Advanced Manufacturing of Amorolfine Hydrochloride via Optimized Heck Reaction and Reductive Amination
Introduction to Advanced Amorolfine Hydrochloride Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes that balance high yield with environmental sustainability, particularly for critical antifungal agents like Amorolfine Hydrochloride. Patent CN102887872A introduces a refined methodology that addresses historical inefficiencies in the production of this key active pharmaceutical ingredient (API) intermediate. By leveraging a specific combination of N-Methylpyrrolidone (NMP) as a reaction medium and sodium triacetoxyborohydride as a selective reducing agent, this technology significantly mitigates the ecological footprint associated with traditional synthesis. The process is designed to overcome the limitations of prior art, such as the use of toxic solvents like DMF and the safety hazards of high-pressure hydrogenation. For global supply chain leaders, this represents a pivotal shift towards greener chemistry without compromising the stringent purity specifications required for regulatory approval. The integration of these optimized conditions ensures a reliable supply of high-quality intermediates, positioning manufacturers to meet the growing demand for effective antifungal treatments while adhering to increasingly rigorous environmental, health, and safety (EHS) standards.
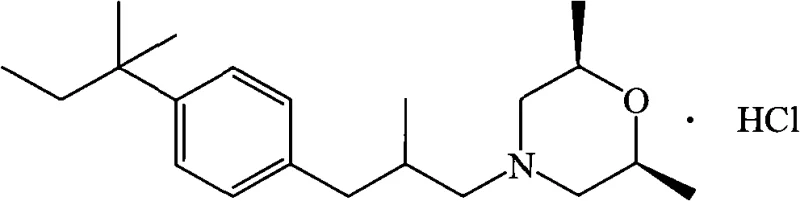
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Amorolfine Hydrochloride has been plagued by significant technical and environmental hurdles that impact both cost and scalability. Early methodologies, such as those disclosed in US Patent No. 7795425B2, relied on complex condensation reactions followed by Heck couplings that often suffered from multiple rearrangement side reactions. These side pathways not only reduced the overall yield but also generated a difficult-to-separate impurity profile, necessitating extensive and costly purification steps. Furthermore, alternative routes described in international applications like WO 2007113218A1 utilized Dimethylformamide (DMF) as the primary solvent. While effective for solubility, DMF is classified as a reproductive toxin and exhibits poor biodegradability, with a BOD5/COD ratio as low as 0.065, creating substantial wastewater treatment burdens. Additionally, the reliance on palladium-on-carbon catalysts for reduction steps mandates the use of high-pressure reactors, introducing significant capital expenditure and operational safety risks related to hydrogen handling. These factors collectively render conventional methods less attractive for modern, large-scale commercial production where efficiency and sustainability are paramount.
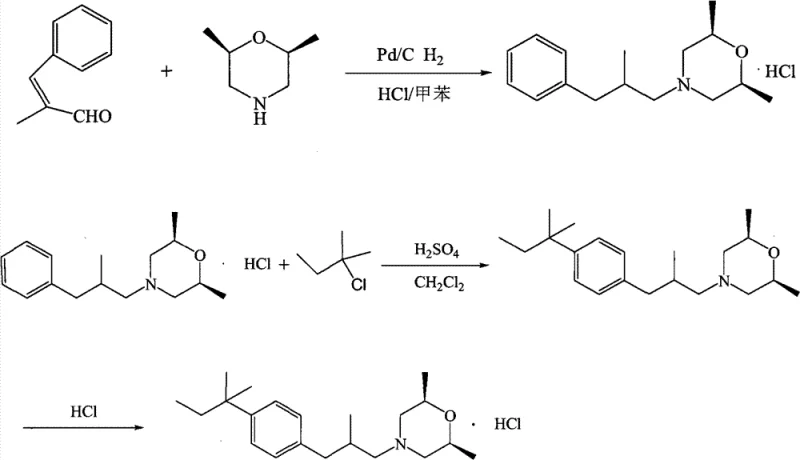
The Novel Approach
The innovative strategy outlined in CN102887872A fundamentally reengineers the synthetic pathway to eliminate these bottlenecks. By substituting DMF with N-Methylpyrrolidone (NMP), the process utilizes a solvent with a higher boiling point (202-204°C) and superior thermal stability, allowing for more efficient reaction kinetics at elevated temperatures without decomposition. Crucially, NMP is far more environmentally benign, possessing a BOD5/COD value of approximately 1.71, which facilitates biological degradation of waste streams and drastically lowers effluent treatment costs. In the reduction phase, the protocol replaces hazardous high-pressure hydrogenation with sodium triacetoxyborohydride. This reagent operates under mild atmospheric conditions, eliminating the need for specialized pressure vessels and removing the risk of uncontrolled hydrogen evolution. The result is a streamlined, safer process that delivers superior selectivity, minimizing byproduct formation and ensuring a consistent, high-purity output suitable for direct pharmaceutical application.
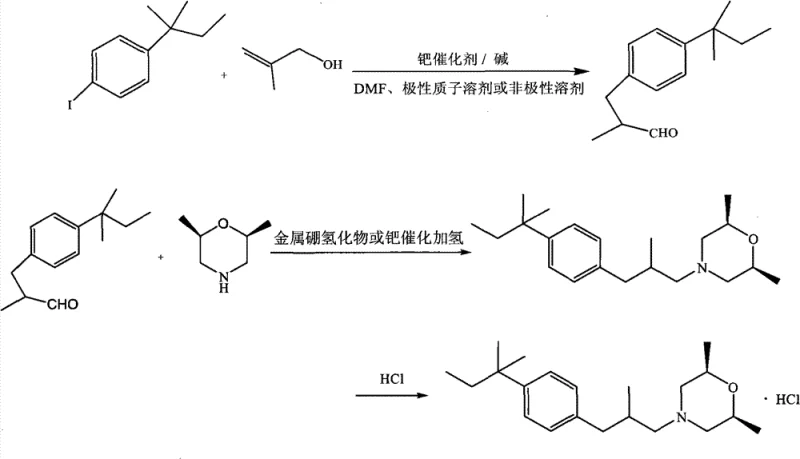
Mechanistic Insights into Mizoroki-Heck Coupling and Reductive Amination
The core of this advanced synthesis lies in the precise execution of the Mizoroki-Heck reaction, which couples 4-iodo-tert-amylbenzene with 2-methylallyl alcohol. In the presence of a palladium catalyst—such as palladium chloride or tetrakis(triphenylphosphine)palladium—and a mild base like sodium bicarbonate, the reaction proceeds through a classic catalytic cycle involving oxidative addition, migratory insertion, and beta-hydride elimination. The choice of NMP as the solvent is mechanistically critical; its high polarity and coordinating ability stabilize the palladium species, preventing premature catalyst deactivation and ensuring high turnover numbers. This stability allows the reaction to proceed efficiently at temperatures between 80°C and 160°C, driving the conversion of the aryl iodide to the corresponding aldehyde intermediate, 3-tert-pentylphenyl-2-methyl propanal, with exceptional fidelity. The careful control of stoichiometry, specifically maintaining a molar ratio of substrate to alcohol between 1:1.0 and 1:5.0, further suppresses oligomerization side reactions, ensuring that the aldehyde is formed with minimal impurities.
Following the coupling step, the process employs a Borch reduction mechanism for the reductive amination of the aldehyde with cis-2,6-dimethylmorpholine. Unlike traditional hydride reductions that might reduce the carbonyl to an alcohol, sodium triacetoxyborohydride selectively reduces the intermediate iminium ion formed between the amine and the aldehyde. This selectivity is driven by the mild acidity of the reaction environment, typically maintained with glacial acetic acid at temperatures between 0°C and 25°C. The mechanism involves the initial formation of a Schiff base, which is protonated to form the reactive iminium species. The hydride donor then transfers a hydride ion specifically to the iminium carbon, yielding the tertiary amine product, amorolfine. This chemoselectivity is vital for maintaining the integrity of the molecular structure and avoiding the formation of alcohol byproducts that would complicate downstream purification. The final step involves simple acidification to form the stable hydrochloride salt, completing the transformation with high atom economy.
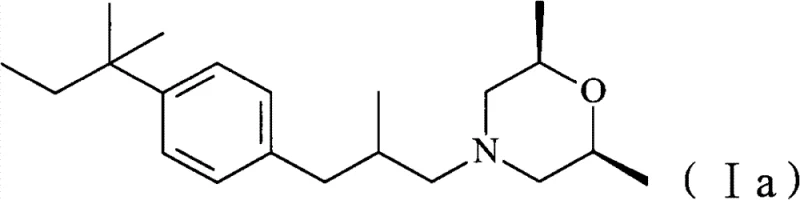
How to Synthesize Amorolfine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive palladium catalyst. The Heck coupling is monitored via gas chromatography (GC) to determine the precise endpoint, ensuring complete consumption of the starting iodide before proceeding. Once the aldehyde intermediate is secured, either through isolation or a telescoped one-pot procedure, the temperature is carefully lowered for the reductive amination step. The addition of sodium triacetoxyborohydride must be controlled to manage the exotherm, although it is significantly milder than metal hydride alternatives. Detailed standard operating procedures regarding reagent addition rates, temperature ramping, and workup protocols are essential for operators to achieve the reported yields exceeding 83%.
- Perform a Mizoroki-Heck reaction between 4-iodo-tert-amylbenzene and 2-methylallyl alcohol using NMP solvent and a palladium catalyst.
- Conduct reductive amination of the resulting aldehyde with cis-2,6-dimethylmorpholine using sodium triacetoxyborohydride.
- Convert the free base amorolfine into amorolfine hydrochloride via acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The transition from DMF to NMP represents a significant reduction in environmental compliance costs. Because NMP is readily biodegradable, the expense associated with treating hazardous wastewater is drastically minimized, directly impacting the bottom line of manufacturing operations. Furthermore, the elimination of high-pressure hydrogenation equipment removes a major capital barrier and reduces the ongoing maintenance and safety inspection costs associated with pressure vessels. This simplification of the infrastructure requirements allows for more flexible manufacturing setups and reduces the risk of production stoppages due to equipment failure or safety audits. The robustness of the process also translates to a more reliable supply continuity, as the reagents used are commodity chemicals with stable global availability, insulating the supply chain from the volatility often seen with specialized catalysts or gases.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts and high-pressure hydrogen gas with sodium triacetoxyborohydride results in substantial operational savings. This reagent is not only safer to handle but also eliminates the need for complex gas handling systems and the associated energy consumption for compression. Additionally, the high selectivity of the reduction step minimizes the loss of valuable starting materials to side products like alcohols, thereby improving the overall mass balance and reducing the cost per kilogram of the final API intermediate. The simplified purification process, driven by higher crude purity, further reduces solvent consumption and processing time, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By utilizing widely available solvents like NMP and ethyl acetate, along with stable solid reagents, the manufacturing process becomes less susceptible to logistical disruptions. The avoidance of gaseous hydrogen removes a critical dependency on industrial gas suppliers and the specialized transport infrastructure they require. This decentralization of supply needs enhances the resilience of the production network, ensuring that manufacturing can continue uninterrupted even during broader supply chain fluctuations. The consistent quality of the output, with purity levels consistently above 99.5%, also reduces the likelihood of batch rejections, ensuring a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The thermal stability of NMP and the mild conditions of the reductive amination make this process highly scalable from pilot plant to commercial tonnage. The absence of cryogenic conditions or extreme pressures simplifies the engineering requirements for scale-up, allowing for faster technology transfer. From an environmental perspective, the improved biodegradability of the waste stream aligns with global sustainability goals and regulatory frameworks like REACH. This proactive approach to green chemistry not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific improvements detailed in the patent documentation, focusing on solvent selection, safety profiles, and product quality metrics. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement teams assessing the reliability of suppliers utilizing this technology.
Q: Why is NMP preferred over DMF in this synthesis?
A: N-Methylpyrrolidone (NMP) offers superior thermal stability and biodegradability compared to DMF. Its BOD5/COD value is significantly higher, making wastewater treatment easier and more environmentally compliant.
Q: What are the safety advantages of using sodium triacetoxyborohydride?
A: Sodium triacetoxyborohydride avoids the need for high-pressure hydrogenation equipment required by palladium carbon catalysts and eliminates the violent hydrogen gas evolution associated with metal borohydrides, enhancing operational safety.
Q: What purity levels can be achieved with this method?
A: The optimized process described in patent CN102887872A consistently achieves HPLC purity exceeding 99.5% with total yields often surpassing 83%, suitable for strict pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amorolfine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102887872A are fully realized in practice. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal and safety requirements of the Heck coupling and reductive amination steps described. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques like HPLC and GC to verify that every batch of Amorolfine Hydrochloride meets the >99.5% purity benchmark required by global pharmacopeias. Our commitment to quality assurance ensures that the material you receive is consistent, safe, and ready for immediate use in your final drug product formulation.
We invite you to collaborate with us to leverage this optimized technology for your supply chain. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this NMP-based process for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your project timelines. By partnering with NINGBO INNO PHARMCHEM, you secure not just a chemical supplier, but a strategic ally dedicated to driving innovation and efficiency in the production of high-value pharmaceutical intermediates.
