Advanced Synthesis of 4-(trans-3-pentene)-4'-alkylcyclohexyl Biphenyl for Commercial Display Applications
The landscape of liquid crystal material manufacturing is undergoing a significant transformation driven by the demand for higher performance display technologies and more efficient production methodologies. A pivotal advancement in this sector is detailed in patent CN111423891A, which discloses a highly efficient synthesis method for 4-(trans-3-pentene)-4'-alkylcyclohexyl biphenyl liquid crystal compounds. This specific class of compounds is critical for modulating the viscosity and response speed of liquid crystal mixtures, directly impacting the performance of modern information display panels. The patented approach addresses longstanding inefficiencies in organic synthesis by streamlining the production pathway into a concise two-step process. By leveraging a robust Suzuki coupling reaction followed by a precise alkaline alkylation, the method achieves exceptional product purity levels reaching up to 99.97% and substantially improved yields compared to historical benchmarks. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a viable pathway to securing a reliable supply of high-performance intermediates while mitigating the costs associated with complex multi-step syntheses.
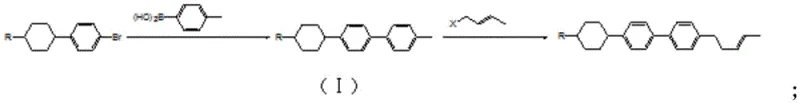
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(trans-3-pentene)-4'-alkylcyclohexyl biphenyl derivatives has been plagued by inefficient reaction pathways that hinder cost-effective mass production. Traditional methods, such as those illustrated in prior art, often rely on lengthy multi-step sequences involving bromination, self-coupling, or complex aldehyde condensations. For instance, earlier routes frequently necessitated a bromination step that inevitably produced undesirable ortho-isomers, complicating the purification process and drastically reducing the overall yield of the desired para-substituted product. Furthermore, alternative conventional strategies involved synthesizing cis-trans mixtures in the penultimate step, requiring additional energy-intensive isomerization reactions to enrich the trans-isomer content. These legacy processes typically resulted in a disappointing total yield ranging merely from 25% to 35%, creating significant bottlenecks in the supply chain for high-purity display materials. The accumulation of impurities at each stage not only increased waste generation but also demanded rigorous and expensive chromatographic purification to meet the stringent quality standards required for optoelectronic applications.
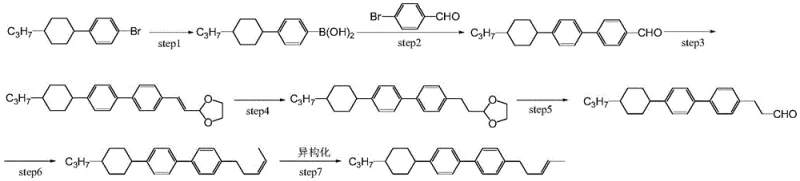
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel methodology outlined in the patent introduces a streamlined strategy that fundamentally reimagines the bond construction logic. The new approach initiates with a high-efficiency Suzuki coupling reaction between alkyl cyclohexyl bromobenzene and methyl phenylboronic acid, rapidly constructing the core biphenyl scaffold with high regioselectivity. This is immediately followed by a direct alkylation step using 1-halogenated-2-butene under carefully controlled alkaline conditions. By bypassing the need for aldehyde intermediates and avoiding the formation of difficult-to-remove self-coupling byproducts, this route dramatically simplifies the workflow. The elimination of the isomerization step is particularly noteworthy, as it removes a major source of yield loss and operational complexity. Consequently, this innovative pathway delivers a total refined yield of 65% to 70%, effectively doubling the efficiency of previous methods. This leap in productivity translates directly into reduced manufacturing costs and a more stable supply of critical liquid crystal monomers for the global display industry.
Mechanistic Insights into Suzuki Coupling and Alkaline Alkylation
The success of this synthesis hinges on the precise optimization of two distinct catalytic and stoichiometric transformations. The first step utilizes a palladium-catalyzed Suzuki-Miyaura cross-coupling, employing specialized catalysts such as PdCl2(AmPhos)2 or Pd(PPh3)4 to facilitate the union of the cyclohexyl-bromobenzene and the boronic acid. The choice of catalyst and the use of phase transfer agents like tetrabutylammonium bromide in a toluene solvent system at 80-85°C ensures rapid conversion while minimizing homocoupling side reactions. This step is critical for establishing the structural integrity of the biphenyl core, achieving intermediate purities exceeding 97% before further processing. The mechanistic elegance lies in the compatibility of the cyclohexyl ring under these conditions, preserving the stereochemistry required for the final liquid crystal properties.
The second transformation involves a nucleophilic substitution facilitated by organolithium chemistry. The intermediate methyl-biphenyl derivative is treated with a strong base, specifically a combination of sodium tert-butoxide and n-butyllithium in tetrahydrofuran (THF). Temperature control is paramount here; maintaining the reaction between -30°C and 10°C during the addition of n-butyllithium and the subsequent 1-halogenated-2-butene is essential to direct the reaction towards the desired trans-alkene configuration. This low-temperature regime suppresses competing elimination reactions and prevents the scrambling of the double bond geometry. The result is a highly selective formation of the trans-3-pentene side chain, which is crucial for the negative dielectric anisotropy and high clearing point characteristics of the final liquid crystal material. This level of mechanistic control allows for the production of material with GC purity levels approaching 99.97% after simple recrystallization.
How to Synthesize 4-(trans-3-pentene)-4'-alkylcyclohexyl Biphenyl Efficiently
Implementing this synthesis requires adherence to strict inert atmosphere conditions and precise stoichiometric ratios to maximize the benefits of the patented route. The process begins with the preparation of the biphenyl intermediate via Suzuki coupling, followed by the critical low-temperature alkylation. Operators must ensure rigorous exclusion of moisture and oxygen, particularly during the organolithium step, to prevent quenching of the reactive intermediates. The following guide outlines the standardized operational parameters derived from the patent examples, ensuring reproducibility and high yield.
- Perform Suzuki coupling between alkyl cyclohexyl bromobenzene and methyl phenylboronic acid using a Pd catalyst in toluene at 80-85°C.
- React the resulting intermediate with 1-halogenated-2-butene under alkaline conditions using n-butyllithium and sodium tert-butoxide in THF.
- Quench the reaction, extract with toluene, and recrystallize using a toluene/ethanol or heptane/ethanol mixture to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers profound strategic advantages beyond mere technical metrics. The reduction of the synthetic route from multiple complex stages to just two primary steps fundamentally alters the cost structure of manufacturing these specialized electronic chemicals. By eliminating the need for expensive aldehyde precursors and removing the isomerization unit operation, the process significantly lowers the consumption of raw materials and utilities. This simplification also reduces the burden on waste treatment facilities, as fewer byproducts and solvent exchanges are required, aligning with increasingly stringent environmental compliance standards in chemical manufacturing.
- Cost Reduction in Manufacturing: The dramatic improvement in overall yield from approximately 30% to nearly 70% implies that significantly less starting material is required to produce the same amount of final product. This efficiency gain directly correlates to a substantial reduction in the cost of goods sold (COGS). Furthermore, the use of readily available and inexpensive industrial solvents like toluene and THF, combined with the avoidance of exotic reagents, ensures that the variable costs remain low and predictable. The elimination of difficult purification steps, such as extensive column chromatography often needed to remove ortho-isomers in older methods, further drives down operational expenditures by saving time and silica gel resources.
- Enhanced Supply Chain Reliability: The reliance on simple, commodity-grade raw materials such as alkyl cyclohexyl bromobenzene and methyl phenylboronic acid mitigates the risk of supply disruptions often associated with custom-synthesized intermediates. Since these starting materials are widely produced for various organic synthesis applications, sourcing is flexible and competitive. Additionally, the robustness of the two-step process enhances production scheduling reliability; shorter cycle times mean that manufacturers can respond more agilely to fluctuations in market demand for liquid crystal displays. This agility is crucial in an industry characterized by rapid technology turnover and tight product launch windows.
- Scalability and Environmental Compliance: The operational simplicity of this method makes it inherently scalable from kilogram-level laboratory batches to multi-ton commercial production without significant re-engineering. The reaction conditions, while requiring temperature control, do not demand extreme pressures or hazardous high-energy inputs that often limit scale-up potential. From an environmental perspective, the higher atom economy and reduced solvent usage per kilogram of product contribute to a smaller carbon footprint. The ability to achieve high purity through simple recrystallization rather than energy-intensive distillation or preparative HPLC further supports sustainable manufacturing practices, which are increasingly becoming a prerequisite for suppliers to major electronics conglomerates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional alternatives in real-world scenarios.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method reduces the synthesis to just two steps, eliminating the need for complex isomerization and purification processes found in older methods, thereby significantly increasing total yield from roughly 30% to over 65%.
Q: How does the process ensure high trans-isomer selectivity?
A: By strictly controlling the reaction temperature between -30°C and 10°C during the alkylation step and utilizing specific alkaline conditions, the process favors the formation of the desired trans-configuration without requiring post-reaction isomerization.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available raw materials and standard solvents like toluene and THF, with simple operational steps that facilitate easy scale-up from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(trans-3-pentene)-4'-alkylcyclohexyl Biphenyl Supplier
As the global demand for high-performance liquid crystal materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in advanced organic synthesis is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge methodologies like the one described in CN111423891A to deliver superior products. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of large-scale panel manufacturers without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for next-generation displays.
We invite potential partners to engage with our technical procurement team to discuss how our optimized synthesis routes can drive value for your specific applications. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how our efficient processes can lower your total acquisition costs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to validate our capabilities and secure a stable, high-quality supply of critical liquid crystal intermediates for your future projects.
