Advanced Synthetic Route for Iloperidone: Enhancing Purity and Scalability for Global Pharmaceutical Markets
Advanced Synthetic Route for Iloperidone: Enhancing Purity and Scalability for Global Pharmaceutical Markets
The pharmaceutical industry continuously seeks robust manufacturing processes for critical antipsychotic agents, and the synthesis of Iloperidone represents a significant area of innovation. Patent CN102070626B introduces a transformative methodology that addresses long-standing challenges in the production of this atypical schizophrenia treatment. By shifting away from traditional cyclization-dependent pathways, this novel approach leverages a highly efficient two-step etherification strategy. The process begins with the activation of a hydroxy-propoxy acetophenone derivative, followed by a precise nucleophilic substitution with a fluorinated benzisoxazole piperidine scaffold. This technical breakthrough not only streamlines the synthetic sequence but also fundamentally alters the impurity profile, offering a pathway to high-purity active pharmaceutical ingredients (APIs) that meets stringent global regulatory standards. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for evaluating supply chain resilience and cost structures in the competitive psychotropic drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iloperidone has been plagued by inefficiencies inherent in early-generation methodologies, particularly those relying on late-stage ring closure. As documented in prior art such as US4355037 and CN101735208, the conventional strategy involves a cyclization reaction as the final step to construct the core structure. While this approach theoretically improves regioselectivity, it suffers from critically low reaction yields, often reported around 58%, which drastically inflates the cost of goods sold (COGS). Furthermore, these cyclization reactions typically require harsh strong base conditions that compromise the stability of the sensitive benzisoxazole moiety, leading to decomposition and complex impurity profiles that are difficult to control. Alternative polymeric synthesis routes, such as those described by Strupczewski, attempt to improve atom economy but introduce severe regioselectivity issues during the alkylation of the phenolic oxygen. These methods often necessitate a large excess of dihaloalkane reagents to drive selectivity, resulting in a mixture of chloro- and bromo-propyl by-products that require difficult and costly separation processes, thereby hindering commercial viability.
The Novel Approach
In stark contrast to these legacy methods, the methodology disclosed in CN102070626B adopts a convergent synthesis strategy that decouples the formation of the ether linkage from the core heterocyclic structure. This novel approach utilizes 6-fluoro-3-(4-piperidinyl)-1,2-benzisoxazole and 3-methoxy-4-(3-hydroxy-1-propoxy)acetophenone as primary building blocks, reacting them through a controlled activation and substitution sequence. By pre-activating the hydroxyl group of the acetophenone derivative into a superior leaving group, such as a tosylate or mesylate, the reaction ensures absolute site specificity during the coupling step. This eliminates the formation of regioisomers and significantly reduces the burden on downstream purification. The result is a process where every reaction step possesses a single dominant reaction site, ensuring that intermediates and the final product exhibit controllable and consistent quality. This shift from a linear, problem-prone cyclization to a modular etherification represents a paradigm shift in the manufacturing of complex psychotropic intermediates, offering a reliable foundation for industrial scale-up.
Mechanistic Insights into Sulfonylation and Nucleophilic Substitution
The core of this innovative synthesis lies in the precise chemical transformation of the hydroxyl functionality into a reactive electrophile, followed by a clean nucleophilic displacement. In the first stage, 3-methoxy-4-(3-hydroxy-1-propoxy)acetophenone undergoes sulfonylation using reagents like p-toluenesulfonyl chloride in the presence of an organic base such as triethylamine. This reaction is conducted at controlled low temperatures, typically ranging from -20°C to 10°C, to prevent side reactions and ensure the exclusive formation of the sulfonate ester. The resulting intermediate, Compound IV, serves as a highly activated substrate where the sulfonate group acts as an excellent leaving group, primed for displacement. This activation step is crucial because the native hydroxyl group is a poor leaving group, and direct substitution would be kinetically unfavorable. The use of mild organic bases and non-protic solvents like dichloromethane preserves the integrity of the acid-sensitive ketone and ether functionalities, preventing degradation that often occurs in more aggressive acidic or basic environments.
The second stage involves the coupling of this activated intermediate with the amine component, 6-fluoro-3-(4-piperidinyl)-1,2-benzisoxazole. This step proceeds via a classic SN2 nucleophilic substitution mechanism, facilitated by the presence of an inorganic base like potassium carbonate and a catalytic amount of potassium iodide. The iodide ion plays a pivotal role by participating in a Finkelstein-type halogen exchange, transiently converting the sulfonate or chloride leaving group into a more reactive iodide species in situ, which accelerates the reaction rate. The reaction is typically performed in a mixed solvent system of acetonitrile and N,N-dimethylformamide at elevated temperatures between 80°C and 90°C. This thermal energy overcomes the activation barrier for the substitution while the polar aprotic solvents stabilize the transition state. The outcome is the formation of the target ether linkage with high fidelity, yielding Iloperidone with a purity exceeding 99.5% after simple recrystallization, demonstrating the exceptional selectivity of this mechanistic pathway.
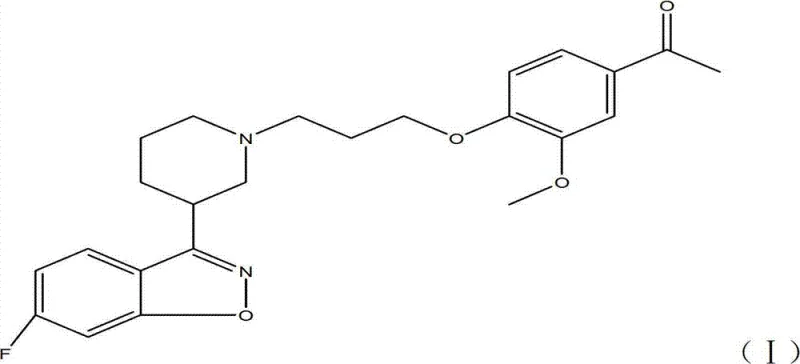
How to Synthesize Iloperidone Efficiently
The operational protocol for this synthesis is designed to maximize yield while minimizing the complexity of workup procedures, making it highly suitable for multi-kilogram production. The process begins with the dissolution of the hydroxy-acetophenone precursor in a chlorinated solvent, followed by the controlled addition of the sulfonylating agent under cooling to manage the exotherm. Once the activated intermediate is isolated and purified via recrystallization, it is suspended in a polar aprotic solvent mixture along with the piperidine derivative salt. The addition of base and iodide catalyst initiates the coupling, which is monitored until completion. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and quenching procedures, are outlined below to ensure reproducibility and safety during technology transfer.
- React 3-methoxy-4-(3-hydroxy-1-propoxy)acetophenone with a sulfonyl chloride (e.g., p-toluenesulfonyl chloride) in the presence of an organic base like triethylamine at low temperatures (-20°C to 10°C) to form the activated sulfonate intermediate.
- Suspend the activated sulfonate intermediate with 6-fluoro-3-(4-piperidinyl)-1,2-benzisoxazole hydrochloride in a mixed solvent system of acetonitrile and DMF.
- Add an inorganic base such as potassium carbonate and a catalytic amount of potassium iodide, then heat the mixture to 80-90°C to facilitate the nucleophilic substitution, followed by recrystallization to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train; because the reaction exhibits such high specificity, the crude product contains minimal by-products, allowing for purification via straightforward recrystallization rather than expensive chromatographic techniques. This elimination of column chromatography significantly reduces solvent consumption, silica gel waste, and processing time, leading to a leaner manufacturing footprint. Furthermore, the use of commercially available and cost-effective starting materials, such as p-toluenesulfonyl chloride and potassium carbonate, ensures that the raw material supply chain is robust and less susceptible to volatility compared to routes requiring exotic or custom-synthesized reagents. The ability to produce intermediates that are stable crystalline solids also enhances inventory management, allowing for the stocking of key precursors without significant degradation risks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high yield and the avoidance of costly separation technologies. By achieving yields approaching 95% in the activation step and nearly 95% in the coupling step, the overall material throughput is maximized, reducing the cost per kilogram of the final API. The removal of the need for large excesses of alkylating agents, which was a requirement in older polymeric routes, further lowers raw material costs. Additionally, the mild reaction conditions reduce energy consumption associated with extreme heating or cooling, and the simplified workup reduces labor hours and utility usage, collectively driving down the operational expenditure for API manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity chemicals that are widely available from multiple global vendors, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining uninterrupted supply to downstream formulation partners. Moreover, the stability of the intermediates allows for flexible production scheduling, where the activated sulfonate can be produced in advance and stored, decoupling the two main reaction steps and providing greater agility in responding to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns well with green chemistry principles by minimizing waste generation. The absence of heavy metal catalysts and the reduction in solvent volume due to higher concentrations and better yields contribute to a lower E-factor (environmental factor). The process is inherently scalable because it avoids hazardous reagents and exothermic runaways that are difficult to manage in large reactors. The ability to recycle solvents like acetonitrile and dichloromethane further enhances the sustainability profile, making it easier for manufacturers to meet increasingly strict environmental regulations and corporate sustainability goals without compromising on production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement teams assessing the quality assurances associated with suppliers utilizing this method.
Q: How does this novel synthesis method improve upon traditional cyclization routes?
A: Traditional methods often rely on a final cyclization step under strong basic conditions, which typically suffers from low yields (around 58%) and stability issues. This novel approach utilizes a convergent etherification strategy that avoids harsh cyclization conditions, resulting in significantly higher yields and improved product stability.
Q: What specific measures are taken to ensure high regioselectivity in this process?
A: The process employs a specific activation of the hydroxyl group via sulfonylation, creating a distinct leaving group. This ensures that the subsequent nucleophilic attack by the piperidine nitrogen occurs exclusively at the intended position, eliminating the regioselectivity problems and by-product formation seen in polymeric synthesis routes.
Q: Can the intermediates in this route be purified without chromatography?
A: Yes, a key advantage of this methodology is that all intermediates are crystalline solids that can be effectively purified through simple recrystallization techniques. This eliminates the need for expensive and time-consuming column chromatography, greatly facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iloperidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in patent CN102070626B can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Iloperidone meets the highest international pharmacopeial standards. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that de-risk your supply chain and accelerate your time to market.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this high-efficiency process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence and our track record of excellence in the production of complex psychiatric pharmaceutical intermediates.
