Industrial Scale Synthesis of Cis-3-Amino-2-Arylpyrrolidine Derivatives via Raney Nickel Catalysis
Introduction to Advanced Pharmaceutical Intermediate Synthesis
The pharmaceutical industry constantly seeks robust and scalable pathways for complex heterocyclic scaffolds, particularly those serving as core structures for neurokinin receptor antagonists. Patent CN102936218B introduces a groundbreaking industrial preparation method for cis-3-amino-2-arylpyrrolidine derivatives, addressing critical bottlenecks in existing manufacturing technologies. This innovation primarily solves the technical problems associated with long synthetic routes, low overall yields, and the prohibitive costs of traditional methods that hinder large-scale production. By leveraging a streamlined sequence involving sodium hydride condensation, bromoacetonitrile alkylation, and a key Raney nickel hydrogenated cyclization, this technology offers a superior alternative for producing high-purity intermediates used in treating depression, anxiety, and chemotherapy-induced nausea. For R&D directors and procurement managers, understanding this shift from hazardous, low-yield processes to efficient catalytic cycles is essential for optimizing supply chains and reducing the cost of goods sold in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cis-3-amino-2-arylpyrrolidine derivatives has been plagued by severe operational challenges and environmental hazards. As illustrated in prior art such as Tetrahedron Letters 45 (2004), traditional routes often employ titanium tetrachloride for the critical ring-closing step. 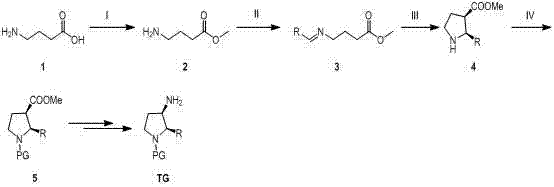 This reliance on TiCl4 presents a major liability; the reagent is severely corrosive, complicates post-treatment operations, and generates substantial environmental pollution, making it unsuitable for modern green chemistry standards. Furthermore, these legacy methods frequently fail to produce a single cis-product, resulting in difficult-to-separate mixtures that require extensive purification efforts, thereby driving down the overall yield and increasing production time. Other methods, such as those utilizing Grubbs second-generation catalysts and platinum dioxide, introduce exorbitant costs due to the use of precious metals, further limiting their feasibility for extensive commercial preparation.
This reliance on TiCl4 presents a major liability; the reagent is severely corrosive, complicates post-treatment operations, and generates substantial environmental pollution, making it unsuitable for modern green chemistry standards. Furthermore, these legacy methods frequently fail to produce a single cis-product, resulting in difficult-to-separate mixtures that require extensive purification efforts, thereby driving down the overall yield and increasing production time. Other methods, such as those utilizing Grubbs second-generation catalysts and platinum dioxide, introduce exorbitant costs due to the use of precious metals, further limiting their feasibility for extensive commercial preparation.
The Novel Approach
In stark contrast, the novel approach detailed in CN102936218B utilizes a concise and economically viable pathway that bypasses these historical hurdles. The new strategy initiates with the condensation of readily accessible aryl methyl ketones with dimethyl carbonate, followed by alkylation and a highly efficient Raney nickel hydrogenated cyclization. 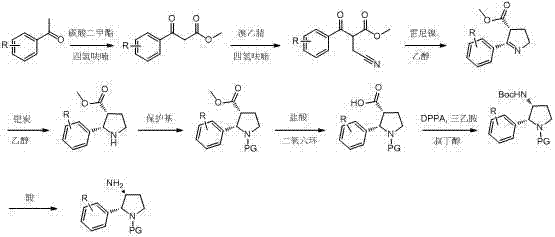 This methodology eliminates the need for dangerous and expensive reagents like titanium tetrachloride or Grubbs catalysts, replacing them with cost-effective and robust alternatives. The process is characterized by fewer reaction steps, mild reaction conditions, and a high gross production rate, specifically targeting the formation of the desired cis-isomer with high selectivity. By avoiding the generation of complex back-mixing compounds and utilizing standard industrial solvents like tetrahydrofuran and ethanol, this route significantly enhances the feasibility of scale-up production, offering a reliable solution for the commercial manufacturing of complex pharmaceutical intermediates.
This methodology eliminates the need for dangerous and expensive reagents like titanium tetrachloride or Grubbs catalysts, replacing them with cost-effective and robust alternatives. The process is characterized by fewer reaction steps, mild reaction conditions, and a high gross production rate, specifically targeting the formation of the desired cis-isomer with high selectivity. By avoiding the generation of complex back-mixing compounds and utilizing standard industrial solvents like tetrahydrofuran and ethanol, this route significantly enhances the feasibility of scale-up production, offering a reliable solution for the commercial manufacturing of complex pharmaceutical intermediates.
Mechanistic Insights into Raney Nickel Catalyzed Cyclization
The core of this technological breakthrough lies in the mechanistic efficiency of the Raney nickel catalyzed hydrogenation and cyclization step. Unlike homogeneous catalysis which often requires rigorous exclusion of air and moisture, the heterogeneous nature of Raney nickel allows for a more forgiving and operationally simple process. The mechanism involves the reduction of the nitrile group and the subsequent intramolecular cyclization to form the pyrrolidine ring system. This transformation occurs under moderate pressure (15-60 psi) and temperature (25-65°C), conditions that are easily manageable in standard stainless steel reactors. The catalyst facilitates the simultaneous reduction of the double bond in the enamine intermediate and the cyclization, ensuring that the thermodynamic preference for the cis-configuration is kinetically trapped effectively. This precise control over stereochemistry is vital for pharmaceutical applications where the biological activity is strictly dependent on the spatial arrangement of the amino and aryl groups on the pyrrolidine ring.
Furthermore, the impurity profile of this novel route is significantly cleaner compared to conventional methods. In traditional TiCl4 mediated reactions, the harsh acidic conditions often lead to side reactions such as polymerization or decomposition of sensitive functional groups, creating a complex impurity spectrum that is difficult to purge. In the Raney nickel system, the reaction environment is neutral to slightly basic depending on the specific workup, minimizing acid-catalyzed degradation. The subsequent steps involving protection with groups like Cbz or Acetyl, followed by acid hydrolysis and rearrangement using DPPA (Diphenylphosphoryl azide), are designed to be orthogonal and high-yielding. This strategic sequencing ensures that intermediates remain stable and purifiable at each stage, ultimately delivering a final product with stringent purity specifications required for clinical grade API intermediates without the need for excessive chromatographic purification.
How to Synthesize Cis-3-Amino-2-Arylpyrrolidine Efficiently
The synthesis of these valuable derivatives follows a logical progression designed for industrial robustness, beginning with the formation of the beta-keto ester backbone and concluding with the installation of the amine functionality. The process leverages common organic transformations that are well-understood in process chemistry, ensuring that technology transfer from lab to plant is seamless. Operators utilize standard equipment for reflux, hydrogenation, and filtration, avoiding the need for specialized cryogenic or high-pressure apparatus. The following guide outlines the critical phases of this synthesis, emphasizing the key parameters for maximizing yield and stereo-selectivity. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the technical documentation below.
- Condense aryl methyl ketone with dimethyl carbonate using sodium hydride, followed by bromoacetonitrile alkylation to form the cyano-ester precursor.
- Perform Raney nickel hydrogenated cyclization and enamine hydrogenated reduction to obtain the cis-2-aryl-pyrrolidine-3-carboxylate core structure.
- Introduce protecting groups, perform acid hydrolysis and rearrangement, and finally remove Boc groups to yield the target cis-3-amino derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The shift away from precious metal catalysts and corrosive reagents fundamentally alters the cost structure of the intermediate, removing volatile price dependencies associated with rare earth metals and specialized hazardous waste disposal. This process optimization ensures a more predictable pricing model for long-term contracts, allowing pharmaceutical companies to better forecast their raw material expenditures. Additionally, the use of commodity chemicals like aryl methyl ketones and dimethyl carbonate ensures a stable supply base, reducing the risk of shortages that often plague niche reagent markets. The simplified workup procedures also mean faster batch turnover times, enhancing the overall agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive Grubbs catalysts and platinum dioxide represents a massive reduction in direct material costs. By substituting these with Raney nickel and palladium on carbon, the process utilizes widely available, lower-cost catalysts that can often be recovered or disposed of more economically. Furthermore, the avoidance of titanium tetrachloride removes the significant downstream costs associated with neutralizing corrosive waste streams and maintaining specialized corrosion-resistant equipment. This holistic reduction in reagent and infrastructure costs leads to substantial savings in the overall cost of goods manufactured, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted aryl ethyl ketones ensures that the supply chain is not vulnerable to the bottlenecks often seen with proprietary or complex precursors. These bulk chemicals are produced by numerous suppliers globally, providing procurement teams with multiple sourcing options to mitigate supply risk. The robustness of the reaction conditions also means that production is less likely to be interrupted by minor fluctuations in utility availability or environmental controls, ensuring a consistent and reliable flow of high-purity intermediates to downstream formulation units.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior, facilitating easier regulatory approval for commercial scale-up. The mild reaction temperatures and pressures reduce energy consumption and operational hazards, while the absence of heavy metal contaminants like titanium simplifies the purification process and wastewater treatment. This alignment with green chemistry principles not only reduces the environmental footprint but also streamlines the audit process for international clients who demand strict adherence to sustainability standards. The process is inherently designed for tonnage production, capable of scaling from pilot batches to multi-ton annual capacities without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent data and practical process chemistry considerations, aimed at clarifying the operational benefits for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this intermediate into your existing drug development pipeline. We encourage technical teams to review these points to assess the alignment with their specific project requirements and quality standards.
Q: How does this new synthesis method improve upon traditional titanium tetrachloride routes?
A: Traditional methods often rely on titanium tetrachloride for ring closure, which is highly corrosive, difficult to treat in post-processing, and generates significant environmental pollution. The novel Raney nickel catalyzed route operates under mild conditions, avoids hazardous reagents, and simplifies purification, making it far more suitable for industrial scale-up.
Q: What are the cost advantages of using Raney Nickel over Grubbs catalysts for this intermediate?
A: Conventional routes utilizing Grubbs second-generation catalysts and platinum dioxide involve extremely expensive precious metals, driving up raw material costs significantly. By substituting these with Raney nickel and palladium on carbon, the process drastically reduces catalyst expenditure while maintaining high stereoselectivity for the cis-product.
Q: Is this synthetic route scalable for commercial API production?
A: Yes, the process is specifically designed for scalability. It utilizes readily available starting materials like aryl methyl ketones and operates at moderate temperatures and pressures. The elimination of difficult-to-separate mixtures and the use of robust heterogeneous catalysts ensure consistent quality and feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-3-Amino-2-Arylpyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our expertise in handling complex heterocyclic chemistry allows us to navigate the intricacies of the Raney nickel cyclization and subsequent protection steps with precision, guaranteeing a consistent supply of the cis-isomer required for your NK-1 antagonist programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation pharmaceuticals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your project's economics. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can accelerate your time to market while reducing overall development risks.
