Transforming Pyridine Still Residue into High-Value Agrochemical Intermediates via Advanced Catalytic Hydrogenation
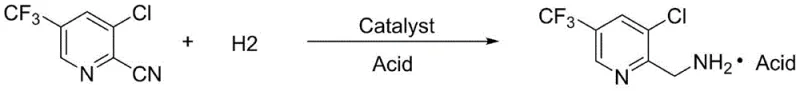
The global fine chemical industry faces a persistent challenge in managing high-value waste streams generated during the purification of complex heterocyclic compounds. Patent CN113735764B, published in early 2023, introduces a groundbreaking methodology specifically designed to address the inefficiencies associated with the rectification of 2-cyano-3-chloro-5-trifluoromethylpyridine. This patent outlines a sophisticated catalytic hydrogenation process that transforms what was previously considered hazardous still residue into a highly valuable agrochemical intermediate, specifically 2-methylamino-3-chloro-5-trifluoromethylpyridine acid salt. For R&D directors and process engineers, this represents a paradigm shift from linear consumption to circular chemical manufacturing, where waste streams are re-engineered into revenue-generating products through precise control of reaction parameters and catalyst selection.
The significance of this technology extends beyond mere waste management; it fundamentally alters the economic calculus of producing fluorinated pyridine derivatives. By recovering the active pharmaceutical or agrochemical ingredients trapped in the distillation bottoms, manufacturers can drastically reduce the raw material intensity of their production lines. This approach not only mitigates environmental liabilities associated with chemical waste disposal but also secures a secondary source of critical intermediates needed for the synthesis of next-generation pesticides like fluoroether bacteria amide. The technical robustness of this method, capable of achieving purities as high as 99.9 percent, demonstrates that recovered materials can meet the stringent quality standards required by top-tier multinational corporations without compromising on performance or safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of 2-cyano-3-chloro-5-trifluoromethylpyridine involves the nucleophilic substitution of fluorine atoms on a pyridine ring, followed by rigorous distillation to isolate the pure product. However, this conventional batch rectification process inevitably generates a substantial amount of still residue, often comprising 40 to 50 percent of the unreacted starting material alongside byproducts like succinonitrile. In standard operating procedures, these residues are frequently subjected to continuous rectification or blended with the next batch in an attempt to recover value. Unfortunately, this practice often leads to severe operational issues, including the polymerization of materials within the rectification kettle, which can form solid blocks that damage equipment and halt production lines. Furthermore, the high unit price of the fluorinated pyridine precursor means that discarding these residues represents a significant financial loss, while the accumulation of impurities like succinonitrile complicates downstream processing and reduces the overall yield of the final active ingredient.
The Novel Approach
The innovative strategy detailed in the patent circumvents these traditional bottlenecks by abandoning the attempt to distill the residue back to the nitrile precursor. Instead, it employs a direct catalytic conversion strategy that leverages the chemical potential of the residue. By introducing a specific catalyst and an organic solvent directly into the still residue, the process dissolves the complex mixture and prepares it for a selective hydrogenation reaction. This approach effectively bypasses the thermal instability issues associated with repeated distillation. The addition of acid prior to hydrogen introduction is a critical differentiator, as it facilitates the formation of the amine salt directly during the reduction phase. This one-pot transformation converts the problematic nitrile groups into stable aminomethyl groups, resulting in 2-methylamino-3-chloro-5-trifluoromethylpyridine acid salt. This novel route turns a disposal problem into a streamlined synthesis step, significantly simplifying the workflow and enhancing the overall atom economy of the manufacturing process.
Mechanistic Insights into Catalytic Hydrogenation of Nitriles
The core of this technological breakthrough lies in the chemoselective catalytic hydrogenation of the nitrile functional group in the presence of other sensitive moieties such as chloro and trifluoromethyl substituents. The reaction mechanism involves the adsorption of the 2-cyano-3-chloro-5-trifluoromethylpyridine molecule onto the surface of the metal catalyst, which can be palladium, molybdenum, nickel, or iron. Under mild pressures of 0.1 to 0.3 MPa and temperatures ranging from 10 to 30 degrees Celsius, hydrogen molecules dissociate on the catalyst surface and sequentially add to the carbon-nitrogen triple bond. The presence of the acid, typically hydrochloric or acetic acid, plays a dual role: it protonates the intermediate imine species to prevent the formation of secondary amines through condensation reactions, and it drives the equilibrium towards the formation of the stable ammonium salt. This acid-mediated pathway is crucial for maintaining high selectivity, ensuring that the reduction stops at the primary amine stage rather than proceeding to over-reduction or causing dehalogenation of the chlorine atom, which is a common side reaction in harsh hydrogenation conditions.
Furthermore, the tolerance of this catalytic system to impurities is a testament to its robustness. The still residue typically contains significant amounts of succinonitrile, a byproduct that could potentially poison sensitive catalysts or participate in unwanted side reactions. However, the optimized catalyst loading, with a weight ratio of catalyst to residue between 1:200 and 5:200, ensures sufficient active sites are available to process the target nitrile efficiently despite the presence of these contaminants. The subsequent crystallization step leverages the solubility differences between the desired pyridinium salt and the impurities. By distilling off a portion of the solvent and adding water, the system induces precipitation of the target product while leaving soluble impurities in the mother liquor. This physical separation mechanism, combined with the chemical selectivity of the hydrogenation, allows for the achievement of exceptional purity levels, consistently reaching 99.9 percent as verified by HPLC analysis, thereby validating the efficacy of the mechanistic design.
How to Synthesize 2-methylamino-3-chloro-5-trifluoromethylpyridine Efficiently
Implementing this recovery process requires precise adherence to the operational parameters defined in the patent to ensure safety and maximum yield. The procedure begins with the dissolution of the rectification kettle residue in a lower alcohol solvent, preferably methanol, which offers an optimal balance of solubility and ease of removal. The addition of the acid must be carefully controlled to maintain the correct stoichiometric ratio relative to the nitrile content, ensuring complete salt formation without excessive acidity that could corrode equipment. The hydrogenation step is conducted in a sealed reactor to maintain the necessary pressure, with reaction progress monitored by hydrogen uptake until saturation is reached. Following the reaction, the workup involves a strategic distillation to concentrate the solution followed by anti-solvent crystallization with water.
- Dissolve the 2-cyano-3-chloro-5-trifluoromethylpyridine rectification residue in an organic solvent like methanol with a catalyst such as nickel or molybdenum.
- Cool the solution, add acid (e.g., hydrochloric acid), and introduce hydrogen gas under sealed conditions until absorption ceases.
- Distill off solvent under vacuum, add water for crystallization, filter the crude acid salt, and recrystallize to obtain the qualified product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recovery technology offers profound strategic benefits that extend well beyond simple cost accounting. By integrating this process, manufacturing facilities can transform a cost center—waste disposal—into a profit center through the generation of saleable intermediates. This internal sourcing of raw materials reduces dependency on external suppliers for the 2-methylamino-3-chloro-5-trifluoromethylpyridine precursor, thereby insulating the supply chain from market volatility and potential shortages. The ability to recycle high-value fluorinated compounds internally creates a more resilient production ecosystem, ensuring continuity of supply even when upstream feedstock availability is constrained. Moreover, the simplified process flow, which eliminates the need for complex purification of the residue prior to conversion, reduces the overall operational complexity and energy consumption associated with waste treatment.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of waste disposal fees and the recovery of expensive fluorinated raw materials. Since the still residue contains nearly half of its weight in valuable nitrile compound, recovering this material effectively lowers the net cost of goods sold for the final amine product. The use of non-precious metal catalysts like nickel or iron further optimizes the cost structure by avoiding the high capital expenditure associated with palladium-based systems. Additionally, the ability to reuse the mother liquor for multiple batches significantly reduces solvent consumption, leading to substantial savings in raw material procurement and waste treatment costs without compromising product quality.
- Enhanced Supply Chain Reliability: Implementing an in-house recovery loop diversifies the sources of critical intermediates, reducing the risk associated with single-source suppliers. The process operates under mild conditions using readily available reagents such as methanol and hydrochloric acid, which are commodity chemicals with stable supply chains. This reduces the logistical burden of sourcing specialized reagents and minimizes the lead time required to ramp up production in response to increased demand. The robustness of the method against feedstock variability ensures that production schedules remain stable even when the composition of the input residue fluctuates slightly, providing a consistent output of high-quality material for downstream synthesis.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with green chemistry principles by maximizing atom economy and minimizing waste generation. The process avoids the use of hazardous reagents and operates at low temperatures, reducing the energy footprint of the manufacturing facility. The high yield and purity achieved mean that less material is lost to waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The scalability of the hydrogenation and crystallization steps is well-established in the fine chemical industry, allowing for seamless transition from pilot scale to multi-ton commercial production without the need for exotic equipment or specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this residue recovery technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and benefits of adopting this method for large-scale agrochemical intermediate production. Understanding these details is crucial for stakeholders evaluating the integration of this process into existing manufacturing workflows.
Q: What is the primary advantage of recovering pyridine still residue?
A: The primary advantage is economic valorization of waste. Instead of discarding expensive residue containing 40-50% active ingredient, this method converts it into a high-purity (99.9%) pesticide intermediate with yields up to 98%.
Q: Which catalysts are effective for this hydrogenation process?
A: The patent specifies that palladium, molybdenum, nickel, and iron are effective. Nickel and molybdenum are preferred due to their balance of activity and cost-effectiveness in industrial settings.
Q: How does this method handle impurities like succinonitrile?
A: The process is designed to tolerate impurities. The residue typically contains 15-20% succinonitrile, but the specific hydrogenation and crystallization steps effectively isolate the target pyridinium salt with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-methylamino-3-chloro-5-trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced recovery technologies like the one described in CN113735764B for optimizing the production of complex fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch of recovered intermediate meets the exacting standards required by the global agrochemical industry. We understand that consistency and reliability are paramount for our partners, and our state-of-the-art facilities are equipped to handle the specific challenges of catalytic hydrogenation and crystallization processes.
We invite forward-thinking organizations to collaborate with us to leverage these efficiency gains in their own supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and waste profiles. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for 2-methylamino-3-chloro-5-trifluoromethylpyridine and related derivatives. Together, we can drive sustainability and profitability through the intelligent application of cutting-edge chemical engineering solutions.
