Advanced Nor-UDCA Manufacturing: Leveraging CO2-Assisted Oxidative Cleavage for Commercial Scale-Up
The pharmaceutical landscape for treating cholestatic liver diseases, particularly Primary Sclerosing Cholangitis (PSC), has seen a paradigm shift with the emergence of Nor-UDCA as a superior therapeutic candidate compared to traditional Ursodeoxycholic Acid (UDCA). Recent intellectual property developments, specifically patent CN115806578A, have unveiled a groundbreaking synthetic methodology that addresses long-standing bottlenecks in the manufacturing of this critical bile acid derivative. This patent introduces a novel pathway starting from Phocacholic Acid (PhCA), utilizing a unique CO2-assisted oxidative cleavage mechanism to efficiently shorten the side chain from C24 to C23. For R&D directors and procurement specialists in the fine chemical sector, this innovation represents a pivotal opportunity to secure a more robust and cost-effective supply chain for high-purity pharmaceutical intermediates. The technical nuances of this route not only enhance reaction selectivity but also simplify the purification protocols, thereby aligning perfectly with the stringent quality requirements of modern API manufacturing.
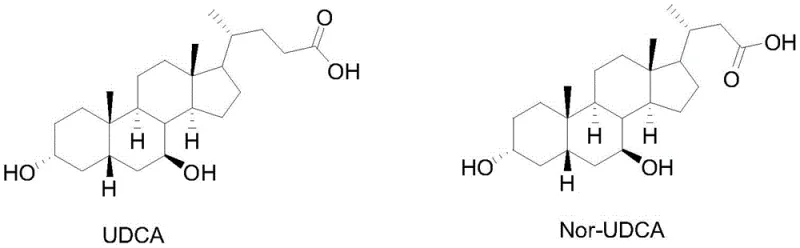
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nor-UDCA has relied heavily on Ursodeoxycholic Acid (UDCA) as the starting material, a strategy that inherently limits economic viability due to the high cost and limited availability of the precursor. Conventional routes, such as those disclosed in earlier patents like WO2012/07268A1, typically involve a multi-step sequence that includes protection and de-protection strategies, leading to accumulated yield losses and increased waste generation. These traditional methods often require harsh reagents for side-chain degradation, which can compromise the stereochemical integrity of the steroid nucleus, resulting in difficult-to-remove impurities that complicate downstream processing. Furthermore, the reliance on expensive starting materials creates a fragile supply chain vulnerable to market fluctuations, making it challenging for manufacturers to offer competitive pricing for the final active pharmaceutical ingredient. The environmental footprint of these older processes is also significant, often involving heavy metal oxidants or chlorinated solvents that necessitate complex waste treatment protocols.
The Novel Approach
In stark contrast, the methodology described in CN115806578A leverages Phocacholic Acid (PhCA), a more accessible and economically favorable raw material, to achieve the desired C23 skeleton through a direct oxidative cleavage strategy. The core innovation lies in the use of carbon dioxide gas sparging during the sodium hypochlorite oxidation step, which serves as a dynamic pH control mechanism without the need for additional buffering salts. This approach facilitates the selective conversion of the C24 side chain into a C23 aldehyde intermediate with remarkable efficiency, as demonstrated by yields reaching up to 80% in optimized embodiments. By eliminating the need for complex protecting groups and utilizing mild, aqueous-based oxidation conditions, this novel route drastically reduces the number of unit operations and solvent consumption. The transition from PhCA to the key aldehyde intermediate sets a solid foundation for the subsequent steps, ensuring that the overall process remains linear, scalable, and environmentally compliant.
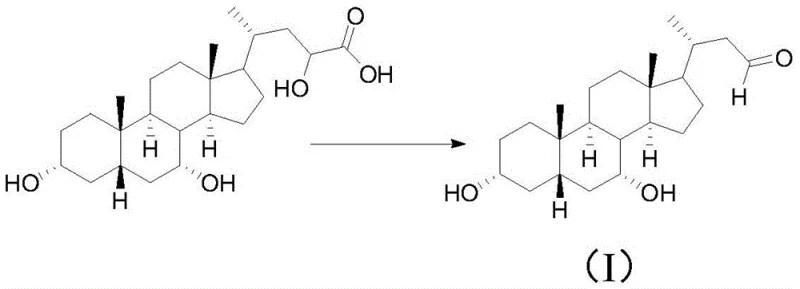
Mechanistic Insights into CO2-Assisted Oxidative Cleavage
The mechanistic elegance of this synthesis centers on the interaction between carbon dioxide, water, and sodium hypochlorite within the reaction matrix. When CO2 is continuously sparged into the methanol-water solvent system containing PhCA, it dissolves to form carbonic acid, establishing a mildly acidic environment that is crucial for the generation of hypochlorous acid (HOCl) from sodium hypochlorite. This in situ generation of the active oxidant ensures a steady, controlled concentration that prevents the violent exotherms often associated with bulk hypochlorite additions. The mild acidity also protonates intermediate species, facilitating the specific cleavage of the terminal carbon-carbon bond on the side chain while preserving the sensitive hydroxyl groups on the steroid ring system. This precise control over the reaction milieu minimizes over-oxidation to carboxylic acids at the early stages, allowing for the isolation of the aldehyde intermediate which is pivotal for the subsequent chain shortening logic. Such mechanistic control is essential for maintaining high purity profiles, as it suppresses the formation of chlorinated by-products that are notoriously difficult to separate from the target molecule.
Furthermore, the continuous flow of CO2 acts as an inert blanket, displacing oxygen and minimizing the risk of radical-mediated side reactions that could degrade the steroid backbone. As the reaction progresses from the aldehyde intermediate to the Nor-CDCA acid and subsequently to the 7-keto derivative, the oxidation state is carefully managed through sequential adjustments in reagent concentration and temperature. The final transformation involves a stereoselective catalytic hydrogenation using palladium on carbon under pressurized hydrogen, where the presence of pyridine plays a critical role in directing the hydride attack to the beta-face of the C7 ketone. This ensures the formation of the therapeutically active 7-beta-hydroxy configuration characteristic of Nor-UDCA. The synergy between the oxidative cleavage and the stereoselective reduction highlights a sophisticated understanding of bile acid chemistry, enabling the production of a high-value intermediate with minimal epimeric contamination.
How to Synthesize Nor-UDCA Efficiently
The practical execution of this synthesis requires careful attention to gas flow rates and reagent addition profiles to maximize the benefits of the CO2-assisted mechanism. Operators must maintain a consistent CO2 sparge rate, typically between 2 mL/min and 10 mL/min per 20 mL of reaction volume, to ensure the pH remains within the optimal window throughout the oxidation phase. The detailed standardized synthesis steps, including specific molar ratios, temperature gradients, and workup procedures required to replicate the high yields reported in the patent data, are outlined below for technical reference.
- Dissolve Phocacholic Acid (PhCA) in a methanol/water solvent system and initiate oxidative cleavage by sparging CO2 gas while adding sodium hypochlorite to generate the C23-aldehyde intermediate.
- Convert the aldehyde intermediate to Nor-CDCA through further oxidation under controlled CO2 flow and elevated temperatures, ensuring high purity before the next stage.
- Perform selective oxidation at the C7 position to form the 7-keto derivative, followed by stereoselective catalytic hydrogenation using Pd/C to yield the final Nor-UDCA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PhCA-based route offers transformative advantages in terms of cost structure and supply security. The shift away from UDCA as a starting material immediately decouples the production cost from the volatile pricing of premium bile acids, allowing for a more predictable and lower cost of goods sold (COGS). The use of commodity chemicals like sodium hypochlorite and carbon dioxide, rather than specialized oxidants or exotic catalysts, further drives down raw material expenses and simplifies logistics. Since the process avoids the use of heavy metal catalysts in the oxidation steps, the burden on wastewater treatment facilities is significantly reduced, leading to lower operational expenditures related to environmental compliance and waste disposal. This streamlined chemical profile makes the process highly attractive for large-scale manufacturing where margin compression is a constant challenge.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials and the reduction in synthetic steps directly translate to substantial cost savings in pharmaceutical intermediate manufacturing. By utilizing Phocacholic Acid, which is generally more abundant and less processed than UDCA, manufacturers can achieve a significantly lower entry price point for the raw material basket. Additionally, the high selectivity of the CO2-assisted oxidation reduces the need for extensive chromatographic purification, which is often the most costly and time-consuming part of fine chemical production. The ability to perform reactions in aqueous methanol mixtures also reduces the consumption of expensive organic solvents, contributing to a leaner and more cost-efficient production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Diversifying the raw material base to include PhCA mitigates the risk of supply disruptions associated with single-source dependencies on UDCA. The reagents required for this synthesis, such as sodium hypochlorite and carbon dioxide, are globally available commodities with robust supply networks, ensuring that production schedules are not held hostage by niche chemical shortages. The simplicity of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum systems, means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant requalification efforts. This flexibility strengthens the resilience of the supply chain, guaranteeing consistent delivery timelines for downstream API producers.
- Scalability and Environmental Compliance: The inherent safety of the CO2-sparging technique makes this route exceptionally scalable from pilot plant to commercial tonnage without encountering the heat transfer limitations typical of batch oxidations. The aqueous nature of the reaction medium aligns well with green chemistry principles, reducing the emission of volatile organic compounds (VOCs) and simplifying the handling of reaction masses. Because the process generates fewer halogenated by-products and avoids toxic heavy metals, the effluent stream is easier to treat, helping manufacturers meet increasingly stringent environmental regulations. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Nor-UDCA synthesis pathway. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's feasibility.
Q: What is the primary advantage of using Phocacholic Acid over UDCA for Nor-UDCA synthesis?
A: Using Phocacholic Acid (PhCA) as the starting material allows for a direct side-chain shortening strategy via oxidative cleavage, bypassing the need for expensive Ursodeoxycholic Acid (UDCA) and reducing the total number of synthetic steps required to reach the C23 backbone.
Q: How does the CO2-assisted oxidation method improve process safety and yield?
A: The continuous introduction of CO2 gas creates a self-buffering acidic environment that controls the reactivity of sodium hypochlorite, preventing over-oxidation and exothermic runaway risks while significantly improving the selectivity for the desired aldehyde intermediate.
Q: Is this synthesis route scalable for industrial production of API intermediates?
A: Yes, the process utilizes common reagents like sodium hypochlorite and standard unit operations such as gas sparging and catalytic hydrogenation, making it highly amenable to commercial scale-up from kilogram to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nor-UDCA Supplier
The technical potential of the CO2-assisted oxidative cleavage route positions Nor-UDCA as a commercially viable candidate for widespread therapeutic application, provided it is manufactured by a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling pressurized hydrogenation and gas-liquid reactions safely, ensuring that the specific conditions required for this synthesis—such as controlled CO2 sparging and 5 MPa hydrogen pressure—are met with precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor the stereochemical integrity of the 7-beta-hydroxyl group and the absence of chlorinated impurities.
We invite global pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this innovative patent technology, we can help you optimize your supply chain for Nor-UDCA and its intermediates. Please contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how we can support your development and commercialization goals with reliability and excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
