residues and the treatment of acidic waste streams pose significant environmental and regulatory challenges for large-scale manufacturing facilities striving to meet green chemistry standards.
The Novel Approach
In stark contrast to these legacy methods, the novel copper-catalyzed protocol described in the patent offers a streamlined, one-pot solution that maximizes resource efficiency and minimizes environmental impact. By employing inexpensive cuprous bromide as the catalyst and molecular oxygen as the terminal oxidant, the reaction proceeds through a radical mechanism that effectively harnesses the hydrogen chloride liberated from the amine salt. This ingenious design allows for the direct chlorination of the maleimide double bond concurrent with the amination step, effectively killing two birds with one stone. The method demonstrates remarkable versatility, successfully accommodating a wide range of structurally diverse secondary amine hydrochlorides, including complex tricyclic antidepressants and other pharmacologically active scaffolds. For instance, the synthesis of complex derivatives like the maprotiline-based maleimide proceeds smoothly to afford the target product in high yield, showcasing the robustness of this catalytic system against steric hindrance and electronic variations. 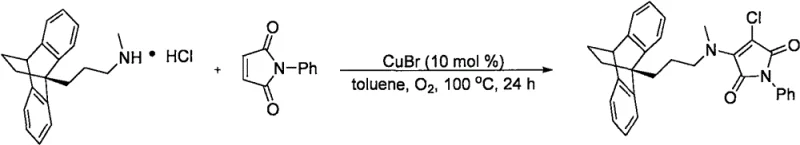
Mechanistic Insights into Copper-Catalyzed Radical Tandem Reaction
The core of this transformative chemistry lies in a sophisticated radical cascade initiated by the interaction between the copper catalyst and molecular oxygen. Under the optimized reaction conditions, the cuprous species activates the oxygen to generate reactive oxygen species, which subsequently facilitate the single-electron oxidation of the amine hydrochloride or the copper center itself. This redox event triggers the homolytic cleavage of the H-Cl bond within the amine salt, generating a chlorine radical that is poised for addition across the electron-deficient double bond of the N-phenylmaleimide. The resulting carbon-centered radical intermediate is then trapped by the amine nitrogen, closing the catalytic cycle and forging the new C-N and C-Cl bonds in a concerted fashion. This mechanism elegantly bypasses the need for external halogen sources, as the chlorine atom incorporated into the final product is derived directly from the hydrochloride counterion of the starting amine. The precise control over the radical propagation steps ensures high regioselectivity, favoring the formation of the 3-chloro-4-amino substitution pattern exclusively.
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms that might lead to polymerization or over-chlorination side products. The use of a mild copper catalyst and controlled oxygen pressure helps maintain a steady concentration of radical species, preventing runaway reactions that could degrade the sensitive maleimide core. Furthermore, the reaction conditions are sufficiently mild to preserve other sensitive functional groups present on the amine substrate, such as ether linkages or fluorine substituents, which are often prone to decomposition under strongly acidic or basic conditions. The high purity of the crude reaction mixtures observed in the patent examples suggests that the catalytic system is highly selective, minimizing the formation of by-products that would otherwise require extensive chromatographic purification. This selectivity is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metal residues must be adhered to, ensuring that the final intermediate meets the rigorous quality standards required for downstream drug synthesis.

How to Synthesize 3-Chloro-4-Amino N-Phenylmaleimide Efficiently
Executing this synthesis requires careful attention to the stoichiometry of the amine hydrochloride and the maintenance of an oxidative atmosphere throughout the reaction duration. The standard protocol involves mixing the N-phenylmaleimide substrate with a three-fold excess of the secondary amine hydrochloride and 10 mol% of cuprous bromide in toluene. The reaction vessel must be thoroughly degassed and backfilled with oxygen to ensure the radical chain reaction can propagate effectively without inhibition by atmospheric nitrogen. Heating the mixture to 100 °C for 24 hours allows sufficient time for the complete consumption of the starting materials and the formation of the thermodynamically stable chloro-amino product. Following the reaction, a simple workup procedure involving dilution with ethyl acetate, filtration to remove inorganic salts, and solvent evaporation yields a crude product that can be readily purified by flash column chromatography.
- Combine N-phenylmaleimide, secondary amine drug molecule hydrochloride (3.0 equiv), and cuprous bromide catalyst (10 mol%) in toluene solvent within a reaction vessel.
- Replace the atmosphere with oxygen three times to ensure an oxidative environment, then stir the mixture at 100 °C for 24 hours to facilitate the radical tandem reaction.
- Upon completion, cool the mixture, dilute with ethyl acetate, filter, remove solvent, and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply security. The elimination of expensive noble metal catalysts and proprietary chlorinating reagents significantly lowers the raw material costs associated with producing these specialized intermediates. Moreover, the ability to use commercially available amine hydrochlorides, which are often stable, inexpensive, and widely stocked, reduces the risk of supply chain disruptions caused by the scarcity of custom-synthesized precursors. The simplified post-treatment process, which avoids complex aqueous workups or neutralization steps, translates into reduced processing time and lower utility consumption at the manufacturing scale. These operational efficiencies collectively contribute to a more robust and cost-effective supply chain for high-value pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with abundant copper salts results in a drastic reduction in catalyst costs, while the dual-use of amine hydrochlorides eliminates the need for purchasing separate chlorinating agents. This consolidation of reagents simplifies inventory management and reduces the overall chemical spend per kilogram of product. Additionally, the high atom economy of the reaction means less raw material is wasted, further driving down the variable costs of production. The use of toluene, a commodity solvent, instead of specialized polar aprotic solvents that may require recycling or special disposal, adds another layer of economic benefit to the process.
- Enhanced Supply Chain Reliability: By relying on generic, off-the-shelf starting materials like N-phenylmaleimide and common amine salts, manufacturers can mitigate the risks associated with single-source suppliers or long lead times for custom intermediates. The robustness of the reaction conditions allows for flexibility in sourcing, as slight variations in reagent grade are less likely to impact the outcome compared to sensitive organometallic couplings. This resilience ensures a consistent flow of materials to downstream production lines, safeguarding against delays that could impact the launch of new drug candidates. The stability of the amine hydrochloride salts also facilitates easier storage and transportation, reducing logistical complexities.
- Scalability and Environmental Compliance: The straightforward nature of this radical tandem reaction makes it highly amenable to scale-up from gram to multi-kilogram quantities without the need for specialized equipment or cryogenic conditions. The absence of stoichiometric acid waste and heavy metal contaminants simplifies the wastewater treatment process, helping facilities meet increasingly stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing organization. The ability to run the reaction under atmospheric oxygen pressure rather than high-pressure gases further lowers the barrier for safe industrial implementation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this copper-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative studies presented in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is critical for process development teams evaluating this technology for integration into their existing manufacturing workflows.
Q: Why is cuprous bromide preferred over other copper salts for this transformation?
A: Experimental data indicates that while various copper salts like cupric acetate and cuprous iodide show activity, cuprous bromide provides the optimal balance of catalytic efficiency and yield, achieving up to 89% conversion in complex substrates compared to lower yields with fluoride or acetate variants.
Q: What is the significance of using amine hydrochlorides instead of free amines?
A: Utilizing amine hydrochlorides serves a dual purpose: it acts as the nitrogen source for amination and simultaneously provides the chlorine source for chlorination via the released hydrochloric acid, thereby eliminating the need for external chlorinating agents and reducing hazardous waste generation.
Q: Can this method be applied to complex drug molecules?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully modifying complex pharmaceutical scaffolds such as maprotiline, nortriptyline, and paroxetine derivatives without requiring protective groups or harsh conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloro-4-Amino N-Phenylmaleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in accelerating drug discovery and development timelines. Our team of expert process chemists has extensively evaluated the copper-catalyzed radical tandem reaction described in CN113372254A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art facilities are designed to handle complex radical chemistries safely, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest industry standards. We understand that every project is unique, and our flexible manufacturing infrastructure allows us to adapt quickly to your specific volume and purity requirements.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient synthetic route. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you optimize your supply chain and reduce your time-to-market with our superior manufacturing capabilities and deep technical expertise in pharmaceutical intermediate synthesis.
