Advanced Photocatalytic Synthesis of Asymmetric Azobenzene Intermediates for Commercial Scale-Up
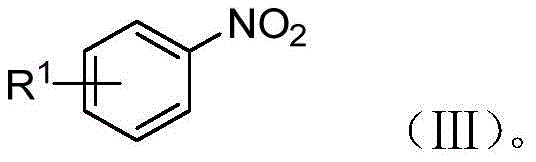
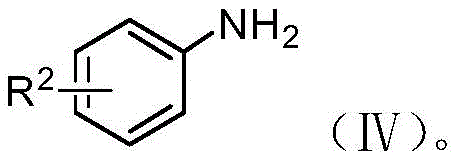
The pharmaceutical and fine chemical industries are constantly seeking safer, more sustainable pathways for synthesizing high-value intermediates, particularly those containing azo linkages which are critical for dyes, liquid crystals, and bioactive molecules. Patent CN110803998B introduces a groundbreaking photocatalytic method for the preparation of asymmetric azobenzene and azobenzene oxide compounds, representing a significant paradigm shift from hazardous traditional chemistry. This technology leverages visible light irradiation and semiconductor photocatalysts to couple aromatic nitro compounds with aromatic amino compounds under inert atmospheres, achieving high atom economy without the need for stoichiometric oxidants or reductants. By utilizing readily available starting materials such as substituted nitrobenzenes and anilines, this process mitigates the severe safety risks associated with unstable diazonium salts and toxic hydrazine derivatives found in legacy synthetic routes. The versatility of this approach allows for the precise tuning of reaction conditions to selectively produce either asymmetric azobenzene or azoxybenzene derivatives, making it an invaluable tool for reliable azobenzene intermediate supplier networks aiming to diversify their portfolio with green chemistry solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of asymmetric azobenzenes has relied heavily on diazotization reactions, a process fraught with inherent dangers and operational complexities that hinder efficient cost reduction in fine chemical manufacturing. The generation of diazonium salt intermediates is notoriously unstable, requiring strict temperature control often near freezing point to prevent violent decomposition and potential explosions, which poses a catastrophic risk in large-scale reactors. Alternative routes utilizing aromatic hydrazines and halogenated hydrocarbons, while avoiding diazonium salts, introduce their own set of severe drawbacks, including the thermal instability of hydrazine compounds which can decompose to release toxic nitrogen oxide flue gas into the environment. Furthermore, these conventional methods often suffer from poor atom utilization, generating substantial amounts of inorganic salt waste and requiring complex downstream purification steps to remove heavy metal catalysts or unreacted hazardous precursors. The inability to selectively control the oxidation state of the nitrogen-nitrogen bond often leads to mixtures of azo and azoxy products, complicating isolation and reducing overall yield, thereby inflating the cost of goods sold for high-purity asymmetric azobenzene products.
The Novel Approach
In stark contrast, the photocatalytic methodology described in the patent data offers a transformative solution by employing a redox-neutral coupling strategy driven by light energy rather than harsh chemical reagents. This novel approach utilizes a semiconductor photocatalyst with a specific bandgap range of 1-4 eV to facilitate the simultaneous reduction of the nitro group and oxidation of the amino group, effectively merging two half-reactions into a single, streamlined process. The reaction proceeds under mild conditions, typically between 0-100°C, and utilizes benign solvents such as water, DMSO, or acetonitrile, significantly lowering the energy footprint and safety barriers for commercial scale-up of complex photochemical intermediates. By simply adjusting the irradiation time, manufacturers can exert precise control over the product distribution, selectively stopping at the azoxy stage or driving the reaction to completion to form the azobenzene linkage with exceptional selectivity. This flexibility, combined with the use of earth-abundant or recyclable catalyst systems like copper/graphene composites, establishes a robust foundation for sustainable manufacturing that aligns with modern environmental regulations and corporate sustainability goals.
Mechanistic Insights into Photocatalytic Redox Coupling
The core of this innovation lies in the sophisticated interplay between the semiconductor photocatalyst and the substrate molecules under photon irradiation, creating a highly efficient electron transfer pathway. When the semiconductor material, such as a metal oxide or sulfide, absorbs photons with energy exceeding its bandgap, electron-hole pairs are generated; the photogenerated electrons reduce the aromatic nitro compound while the holes oxidize the aromatic amino compound. This dual activation mechanism ensures that no external oxidizing or reducing agents are consumed, as the substrates serve as mutual redox partners, leading to theoretically 100% atom economy regarding the nitrogen source. The presence of a base, such as potassium hydroxide or sodium carbonate, plays a critical role in facilitating the deprotonation steps and stabilizing intermediate radical species, thereby accelerating the coupling kinetics without introducing corrosive acidic waste streams. The inert atmosphere, maintained by gases like nitrogen or argon, prevents the quenching of reactive radical intermediates by atmospheric oxygen, ensuring that the reaction pathway remains directed exclusively toward the formation of the N=N or N(O)=N bonds.
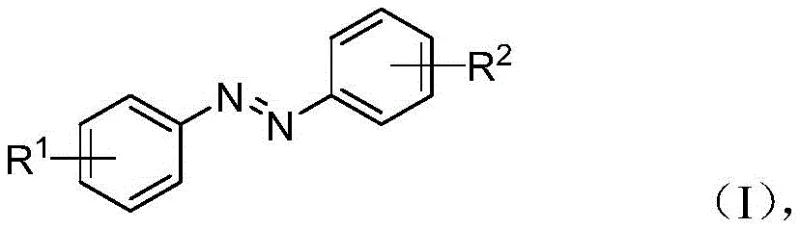
Impurity control in this system is inherently superior due to the mild reaction conditions and the specificity of the photocatalytic surface interactions. Unlike thermal reactions that often promote non-selective radical scrambling or over-oxidation, the photo-driven process limits side reactions primarily to the desired coupling, resulting in a cleaner crude reaction profile. The selectivity between the azoxy (Formula II) and azo (Formula I) products is kinetically controlled; shorter reaction times favor the accumulation of the partially reduced azoxy species, while prolonged irradiation drives the dehydration and further reduction required to form the stable azobenzene linkage. This mechanistic understanding allows process chemists to tailor the synthesis specifically for the target molecule, whether it be a precursor for liquid crystal displays requiring the azoxy motif or a pharmacophore needing the planar azobenzene structure. The ability to tune the catalyst composition, such as doping with noble metals like Pt or Au, further refines the charge separation efficiency, minimizing recombination losses and maximizing the turnover number of the catalytic system.
How to Synthesize Asymmetric Azobenzene Efficiently
Implementing this photocatalytic protocol requires careful attention to the mixing of reagents and the optimization of light intensity to ensure uniform irradiation throughout the reaction vessel. The standard procedure involves dispersing the aromatic nitro and amino substrates along with a base and the semiconductor photocatalyst in a suitable solvent system, followed by ultrasonic treatment to create a homogeneous suspension. Detailed standardized synthesis steps for optimizing yield and selectivity are provided in the guide below, outlining the precise molar ratios and reaction parameters validated in the patent examples.
- Mix aromatic nitro compounds, aromatic amino compounds, and a base in a molar ratio of 1: (0.1-10):(1-10) with a solvent and semiconductor photocatalyst.
- Stir the mixture under inert gas protection (e.g., N2, Ar) while irradiating with light (0.001-50W/cm2) at temperatures between 0-100°C.
- Control reaction time to selectively obtain either azoxybenzene (shorter time) or azobenzene (longer time), then dry and concentrate the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology translates directly into tangible operational improvements and risk mitigation strategies that enhance overall business resilience. By eliminating the need for hazardous diazonium salts and unstable hydrazines, facilities can drastically reduce the costs associated with specialized safety infrastructure, explosive-proof equipment, and toxic waste disposal protocols. The use of common, commodity-grade starting materials like nitrobenzenes and anilines ensures a stable and diversified supply base, reducing lead time for high-purity azo compounds by removing reliance on bespoke, low-volume reagents that are prone to market volatility. Furthermore, the ambient pressure and moderate temperature requirements lower the energy consumption per kilogram of product, contributing to a leaner cost structure that improves margin potential in competitive markets.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and reductants removes a significant line item from the raw material bill, while the recyclability of the heterogeneous photocatalyst further drives down long-term operational expenses. The simplified workup procedure, often requiring only filtration and concentration, reduces solvent usage and processing time, leading to substantial cost savings compared to multi-step traditional syntheses. Additionally, the high atom economy minimizes waste generation fees, providing a double benefit of lower input costs and reduced environmental compliance expenditures.
- Enhanced Supply Chain Reliability: Sourcing aromatic nitro and amino compounds is straightforward due to their widespread production for other industrial applications, ensuring consistent availability and shielding the supply chain from single-source bottlenecks. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or pressure compared to cryogenic diazotization, thereby improving on-time delivery performance. This reliability is crucial for maintaining continuous production lines for downstream customers in the pharmaceutical and agrochemical sectors who demand uninterrupted material flow.
- Scalability and Environmental Compliance: The modular nature of photoreactors allows for easy scale-out strategies, enabling production capacity to be increased by adding more light units rather than building massive, risky batch reactors. The absence of heavy metal catalysts and toxic byproducts simplifies regulatory approval processes and aligns with increasingly stringent global environmental standards, future-proofing the manufacturing asset. This green chemistry profile enhances the brand value of the final product, appealing to end-users who prioritize sustainability in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route, based on the detailed experimental data and claims within the patent documentation. These insights are designed to clarify the feasibility and advantages of transitioning from legacy methods to this advanced photochemical platform.
Q: How does this photocatalytic method improve safety compared to traditional diazotization?
A: Traditional methods involve unstable diazonium salts prone to explosion and hydrazine derivatives that release toxic nitrogen oxides. This photocatalytic route uses stable nitro and amino precursors under mild conditions, eliminating explosion risks and toxic gas emissions.
Q: Can the selectivity between azobenzene and azoxybenzene be controlled?
A: Yes, selectivity is time-dependent. Shorter reaction times (e.g., 6 hours) favor the formation of azoxybenzene derivatives, while extended irradiation (e.g., 12 hours) drives the reaction toward the fully reduced asymmetric azobenzene products with high conversion rates.
Q: What types of photocatalysts are suitable for this industrial process?
A: The process utilizes semiconductor materials with a bandgap of 1-4 eV, including metal oxides, sulfides, perovskites, or carbon-based polymers. Specific examples include copper/graphene composites or Pt/GaN, which ensure high atom economy and recyclability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Azobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in reshaping the landscape of fine chemical production, and we are uniquely positioned to bring this innovation to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of asymmetric azobenzene or azoxybenzene meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs, unlocking new levels of efficiency and safety in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements, and ask for specific COA data and route feasibility assessments to verify how this technology can optimize your manufacturing operations.
