Advanced Ag-Catalyzed Radical Cyclization for Scalable Cyclopentadienodihydroquinoline Production
Introduction to Next-Generation Heterocycle Synthesis
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for constructing bioactive cores found in potential therapeutic agents. Patent CN109796452B introduces a groundbreaking methodology for the preparation of cyclopentadienodihydroquinoline compounds, a structural motif known for its unique physical, chemical, and biological activities. This innovation addresses long-standing challenges in the field by utilizing a silver-catalyzed radical cascade cyclization strategy that operates under remarkably mild conditions. The core structural architecture of these valuable intermediates, characterized by a fused cyclopentane-dihydroquinoline system, is depicted below, highlighting the versatility of the substitution patterns achievable through this novel pathway.
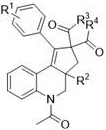
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such transformative chemistries is critical for ensuring a robust pipeline of high-quality building blocks. The patent details a process that not only simplifies the synthetic workflow but also significantly enhances the environmental profile of the manufacturing process. By leveraging a dual catalytic system involving potassium persulfate and silver nitrate, the method bypasses the need for pre-functionalized halogenated substrates, which are often costly and generate significant waste. This represents a paradigm shift from classical approaches, offering a streamlined route that is highly attractive for the commercial scale-up of complex heterocycles required in drug discovery and development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclopentadienodihydroquinoline derivatives has been plagued by significant operational hurdles that impede efficient production. Traditional methodologies often necessitate harsh reaction conditions, including extreme temperatures or the use of strong bases and acids, which can compromise the integrity of sensitive functional groups on the molecular scaffold. Furthermore, prior art frequently relies on the use of pre-activated substrates, such as 2-bromo-1,3-dicarbonyl compounds, to facilitate the cyclization event. This requirement for pre-functionalization introduces additional synthetic steps, thereby reducing the overall atom economy and increasing the cumulative cost of goods. The reliance on stoichiometric amounts of heavy metals or expensive transition metal catalysts in older protocols further exacerbates the economic and environmental burden, creating bottlenecks in cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN109796452B offers a streamlined, one-pot solution that elegantly circumvents these historical inefficiencies. The novel approach employs a radical cascade cyclization between readily available 1,7-enynes and 1,3-dicarbonyl compounds, driven by a catalytic amount of silver nitrate and potassium persulfate as the oxidant. This strategy eliminates the need for pre-halogenation, directly assembling the complex polycyclic framework with high regioselectivity. The general reaction scheme illustrates the convergence of these simple starting materials into the target scaffold under benign conditions.
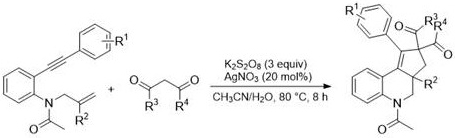
The operational simplicity of this new route is a major advantage for supply chain continuity. By utilizing a mixed solvent system of acetonitrile and water, the process aligns with green chemistry principles, reducing the reliance on purely organic solvents and simplifying downstream processing. The reaction proceeds efficiently at a moderate temperature of 80°C over an 8-hour period, yielding the desired products in substantial quantities. This robustness ensures that the production of high-purity cyclopentadienodihydroquinolines can be maintained consistently, minimizing the risk of batch failures that often plague more sensitive traditional syntheses.
Mechanistic Insights into Ag-Catalyzed Radical Cascade Cyclization
The success of this transformation hinges on the intricate interplay between the silver catalyst and the persulfate oxidant, which generates reactive radical species capable of initiating the cascade. Mechanistically, the silver(I) species likely facilitates the single-electron transfer (SET) process with the persulfate anion, generating sulfate radical anions. These potent oxidants abstract hydrogen or interact with the substrate to generate carbon-centered radicals on the 1,7-enyne framework. The resulting radical species undergoes an intramolecular addition to the alkyne moiety, followed by a subsequent cyclization onto the aromatic ring or the carbonyl component, depending on the specific substrate geometry. This cascade sequence is highly concerted, ensuring that the formation of the cyclopentane ring and the dihydroquinoline core occurs in a synchronized manner, which is crucial for maintaining high stereochemical and regiochemical fidelity.
From an impurity control perspective, the high selectivity of this radical pathway is paramount for R&D directors focused on purity profiles. The mild nature of the reaction conditions prevents the degradation of sensitive functional groups such as esters, halides, and ethers, which are often present in advanced intermediates. The use of water as a co-solvent may also play a role in stabilizing polar transition states or facilitating proton transfer steps, thereby suppressing side reactions like polymerization or over-oxidation. This inherent selectivity translates to a cleaner crude reaction mixture, reducing the burden on purification processes and ensuring that the final isolated material meets stringent purity specifications required for pharmaceutical applications without extensive recrystallization or chromatographic effort.
How to Synthesize Cyclopentadienodihydroquinoline Efficiently
The practical execution of this synthesis is designed for ease of adoption in both laboratory and pilot plant settings. The protocol involves dissolving the 1,7-enyne and 1,3-dicarbonyl precursors in a specific ratio of acetonitrile to water, followed by the sequential addition of the oxidant and catalyst. The reaction is monitored via thin-layer chromatography to ensure complete conversion before workup. Detailed standardized operating procedures regarding exact molar ratios, stirring rates, and quenching protocols are essential for reproducibility.
- Dissolve the 1,7-enyne compound and 1,3-dicarbonyl compound in a mixed solvent of acetonitrile and water (3: 1 ratio).
- Add potassium persulfate (3 equivalents) and silver nitrate (20 mol%) to the reaction mixture.
- Stir the reaction at 80°C for 8 hours, then extract, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The shift towards this catalytic system directly addresses the industry's demand for sustainable and cost-effective manufacturing processes. By eliminating the need for expensive, pre-functionalized starting materials and reducing the number of synthetic steps, the overall cost structure of the intermediate is significantly optimized. The use of commodity chemicals like potassium persulfate and silver nitrate, which are globally sourced and stable, mitigates the risk of supply disruptions associated with specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the synthetic route. By avoiding the separate halogenation step required in conventional methods, manufacturers save on reagent costs, labor hours, and equipment usage time. Furthermore, the catalytic nature of the silver species means that only small quantities are needed relative to the substrate, preventing the accumulation of heavy metal waste that requires costly disposal procedures. This lean approach to synthesis ensures that the cost of goods sold (COGS) is minimized, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available raw materials such as 1,7-enynes and simple 1,3-dicarbonyls ensures a resilient supply chain. Unlike specialized brominated intermediates that may have limited suppliers and long lead times, the precursors for this reaction are commodity chemicals produced at scale by multiple vendors worldwide. This diversification of the supply base reduces the risk of single-source dependency and ensures that production schedules can be met consistently. Additionally, the robustness of the reaction conditions means that manufacturing can proceed with high reliability, reducing the lead time for high-purity intermediates and enabling faster response to market demands.
- Scalability and Environmental Compliance: The incorporation of water into the solvent system is a critical factor for industrial scalability. Aqueous mixtures generally have higher heat capacities and are safer to handle on a large scale compared to volatile organic solvents alone. This feature facilitates easier temperature control during exothermic events, enhancing process safety. Moreover, the reduced organic solvent load simplifies wastewater treatment and solvent recovery processes, aligning with increasingly strict environmental regulations. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, a key metric for modern supply chain management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this Ag-catalyzed method over traditional synthesis?
A: Unlike traditional methods that require harsh conditions or pre-functionalized substrates like 2-bromo-1,3-dicarbonyls, this method utilizes readily available 1,7-enynes and 1,3-dicarbonyls under mild conditions (80°C) with high atom economy.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses inexpensive catalysts (AgNO3, K2S2O8) and a benign solvent system (MeCN/H2O), making it highly scalable and environmentally compliant for industrial production.
Q: What is the substrate scope for this reaction?
A: The reaction tolerates a wide range of substituents on the enyne moiety, including methyl, methoxy, fluorine, chlorine, and bromine, as well as various 1,3-dicarbonyl partners like barbituric acids and cyclic ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentadienodihydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Ag-catalyzed radical cyclization described in CN109796452B for the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex heterocyclic synthesis allows us to offer customized solutions that meet the specific needs of your drug development pipeline.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
