Revolutionizing Benzofuran Intermediate Production via Novel Phenol-Based Cyclization Strategies
Revolutionizing Benzofuran Intermediate Production via Novel Phenol-Based Cyclization Strategies
The pharmaceutical and agrochemical industries continuously seek robust, scalable, and cost-efficient pathways for constructing complex heterocyclic scaffolds, among which the benzofuran core remains a privileged structure due to its prevalence in bioactive molecules. A pivotal advancement in this domain is detailed in Chinese Patent CN103450125A, titled "Synthesis method for 5-substituted benzofuran-2-carboxylic acid and derivatives thereof," published in late 2013. This intellectual property introduces a transformative four-step synthetic strategy that circumvents the economic and technical bottlenecks associated with legacy methodologies. By shifting the synthetic entry point from expensive salicylaldehyde derivatives to readily available substituted phenols, this technology offers a compelling value proposition for global supply chains. For R&D directors and procurement specialists alike, understanding the mechanistic elegance and commercial viability of this route is essential for optimizing the sourcing of critical intermediates used in the production of drugs such as Ramelteon, Darifenacin, and Vilazodone.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzofuran-2-carboxylic acid scaffold has relied heavily on the condensation of 5-substituted salicylaldehydes with activated methylene compounds, such as ethyl bromoacetate or diethyl malonate, under basic conditions. While chemically sound, this classical approach suffers from severe economic and logistical drawbacks that hinder its application in modern, cost-sensitive manufacturing environments. The primary bottleneck is the starting material itself; 5-substituted salicylaldehydes are not commodity chemicals but rather specialized intermediates that command high market prices and often suffer from inconsistent supply availability. Furthermore, the reaction conditions required for these condensations can be quite harsh, often necessitating strong bases and elevated temperatures that complicate process safety and waste management. As illustrated in the conventional reaction schemes below, the reliance on these specific aldehyde precursors creates a fragile supply chain link that is vulnerable to price volatility and raw material shortages.
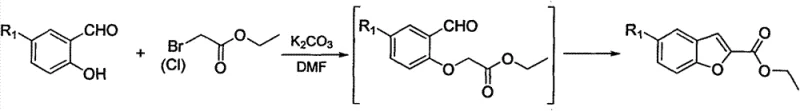
The Novel Approach
In stark contrast to the limitations of the past, the methodology disclosed in CN103450125A represents a paradigm shift by utilizing 4-substituted phenols as the foundational building blocks. Phenols are ubiquitous industrial chemicals produced on a massive scale, ensuring a stable, low-cost, and continuous supply stream that is immune to the fluctuations affecting specialty aldehydes. The novel route ingeniously converts these simple phenols into the target benzofuran system through a sequence involving halomethylation, phosphonium salt formation, and a unique cyclization with trichloroacetyl chloride. This strategic pivot not only slashes raw material costs but also simplifies the operational complexity of the synthesis. The process operates under relatively mild conditions, with reaction temperatures generally maintained between 0°C and 80°C, thereby reducing energy consumption and enhancing operational safety. This approach effectively decouples the production of high-value benzofuran intermediates from the scarcity of expensive precursors, offering a sustainable pathway for commercial scale-up.
Mechanistic Insights into Phenol-Based Benzofuran Cyclization
The chemical ingenuity of this patent lies in its multi-step transformation which effectively builds the furan ring onto the phenolic core through a series of well-controlled electrophilic and nucleophilic events. The process initiates with the halomethylation of the 4-substituted phenol, where formaldehyde or paraformaldehyde reacts with hydrogen halides to install a reactive halomethyl group ortho to the hydroxyl functionality. This intermediate is then quaternized with triphenylphosphine to generate a benzyltriphenyl phosphonium salt, a key species that activates the benzylic position for subsequent cyclization. The critical ring-closing step involves the reaction of this phosphonium salt with trichloroacetyl chloride in the presence of a base such as triethylamine or pyridine. This step likely proceeds via an intramolecular Wittig-type or nucleophilic attack mechanism where the phenoxide oxygen attacks the activated carbonyl or the alpha-carbon of the trichloroacetyl moiety, followed by elimination to form the trichloromethyl-benzofuran intermediate. Finally, acid-catalyzed alcoholysis converts the trichloromethyl group into the desired ester functionality, completing the synthesis with high atom economy regarding the carbon skeleton assembly.
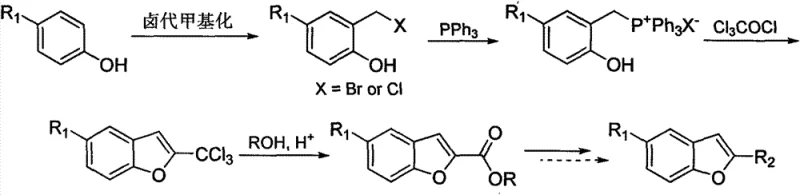
Beyond the primary bond-forming events, the patent details a robust mechanism for impurity control and functional group tolerance that is critical for pharmaceutical grade production. The use of trichloroacetyl chloride serves a dual purpose: it acts as a two-carbon synthon for the furan ring and provides a leaving group that is easily displaced during the final alcoholysis step. This specific choice of reagent minimizes the formation of polymeric byproducts often seen in direct aldol-type condensations. Furthermore, the reaction conditions are sufficiently mild to tolerate a wide array of substituents on the aromatic ring, including sensitive groups like nitro, cyano, and various halogens, without requiring extensive protecting group strategies. The ability to carry out the alcoholysis step under acidic reflux allows for the direct conversion of the trichloromethyl intermediate into various esters or even the free acid via hydrolysis, providing a versatile platform for generating diverse derivative libraries. This mechanistic flexibility ensures that the process can be adapted to produce specific analogues required for different drug candidates without fundamental changes to the core protocol.
How to Synthesize 5-Substituted Benzofuran-2-Carboxylic Acid Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and temperature control, particularly during the exothermic halomethylation and cyclization steps. The patent provides explicit guidance on solvent selection, recommending chlorinated solvents like chloroform or dichloroethane for the phosphonium salt formation to ensure solubility and reaction efficiency. For the final esterification, the choice of alcohol dictates the final ester product, allowing for the direct synthesis of methyl, ethyl, or higher alkyl esters in a single pot. The standardized procedure outlined in the patent examples demonstrates that crude intermediates can often be telescoped into the next step without purification, significantly reducing solvent usage and processing time. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures required to replicate this high-yielding process, please refer to the technical guide below.
- Perform chloromethylation or bromomethylation on 4-substituted phenol using formaldehyde and hydrohalic acid to generate 2-halomethyl-4-substituted phenol.
- React the halomethyl phenol with triphenylphosphine to form the corresponding 2-hydroxyl-5-substituted benzyltriphenyl phosphonium salt.
- Execute a ring-closing reaction with trichloroacetyl chloride under alkaline conditions to obtain 2-trichloromethyl-5-substituted benzofuran.
- Conduct acid-catalyzed alcoholysis on the trichloromethyl intermediate to yield the final 5-substituted benzofuran-2-carboxylic acid ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this phenol-based synthesis route offers tangible strategic benefits that extend far beyond simple unit price reductions. The most significant advantage is the stabilization of the supply chain through the utilization of commodity-grade starting materials. Unlike 5-substituted salicylaldehydes, which are produced by limited suppliers and subject to volatile pricing, 4-substituted phenols are manufactured globally in massive quantities for diverse industries, ensuring a reliable and continuous flow of raw materials. This shift effectively de-risks the procurement process, eliminating the potential for production stoppages caused by upstream shortages of niche intermediates. Additionally, the simplified process flow, which avoids harsh reaction conditions and complex purification steps, translates directly into lower manufacturing overheads and reduced waste disposal costs, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The economic impact of switching to this methodology is profound, primarily driven by the drastic difference in raw material costs between commodity phenols and specialty salicylaldehydes. By eliminating the need for expensive aldehyde precursors, the overall bill of materials is significantly lowered, allowing for more competitive pricing of the final active pharmaceutical ingredients. Furthermore, the process avoids the use of precious metal catalysts or exotic reagents, relying instead on inexpensive and widely available chemicals like triphenylphosphine and trichloroacetyl chloride. The ability to telescope steps and use crude intermediates without intermediate isolation further reduces solvent consumption, energy usage, and labor hours, resulting in substantial operational expenditure savings that enhance the overall profit margin of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by decoupling production from the constrained market of specialty aldehydes. Since the key starting materials are bulk chemicals with multiple global sources, the risk of supply disruption due to single-supplier dependency is virtually eliminated. This redundancy allows procurement teams to negotiate better terms and secure long-term contracts with greater confidence. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch quality and reliable delivery schedules. This stability is crucial for maintaining uninterrupted production lines for downstream API manufacturing, preventing costly delays and inventory stockouts.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this route offers distinct advantages for large-scale operations. The reaction conditions are mild, operating at atmospheric pressure and moderate temperatures, which simplifies the engineering requirements for reactor design and reduces the energy footprint of the process. The avoidance of heavy metal catalysts eliminates the need for complex and costly metal scavenging steps, simplifying waste treatment and ensuring compliance with strict residual metal limits in pharmaceutical products. The use of standard organic solvents and the potential for solvent recovery further aligns with green chemistry principles, reducing the volume of hazardous waste generated. These factors collectively facilitate easier regulatory approval and smoother technology transfer from laboratory to commercial-scale production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzofuran synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the practical aspects of adopting this route. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary cost advantages of this benzofuran synthesis method compared to traditional routes?
A: The primary advantage lies in the starting material. Traditional methods rely on 5-substituted salicylaldehydes, which are expensive and less available. This patent utilizes 4-substituted phenols, which are commodity chemicals with significantly lower market prices and higher availability, drastically reducing raw material costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the route's suitability for industrial amplification. The reaction conditions are mild (temperatures ranging from 0°C to 80°C), avoiding extreme pressures or temperatures. Furthermore, the intermediates can often be used directly in subsequent steps without rigorous purification, streamlining the workflow for ton-scale manufacturing.
Q: What types of substituents are compatible with this synthetic methodology?
A: The method demonstrates broad substrate scope. The R1 group on the phenol ring can be hydroxyl, alkoxyl, alkyl, haloalkyl, halogen, trifluoromethyl, nitro, cyano, aldehyde, acyl, carboxyl, or ester groups. This versatility allows for the synthesis of a wide array of derivatives needed for different pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Substituted Benzofuran-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the broader pharmaceutical value chain. Our technical team has thoroughly analyzed the methodology described in CN103450125A and possesses the expertise to execute this route with precision and reliability. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzofuran intermediate delivered meets the highest industry standards for downstream API synthesis.
We invite you to collaborate with us to leverage this cost-effective technology for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
