Advanced Synthesis of 2-Methoxymethyl-1,4-Diphenylamine for Commercial Oxidative Hair Colorant Production
Advanced Synthesis of 2-Methoxymethyl-1,4-Diphenylamine for Commercial Oxidative Hair Colorant Production
The global demand for high-performance oxidative hair colorants continues to drive innovation in the synthesis of key aromatic amine intermediates. Patent CN103124718A introduces a transformative methodology for preparing 2-substituted-1,4-diphenylamines, specifically targeting 2-methoxymethyl-1,4-diphenylamine, which serves as a critical coupler in modern hair dye formulations. This technical disclosure outlines a robust pathway that bypasses the cumbersome protection-deprotection strategies of legacy technologies, offering a direct route from readily available chlorinated precursors. For R&D directors and procurement specialists, understanding the mechanistic nuances and supply chain implications of this patent is essential for securing a competitive edge in the personal care chemical market. The following analysis dissects the chemical engineering breakthroughs and commercial viability embedded within this intellectual property.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted-1,4-diphenylamines has relied on multi-step sequences that are both time-consuming and economically inefficient. As illustrated in prior art such as US Patent 4,997,451 and US Patent 6,648,923, traditional routes often necessitate the use of protecting groups to manage the reactivity of multiple functional sites on the benzene ring. 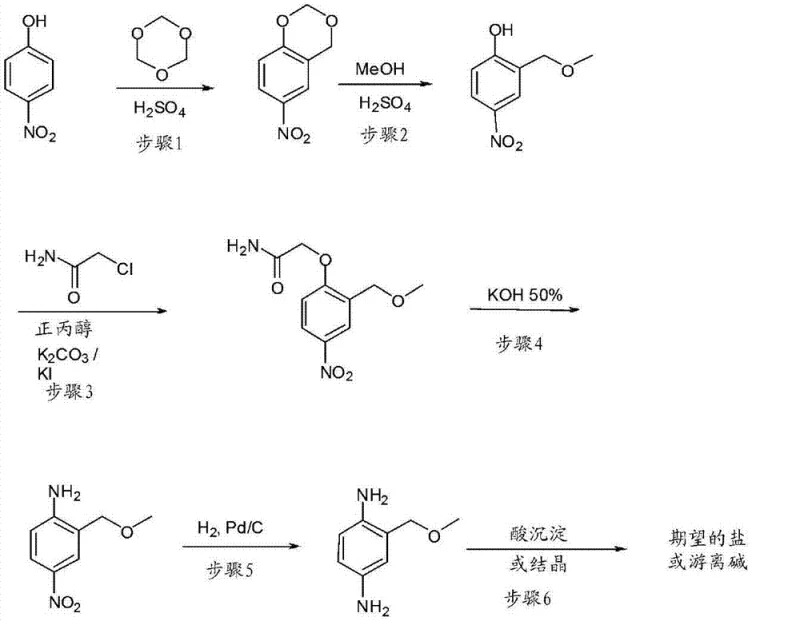 These legacy processes typically involve the initial protection of phenolic hydroxyl groups, followed by nitration, reduction, and subsequent deprotection, which significantly increases the number of unit operations. Each additional step introduces potential yield losses, generates higher volumes of chemical waste, and complicates the purification landscape, ultimately inflating the cost of goods sold (COGS). Furthermore, the reliance on specialized reagents for protection and deprotection creates supply chain vulnerabilities, as these materials may be subject to market volatility or regulatory scrutiny. For a procurement manager, these factors translate into higher raw material costs and less predictable lead times, making the conventional approach less attractive for large-scale commercial manufacturing.
These legacy processes typically involve the initial protection of phenolic hydroxyl groups, followed by nitration, reduction, and subsequent deprotection, which significantly increases the number of unit operations. Each additional step introduces potential yield losses, generates higher volumes of chemical waste, and complicates the purification landscape, ultimately inflating the cost of goods sold (COGS). Furthermore, the reliance on specialized reagents for protection and deprotection creates supply chain vulnerabilities, as these materials may be subject to market volatility or regulatory scrutiny. For a procurement manager, these factors translate into higher raw material costs and less predictable lead times, making the conventional approach less attractive for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN103124718A leverages the activating power of the nitro group to facilitate direct nucleophilic aromatic substitution, thereby streamlining the entire synthetic sequence. 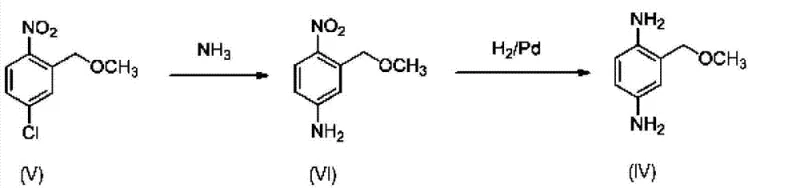 This innovative approach eliminates the need for protecting groups entirely, allowing for the direct introduction of the amino moiety onto the nitro-activated chlorobenzene scaffold. By starting from cost-effective precursors like 4-nitro-2-substituted chlorobenzene, the process reduces the total number of reaction steps, which directly correlates to improved overall yield and reduced operational expenditure. The strategic placement of the methoxymethyl group prior to the ammoniation step ensures that the subsequent nucleophilic attack by ammonia occurs selectively at the position ortho to the nitro group, driven by strong electronic activation. This simplification not only enhances the atom economy of the process but also significantly lowers the environmental footprint by minimizing solvent usage and waste generation. For supply chain heads, this translates to a more resilient manufacturing model with fewer dependency points and a faster time-to-market for the final active ingredient.
This innovative approach eliminates the need for protecting groups entirely, allowing for the direct introduction of the amino moiety onto the nitro-activated chlorobenzene scaffold. By starting from cost-effective precursors like 4-nitro-2-substituted chlorobenzene, the process reduces the total number of reaction steps, which directly correlates to improved overall yield and reduced operational expenditure. The strategic placement of the methoxymethyl group prior to the ammoniation step ensures that the subsequent nucleophilic attack by ammonia occurs selectively at the position ortho to the nitro group, driven by strong electronic activation. This simplification not only enhances the atom economy of the process but also significantly lowers the environmental footprint by minimizing solvent usage and waste generation. For supply chain heads, this translates to a more resilient manufacturing model with fewer dependency points and a faster time-to-market for the final active ingredient.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Hydrogenation
The core chemical transformation in this novel pathway is the nucleophilic aromatic substitution (SnAr) of a chlorine atom by ammonia, a reaction that is kinetically sluggish under normal conditions but becomes highly favorable in the presence of strong electron-withdrawing groups. In the specific case of 4-nitro-2-methoxymethylchlorobenzene, the nitro group at the para-position exerts a powerful inductive and resonance effect that stabilizes the Meisenheimer complex intermediate formed during the attack of the ammonia nucleophile. This electronic activation allows the reaction to proceed at elevated temperatures, typically between 150°C and 220°C, under autogenous pressure in a sealed reactor system. The choice of solvent, such as formamide or acetonitrile, plays a critical role in solubilizing the reactants and stabilizing the transition state, ensuring high conversion rates. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters, as slight deviations in temperature or pressure can impact the ratio of desired product to potential by-products like diarylamines formed from over-alkylation.
Following the successful introduction of the amino group, the final step involves the catalytic hydrogenation of the remaining nitro group to an amine, completing the 1,4-diamine structure. This reduction is typically carried out using heterogeneous catalysts such as palladium on carbon (Pd/C) or Raney nickel under a hydrogen atmosphere of approximately 50-60 psig. The chemoselectivity of this step is paramount; the catalyst must reduce the nitro group efficiently without affecting the ether linkage or causing hydrogenolysis of the benzylic C-O bond. The patent specifies conditions that maintain mild temperatures, generally between 20°C and 80°C, to preserve the integrity of the methoxymethyl substituent. From a quality control perspective, monitoring the hydrogen uptake rate is essential to determine the reaction endpoint and prevent over-reduction or catalyst poisoning. The resulting crude product can then be purified via acid precipitation, forming stable salts such as the malate salt, which enhances the stability and handling properties of the final intermediate for downstream formulation into hair dye kits.
How to Synthesize 2-Methoxymethyl-1,4-Diphenylamine Efficiently
The practical implementation of this synthesis route requires careful attention to the sequential addition of reagents and the control of exothermic events, particularly during the nitration and etherification stages. The process begins with the nitration of 2-chlorobenzyl chloride using concentrated nitric acid and sulfuric acid at low temperatures to prevent oxidation of the benzylic position, followed by an etherification step where the chloromethyl group is converted to a methoxymethyl group using sodium methoxide in methanol. 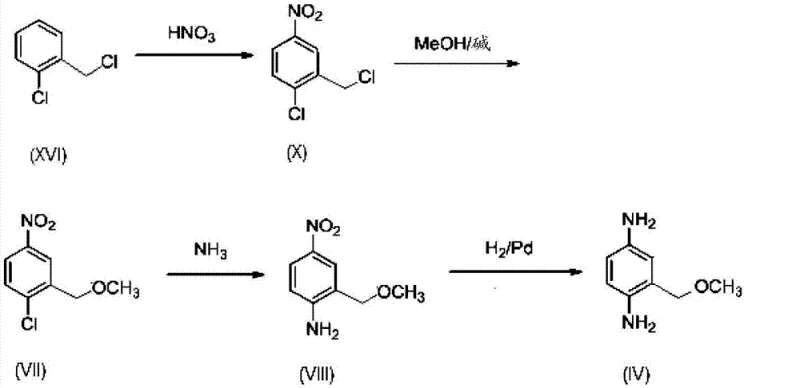 This sequence generates the key intermediate, 4-nitro-2-methoxymethylchlorobenzene, which is then subjected to the high-pressure ammoniation step described previously. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and workup procedures, are critical for reproducibility and safety. Operators must be trained to handle liquid ammonia and high-pressure reactors in compliance with all relevant safety regulations. The final isolation of the product often involves crystallization from organic solvents or precipitation as a salt, ensuring that the purity specifications meet the stringent requirements for cosmetic applications. For a comprehensive guide on the exact operational parameters and safety protocols, please refer to the structured synthesis guide below.
This sequence generates the key intermediate, 4-nitro-2-methoxymethylchlorobenzene, which is then subjected to the high-pressure ammoniation step described previously. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and workup procedures, are critical for reproducibility and safety. Operators must be trained to handle liquid ammonia and high-pressure reactors in compliance with all relevant safety regulations. The final isolation of the product often involves crystallization from organic solvents or precipitation as a salt, ensuring that the purity specifications meet the stringent requirements for cosmetic applications. For a comprehensive guide on the exact operational parameters and safety protocols, please refer to the structured synthesis guide below.
- Perform nitration of 2-chlorobenzyl chloride followed by etherification with methanol to form 4-nitro-2-methoxymethylchlorobenzene.
- Conduct nucleophilic aromatic substitution using liquid ammonia at elevated temperatures (150-220°C) to introduce the amino group.
- Execute catalytic hydrogenation using Pd/C under moderate pressure to reduce the nitro group and finalize the diamine structure.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this patented synthesis route offers substantial strategic benefits for organizations looking to optimize their sourcing strategies for oxidative hair dye intermediates. By shifting away from complex, multi-step legacy processes to this streamlined approach, companies can achieve significant cost reductions in manufacturing without compromising on product quality or purity. The elimination of protecting group chemistry removes the need for expensive reagents and the associated waste disposal costs, directly improving the gross margin of the final product. Furthermore, the use of commodity starting materials like 2-chlorobenzyl chloride ensures a stable and reliable supply base, reducing the risk of production stoppages due to raw material shortages. This resilience is crucial for supply chain heads who must guarantee continuous delivery to downstream formulators in the highly competitive personal care market.
- Cost Reduction in Manufacturing: The simplified process flow significantly lowers operational expenditures by reducing the number of reaction vessels, filtration steps, and solvent exchanges required. Since the route avoids the use of specialized protecting agents and the associated deprotection reagents, the overall material cost per kilogram of product is drastically reduced. Additionally, the high atom economy of the direct substitution reaction means less raw material is wasted as by-products, further enhancing the economic efficiency of the process. These savings can be passed on to customers or reinvested into R&D to develop next-generation colorant technologies.
- Enhanced Supply Chain Reliability: Relying on widely available bulk chemicals such as chlorobenzene derivatives and methanol mitigates the risk of supply chain disruptions often associated with niche fine chemical intermediates. The robustness of the reaction conditions allows for flexible manufacturing schedules, enabling producers to scale up or down based on market demand without extensive retooling. This flexibility ensures that procurement managers can secure long-term contracts with favorable terms, knowing that the production technology is not dependent on single-source suppliers for exotic reagents. Consequently, lead times for high-purity dye intermediates can be shortened, improving responsiveness to customer needs.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard chemical engineering unit operations that are well-understood in the industry. The reduction in waste generation aligns with increasingly stringent environmental regulations, lowering the compliance burden and associated fees for waste treatment. By minimizing the use of hazardous solvents and reagents, the facility's environmental footprint is reduced, supporting corporate sustainability goals. This eco-friendly profile is becoming a key differentiator in the B2B chemical market, where end-brands are demanding greener supply chains for their consumer products.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of frequently asked questions based on the patent data and industry standards. These answers provide clarity on safety, purity, and scalability, helping stakeholders make informed decisions about integrating this intermediate into their supply chains. Understanding these details is crucial for risk assessment and quality assurance planning.
Q: What are the key safety considerations for the ammoniation step in this synthesis?
A: The ammoniation step involves liquid ammonia at pressures between 75-100 psig and temperatures up to 220°C. Strict pressure vessel protocols and leak detection systems are mandatory to ensure operational safety during this high-energy nucleophilic substitution.
Q: How does this patent route improve impurity profiles compared to conventional methods?
A: By utilizing direct nucleophilic substitution on a nitro-activated ring, the process avoids complex protection and deprotection sequences found in older US patents. This reduction in synthetic steps minimizes the formation of side-products and simplifies downstream purification, leading to a cleaner impurity profile.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process relies on commodity starting materials like 2-chlorobenzyl chloride and uses standard unit operations such as nitration, etherification, and catalytic hydrogenation. The elimination of exotic reagents and the robustness of the reaction conditions make it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxymethyl-1,4-Diphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance and safety of final consumer products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methoxymethyl-1,4-diphenylamine meets the highest industry standards for oxidative hair coloration. Our commitment to excellence extends beyond mere compliance; we actively collaborate with clients to optimize formulations and troubleshoot processing challenges, acting as a true extension of your R&D department.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. By leveraging our expertise in this patented synthesis route, we can help you identify opportunities to reduce costs and improve efficiency in your supply chain. Please reach out to us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your growth in the global personal care market.
