Revolutionizing Indole Oxide Production: A Metal-Free Cascade Strategy for Commercial Scale
Revolutionizing Indole Oxide Production: A Metal-Free Cascade Strategy for Commercial Scale
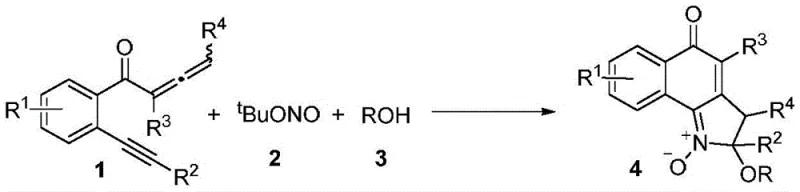
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant paradigm shift, driven by the urgent need for greener, more economical, and scalable manufacturing processes. Patent CN109896989B introduces a groundbreaking methodology for the synthesis of 5-oxo-2H-aromatic ring [g] indole-1-oxides, a class of compounds with profound biological relevance in the treatment of breast cancer and infectious diseases. This technology leverages a novel multi-component cascade reaction that directly constructs the intricate indole oxide core from readily available o-alkynyl-substituted aryl allenone compounds, tert-butyl nitrite, and alcohols. Unlike traditional routes that rely on harsh oxidants or toxic heavy metals, this approach operates under mild conditions in an air atmosphere, demonstrating exceptional atom economy and operational simplicity. For R&D directors and procurement specialists alike, this represents a critical advancement in accessing high-value pharmaceutical intermediates with reduced environmental impact and lower production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzindole-1-oxides and related alkaloids like stephacidin B has been fraught with significant technical and economic challenges that hinder efficient commercialization. Conventional strategies often depend on the oxidation of pre-formed benzindoles or zinc-catalyzed intramolecular reductive condensation reactions, which inherently suffer from low oxidation selectivity and the generation of substantial hazardous waste. The reliance on transition metal catalysts not only inflates raw material costs but also necessitates rigorous and expensive downstream purification processes to meet stringent regulatory limits on residual metals in pharmaceutical ingredients. Furthermore, many existing protocols require strictly anhydrous conditions, inert gas atmospheres, and elevated temperatures, creating substantial barriers to safe and reliable scale-up in a standard multipurpose chemical plant. These inefficiencies collectively result in prolonged lead times and unpredictable supply chains for critical drug intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN109896989B offers a streamlined, one-pot solution that elegantly bypasses these historical bottlenecks through a metal-free multi-component cascade mechanism. By utilizing tert-butyl nitrite as a dual-function reagent for both nitrosation and oxidation, the process eliminates the need for any external metal catalyst, thereby fundamentally altering the cost structure of the synthesis. The reaction proceeds smoothly in alcohol solvents, which serve a dual purpose as both the reaction medium and a nucleophile to introduce an alkoxy group, forming a valuable quaternary carbon center in a single step. This innovative route allows for the direct conversion of simple starting materials into complex 5-oxo-2H-aromatic ring [g] indole-1-oxides with high efficiency and minimal waste generation. The operational simplicity is further enhanced by the fact that the reaction tolerates air and moisture, removing the need for specialized equipment and making it ideally suited for robust industrial application.
Mechanistic Insights into Metal-Free Multi-Component Cascade Cyclization
The core of this technological breakthrough lies in the intricate yet efficient mechanistic pathway that orchestrates the formation of the indole oxide scaffold without metallic assistance. The reaction initiates with the interaction between the o-alkynyl-substituted aryl allenone and tert-butyl nitrite, likely generating a reactive nitroso intermediate that triggers an intramolecular cyclization. This is followed by a nucleophilic attack by the alcohol molecule, which traps the cationic species to form the stable quaternary carbon center observed in the final product structure. The entire sequence is a testament to the power of cascade chemistry, where multiple bond-forming events occur in a single operational step, maximizing atom economy and minimizing the accumulation of intermediate byproducts. For process chemists, understanding this mechanism is crucial as it highlights the tolerance of the system to various electronic environments on the aromatic rings, allowing for broad substrate scope without compromising yield or purity.
From an impurity control perspective, the absence of metal catalysts significantly simplifies the impurity profile of the crude reaction mixture. Traditional metal-catalyzed reactions often generate complex mixtures containing metal-ligand complexes, homocoupling byproducts, and over-oxidized species that are difficult to separate. In this metal-free protocol, the primary byproducts are volatile organic compounds or simple salts that can be easily removed during the workup phase. The high selectivity of the cascade reaction ensures that the desired 5-oxo-2H-aromatic ring [g] indole-1-oxide is formed as the major product, often precipitating directly from the reaction mixture upon cooling. This inherent purity reduces the burden on analytical laboratories for extensive impurity profiling and allows for a more straightforward validation of the manufacturing process, ensuring consistent quality batch after batch.
How to Synthesize 5-oxo-2H-aromatic ring [g] indole-1-oxide Efficiently
The practical implementation of this synthesis route is designed for maximum operational ease, enabling laboratory and pilot plant teams to produce high-quality intermediates with minimal training or specialized infrastructure. The standardized protocol involves simply dissolving the o-alkynyl substituted aryl allenone precursor and tert-butyl nitrite in a commercially available alcohol solution, such as 95% ethanol, which acts as both solvent and reactant. The mixture is then stirred at ambient temperature or slightly elevated temperatures up to 70°C under normal air atmosphere for a short duration, typically around 3 hours, allowing the cascade transformation to reach completion. Detailed standard operating procedures regarding stoichiometry, specific temperature ramps, and safety handling of tert-butyl nitrite are critical for reproducibility and are outlined in the technical guide below.
- Dissolve the o-alkynyl substituted aryl allenone compound and tert-butyl nitrite in an alcohol compound (acting as both reactant and solvent).
- Stir the reaction mixture at room temperature to 70°C under an air atmosphere for approximately 3 hours to allow the cascade cyclization to proceed.
- Cool the system to 0°C to precipitate the product, then filter, wash with ethanol, and dry to obtain the high-purity target indole oxide without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The elimination of transition metal catalysts removes a significant cost driver associated with both the purchase of expensive noble or base metals and the subsequent disposal or recovery of metal-contaminated waste streams. Furthermore, the simplified workup procedure, which relies on precipitation and filtration rather than column chromatography, drastically reduces solvent consumption and processing time, leading to substantial cost reduction in pharmaceutical intermediate manufacturing. This efficiency gain allows for faster turnover of production vessels and increased overall plant capacity without the need for capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the removal of costly catalytic systems and the simplification of purification workflows. By avoiding the use of metals like zinc or palladium, the raw material bill of materials is significantly lowered, and the expense associated with meeting strict residual metal specifications is entirely eradicated. Additionally, the ability to use technical grade alcohols as solvents and the avoidance of energy-intensive distillation or chromatographic separation steps contribute to a leaner, more cost-effective production model that enhances profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures a more reliable supply of critical indole oxide building blocks for downstream drug synthesis. Since the reaction does not require sensitive reagents or inert atmosphere conditions, it is less susceptible to disruptions caused by utility failures or operator error, ensuring consistent output. The wide substrate scope demonstrated in the patent means that a single production line can be adapted to manufacture various derivatives by simply changing the starting allenone, providing flexibility to respond to changing market demands for different API precursors without retooling.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by its exothermic profile and lack of hazardous reagents, aligning perfectly with modern green chemistry principles. The high atom economy and minimal waste generation reduce the environmental footprint of the manufacturing site, simplifying compliance with increasingly stringent environmental regulations. This sustainability advantage not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, making the final pharmaceutical products more attractive to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed experimental data provided in the patent literature. These insights are intended to clarify the operational parameters and potential applications for partners considering the integration of this route into their supply chain. Understanding these nuances is essential for making informed decisions about process adoption and long-term sourcing strategies.
Q: Does this synthesis method require transition metal catalysts?
A: No, the process described in patent CN109896989B is entirely metal-free, utilizing tert-butyl nitrite as the key reagent for nitrosation and cyclization, which eliminates the need for expensive metal removal steps.
Q: What is the purification method for the final indole oxide products?
A: The reaction design allows for simple precipitation upon cooling. The product can be isolated via suction filtration and washing, completely avoiding the need for time-consuming and solvent-intensive column chromatography.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions (room temperature to 70°C) in an air atmosphere without strict anhydrous requirements, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-oxo-2H-aromatic ring [g] indole-1-oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free cascade synthesis described in CN109896989B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 5-oxo-2H-aromatic ring [g] indole-1-oxide meets the exacting standards required for global pharmaceutical applications, providing you with a secure and high-quality source for your critical drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free protocol for your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the performance of this technology, ensuring a seamless transition to a more efficient and sustainable supply chain for your next-generation therapeutics.
