Advanced Synthetic Route for 2-Benzoxazepine Compounds: Enabling Efficient CNS Drug Manufacturing
Advanced Synthetic Route for 2-Benzoxazepine Compounds: Enabling Efficient CNS Drug Manufacturing
The pharmaceutical landscape is continuously evolving, driven by the demand for more efficient and sustainable manufacturing processes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN112851624A, which discloses a novel synthetic method for 2-benzoxazepine compounds. These structures serve as critical pharmacophores in a wide array of central nervous system (CNS) agents, including well-known drugs like Doxepin and Olopatadine. The patented technology introduces a selenium-mediated cyclization strategy that operates under remarkably mild conditions, utilizing N-phenylselenophthalimide (NPSP) and zinc chloride as a catalyst. This approach represents a paradigm shift from traditional harsh acidic methods, offering a pathway to high-purity intermediates with exceptional yields. For R&D directors and procurement specialists alike, understanding this methodology is key to optimizing supply chains for next-generation CNS therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-benzoxazepine core has been a challenging endeavor for synthetic chemists, often plagued by severe operational constraints and environmental drawbacks. Traditional routes frequently rely on strong mineral acids like sulfuric acid or aggressive Lewis acids such as aluminum trichloride and boron trifluoride diethyl etherate. For instance, earlier methodologies described in literature required reaction temperatures as low as minus 30 degrees Celsius to control selectivity, necessitating energy-intensive cryogenic cooling systems that drastically increase operational expenditures. Furthermore, these harsh conditions often lead to significant side reactions, resulting in lower yields—sometimes hovering around 42 percent—and complex impurity profiles that are difficult to purge. The use of corrosive reagents also accelerates equipment degradation and generates substantial hazardous waste, posing serious challenges for environmental compliance and waste disposal in large-scale facilities.
The Novel Approach
In stark contrast, the method disclosed in CN112851624A leverages the unique reactivity of organoselenium reagents to achieve intramolecular cyclization at room temperature. By employing N-phenylselenophthalimide (NPSP) in conjunction with a zinc chloride catalyst, the process activates the olefinic bond of the cinnamyl ether substrate without the need for extreme thermal or acidic conditions. This mild environment not only preserves sensitive functional groups but also simplifies the reaction setup, allowing it to proceed in common solvents like dichloromethane. The result is a robust transformation that consistently delivers high yields, with specific examples in the patent demonstrating conversion rates exceeding 90 percent. This technological leap transforms the manufacturing landscape, turning a previously cumbersome multi-step sequence into a streamlined, industrially viable operation.
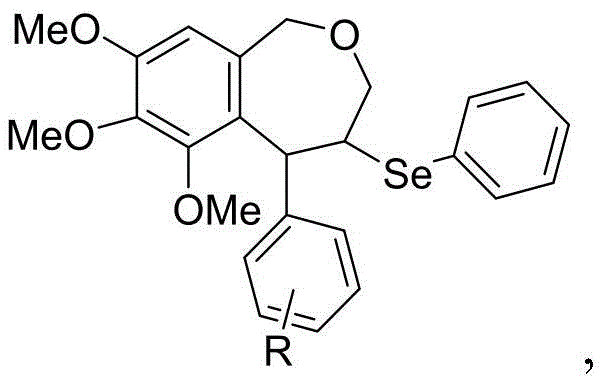
Mechanistic Insights into Selenium-Mediated Cyclization
The success of this synthetic route lies in the precise mechanistic interplay between the electrophilic selenium species and the electron-rich aromatic substrate. Upon interaction with the Lewis acid catalyst, the N-phenylselenophthalimide generates a highly reactive electrophilic selenium intermediate. This species selectively attacks the carbon-carbon double bond of the cinnamyl moiety within the substrate, forming a transient selenonium ion. This cyclic three-membered intermediate is pivotal, as it activates the adjacent carbon atoms for nucleophilic attack. Unlike traditional carbocation pathways that can suffer from rearrangements, the selenonium ion provides a rigid template that directs the subsequent cyclization with high fidelity.
Following the formation of the selenonium ion, the intramolecular nucleophilic attack occurs. The benzene ring of the 3,4,5-trimethoxybenzyl group, activated by the strong electron-donating effects of the methoxy substituents, acts as a potent nucleophile. It attacks the benzylic position of the selenonium intermediate, leading to the closure of the seven-membered oxazepine ring. This step is facilitated by the specific orientation of the substrate, ensuring that the carbon-oxygen and carbon-carbon bonds form in the correct geometry to yield the target 2-benzoxazepine structure. The phthalimide group is subsequently eliminated or transformed during the workup, leaving behind the desired heterocyclic core with minimal byproduct formation, thereby ensuring high purity suitable for pharmaceutical applications.
![Chemical structure of the key substrate 1-[(cinnamoyloxy)methyl]-3,4,5-trimethoxybenzene prior to cyclization](/insights/img/2-benzoxazepine-synthesis-pharma-supplier-20260306010615-011.png)
How to Synthesize 2-Benzoxazepine Efficiently
Implementing this advanced synthesis requires careful attention to reagent preparation and reaction stoichiometry to maximize the benefits of the selenium-mediated pathway. The process begins with the in-situ or separate preparation of the key organoselenium reagent, NPSP, followed by the assembly of the cinnamyl ether substrate. The convergence of these two components in the presence of zinc chloride triggers the cascade cyclization. While the general concept is straightforward, the devil is in the details regarding solvent dryness, temperature control during reagent addition, and the specific quenching protocol to ensure safe handling of selenium byproducts. For a comprehensive, step-by-step technical breakdown suitable for your process chemistry team, please refer to the standardized protocol below.
- Preparation of N-phenylselenophthalimide (NPSP) by reacting phenyl selenium bromide with potassium phthalimide in petroleum ether and dichloromethane.
- Synthesis of the key substrate 1-[(cinnamoyloxy)methyl]-3,4,5-trimethoxybenzene via etherification of cinnamyl alcohol and 1-bromomethyl-3,4,5-trimethoxybenzene.
- Cyclization reaction by mixing the substrate with NPSP and zinc chloride catalyst in anhydrous dichloromethane at room temperature, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of cryogenic requirements and harsh corrosive acids translates directly into reduced capital expenditure on specialized reactor linings and cooling infrastructure. Moreover, the simplified workup procedure, which utilizes standard aqueous bicarbonate quenching rather than complex neutralization of strong Lewis acids, streamlines the production cycle time. This efficiency gain allows for faster batch turnover and improved asset utilization, making the supply of these critical intermediates more resilient against market fluctuations and demand surges.
- Cost Reduction in Manufacturing: The shift from energy-intensive cryogenic conditions to ambient temperature processing results in substantial utility savings. Additionally, the high selectivity of the selenium-mediated reaction minimizes the formation of difficult-to-remove isomers, reducing the load on purification columns and lowering solvent consumption. By avoiding expensive and hazardous reagents like boron trifluoride, the overall raw material cost profile is optimized, delivering a more competitive cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, such as zinc chloride and substituted cinnamyl alcohols, are commodity chemicals with robust global supply chains. This reduces the risk of bottlenecks associated with specialty catalysts that may have single-source suppliers. The mild reaction conditions also mean that the process can be manufactured in a wider range of facilities, including multipurpose plants that may not be equipped for extreme low-temperature chemistry, thereby diversifying the potential manufacturing base and securing supply continuity.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this method mitigates those concerns. The absence of exothermic spikes typical of strong acid catalysis enhances process safety at the 100 MT scale. Furthermore, the reduced generation of acidic wastewater and the potential for recycling selenium species align with modern green chemistry principles. This ensures that production remains compliant with increasingly stringent environmental regulations, safeguarding the license to operate for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding the practical application, safety, and scalability of the selenium-mediated cyclization method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this selenium-mediated method over traditional acid-catalyzed routes?
A: Unlike traditional methods requiring harsh Lewis acids like aluminum trichloride or cryogenic conditions (-30°C) with boron trifluoride, this novel approach utilizes zinc chloride at room temperature. This significantly reduces energy consumption, minimizes equipment corrosion, and achieves higher yields (up to 92%) with simpler workup procedures.
Q: How does this process address impurity control in pharmaceutical intermediate manufacturing?
A: The reaction exhibits high regioselectivity due to the specific electrophilic nature of the organoselenium species and the activation of the trimethoxy-substituted benzene ring. This specificity effectively avoids the formation of isomeric byproducts common in Friedel-Crafts type cyclizations, ensuring a cleaner crude profile and reducing the burden on downstream purification.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The use of room temperature conditions eliminates the need for expensive cryogenic cooling infrastructure. Furthermore, the reagents are commercially accessible, and the quenching process using saturated sodium bicarbonate is straightforward and safe for large-volume reactors, facilitating seamless technology transfer from lab to plant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzoxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in CN112851624A for the production of high-value CNS drug intermediates. As a premier CDMO partner, we possess the technical expertise to adapt and optimize this selenium-mediated route for your specific project needs. Our facilities are equipped to handle complex organoselenium chemistry with the utmost safety and precision, ensuring that the transition from laboratory bench to commercial plant is seamless. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, backed by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your pipeline. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can improve your margins. Please contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable, cost-effective supply of high-purity 2-benzoxazepine intermediates for your global operations.
