Scalable Palladium-Catalyzed Synthesis of 4,5-Diphenylimidazoline for High-Purity Pharmaceutical Intermediates
Scalable Palladium-Catalyzed Synthesis of 4,5-Diphenylimidazoline for High-Purity Pharmaceutical Intermediates
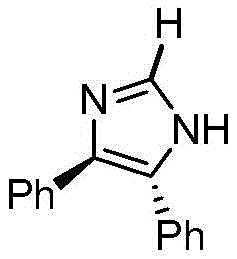
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity, stereochemical control, and cost-effective scalability. A pivotal advancement in this domain is documented in patent CN111995544B, which outlines a robust synthetic methodology for 4,5-diphenylimidazoline. This nitrogen-containing heterocyclic compound serves as a critical building block in the construction of complex bioactive molecules. The patent details a sophisticated approach utilizing a chiral palladium complex to facilitate the cyclization of benzil with ammonium formate. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented route is essential. The method not only ensures the production of high-purity material suitable for downstream API synthesis but also establishes a foundation for consistent quality control in large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted imidazoles and imidazolines has relied heavily on multicomponent reactions catalyzed by solid acids or zeolites, such as the H-BEA zeolite mentioned in prior literature. While these methods offer a degree of atom economy, they frequently encounter significant hurdles regarding product isolation and stereochemical purity. Traditional thermal cyclizations often require harsh conditions that can lead to the degradation of sensitive functional groups or the formation of unwanted regioisomers. Furthermore, the removal of solid catalyst residues from the final product stream can introduce additional purification steps, increasing both processing time and operational costs. For supply chain managers, these inefficiencies translate into longer lead times and potential variability in batch-to-batch consistency, which is unacceptable for GMP-compliant pharmaceutical manufacturing.
The Novel Approach
The methodology presented in CN111995544B represents a paradigm shift by employing a homogeneous chiral palladium catalyst system. This approach allows for the precise activation of the carbonyl groups in benzil under relatively mild reflux conditions in chlorobenzene. By utilizing ammonium formate as both a nitrogen source and a reducing agent, the reaction proceeds through a streamlined mechanism that minimizes side reactions. The result is a highly selective formation of the 4,5-diphenylimidazoline core with a reported isolated yield of 75%. This level of efficiency significantly reduces the burden on downstream purification processes. For partners looking for cost reduction in pharmaceutical intermediate manufacturing, this transition from heterogeneous to optimized homogeneous catalysis offers a clear pathway to improved throughput and reduced waste generation.

Mechanistic Insights into Pd-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the unique behavior of the chiral palladium complex, specifically the bis{[1,4-(4S)-diisopropyl-2-oxazolinylbenzene]palladium chloride} derivative. Under anhydrous and anaerobic conditions, this catalyst activates the dicarbonyl substrate, facilitating a nucleophilic attack by the ammonia species generated in situ from ammonium formate. The reaction mechanism involves the initial formation of an imine intermediate, which subsequently undergoes an intramolecular cyclization assisted by the metal center. This coordination stabilizes the transition state, ensuring that the reaction proceeds towards the desired 4,5-disubstituted product rather than alternative isomers. The presence of the bulky diisopropyl groups on the oxazoline ligands provides the necessary steric environment to enforce chirality, which is crucial for applications requiring enantiomerically pure intermediates.
Furthermore, the utility of the resulting 4,5-diphenylimidazoline extends beyond its role as a static building block; it functions as an effective organocatalyst in its own right. As demonstrated in the patent data, the compound exhibits notable catalytic activity in the Strecker reaction, specifically promoting the condensation of benzophenone imine with trimethylsilyl nitrile. In this application, the imidazoline derivative achieves a conversion rate of 66%, significantly outperforming uncatalyzed controls which only reach 30% conversion under identical conditions. This dual functionality highlights the compound's value in asymmetric synthesis, offering R&D teams a versatile tool for constructing chiral amines. The ability to produce a catalyst that can subsequently drive other high-value transformations adds a layer of strategic value to the supply chain.
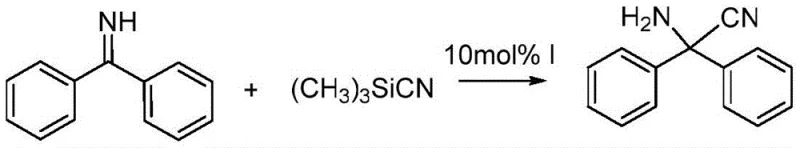
How to Synthesize 4,5-Diphenylimidazoline Efficiently
The practical implementation of this synthesis requires careful attention to the preparation of the catalytic species and the maintenance of inert atmospheres. The process begins with the synthesis of the chiral ligand from 1,4-dicyanobenzene and L-valinol, followed by complexation with palladium chloride. Once the catalyst is secured, the main cyclization reaction is straightforward, involving the reflux of benzil and ammonium formate in chlorobenzene for 30 hours. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to ensure reproducibility and safety in your pilot plant operations.
- Preparation of the chiral palladium catalyst complex under anhydrous and anaerobic conditions using zinc chloride and L-valinol derivatives.
- Refluxing benzil (0.5mmol) and ammonium formate (5.0g) in chlorobenzene solvent with 1mol% of the palladium catalyst for 30 hours.
- Purification of the crude reaction mixture via column chromatography using petroleum ether and dichloromethane to isolate single crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN111995544B offers tangible benefits that extend beyond mere chemical yield. The shift towards this palladium-catalyzed route addresses several critical pain points associated with traditional heterocycle synthesis. By optimizing the reaction conditions to utilize commercially available starting materials like benzil and ammonium formate, the process mitigates the risks associated with sourcing exotic or unstable reagents. This stability in raw material supply is paramount for maintaining continuous production schedules and avoiding costly delays. Moreover, the high selectivity of the reaction reduces the complexity of the impurity profile, simplifying the analytical burden and accelerating the release of batches for further processing.
- Cost Reduction in Manufacturing: The implementation of this catalytic cycle eliminates the need for excessive purification steps often required to remove solid catalyst residues or separate difficult isomers. By achieving a high yield of 75% directly from the reaction mixture, the overall consumption of solvents and silica for chromatography is drastically reduced. This efficiency translates into substantial cost savings in terms of raw material usage and waste disposal fees. Additionally, the recovery and potential recycling of the palladium catalyst, given its high value, can further enhance the economic viability of the process on a multi-ton scale.
- Enhanced Supply Chain Reliability: The reliance on robust, well-characterized starting materials ensures that the supply chain remains resilient against market fluctuations. Benzil and ammonium formate are commodity chemicals with stable global availability, reducing the risk of bottlenecks that often plague specialty reagent-dependent syntheses. The 30-hour reflux time, while significant, fits well within standard batch processing cycles, allowing for predictable production planning. This reliability is crucial for long-term contracts where consistent delivery of high-purity pharmaceutical intermediates is mandatory.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from gram-scale laboratory optimization to kilogram and ton-scale commercial production. The use of chlorobenzene, while requiring appropriate handling, is a standard industrial solvent with established recovery protocols. The reduction in byproduct formation aligns with green chemistry principles by minimizing the E-factor of the process. For facilities operating under strict environmental regulations, this cleaner profile simplifies compliance and reduces the load on effluent treatment plants, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,5-diphenylimidazoline. These insights are derived directly from the experimental data and claims within patent CN111995544B, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this intermediate into their existing workflows.
Q: What is the primary advantage of the Pd-catalyzed method in CN111995544B?
A: The method utilizes a specialized chiral palladium complex to achieve a 75% yield under reflux conditions, offering a cleaner alternative to traditional zeolite-catalyzed routes which often suffer from lower selectivity.
Q: Can this intermediate be used as a catalyst itself?
A: Yes, the synthesized 4,5-diphenylimidazoline demonstrates significant catalytic activity in Strecker-type reactions, achieving a 66% conversion rate in the condensation of benzophenone imine and trimethylsilyl nitrile.
Q: What are the critical reaction conditions for scale-up?
A: The process requires strict anhydrous and anaerobic conditions during the catalyst preparation phase, followed by a 30-hour reflux in chlorobenzene, which is readily adaptable to standard industrial reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5-Diphenylimidazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4,5-diphenylimidazoline meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence means we can replicate the patented palladium-catalyzed process with precision, delivering material that supports your regulatory filings and clinical trials.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the global fine chemical industry. Let us collaborate to bring your next generation of therapeutics to market faster and more efficiently.
