Advanced Manufacturing of 1-Beta-Methyl Carbapenem Bicyclic Mother Nucleus for Global Antibiotic Production
Advanced Manufacturing of 1-Beta-Methyl Carbapenem Bicyclic Mother Nucleus for Global Antibiotic Production
The pharmaceutical landscape for broad-spectrum antibiotics relies heavily on the efficient production of carbapenem intermediates, specifically the 1-beta-methyl carbapenem bicyclic mother nucleus. Patent CN101723971B, published in late 2011, introduces a transformative preparation method that addresses critical bottlenecks in stereocontrol, safety, and scalability inherent in previous generations of synthetic routes. This technology enables the production of key precursors for major commercial antibiotics such as Meropenem, Ertapenem, Biapenem, and Doripenem, all of which share this specific bicyclic parent nucleus structure. By optimizing the condensation and cyclization steps, the disclosed method achieves a remarkable overall yield of up to 46 percent while maintaining exceptional purity standards required for regulatory compliance in active pharmaceutical ingredient (API) manufacturing.
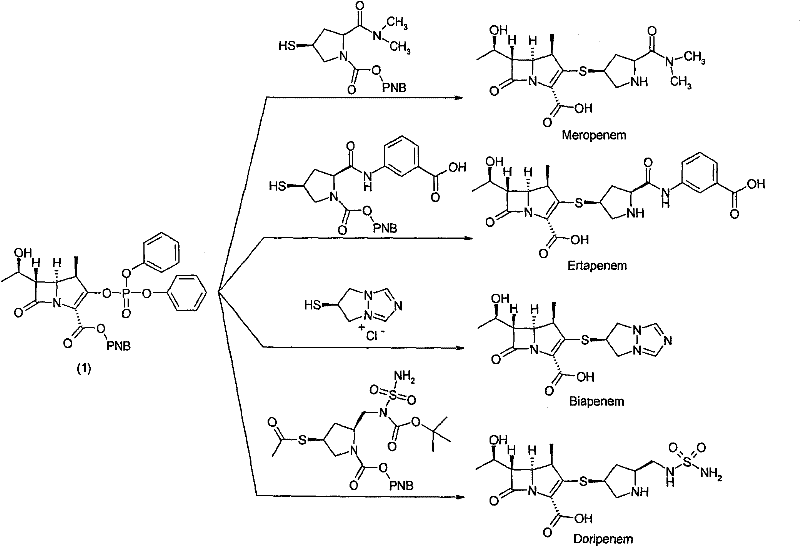
The strategic value of this patent lies in its ability to streamline the supply chain for these life-saving drugs. For R&D directors and process chemists, the detailed mechanistic improvements offer a robust pathway to minimize impurities that often plague carbapenem synthesis. For procurement and supply chain leaders, the shift towards simpler operations and safer reagents translates directly into reduced operational expenditures and enhanced continuity of supply. As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for evaluating the long-term viability and cost-effectiveness of sourcing strategies for complex beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the 1-beta-methyl carbapenem nucleus has been fraught with challenges related to stereoselectivity and hazardous reaction conditions. For instance, the synthetic route reported by Sumitomo Pharmaceuticals, as illustrated in the prior art, suffers from significant inefficiencies during the formation of intermediate compounds. Specifically, the process generates an equivalent amount of optically active isomers during the synthesis of compound (10), which drastically reduces the availability of the desired starting material and inflates production costs due to the loss of valuable chiral resources. Furthermore, other established routes, such as those utilizing thiazolidine derivatives reported by Japan Lederle Ltd., rely on raw materials that are notoriously difficult to produce and functionalize, leading to complex recovery processes and high preparation costs that restrict their industrial implementation.
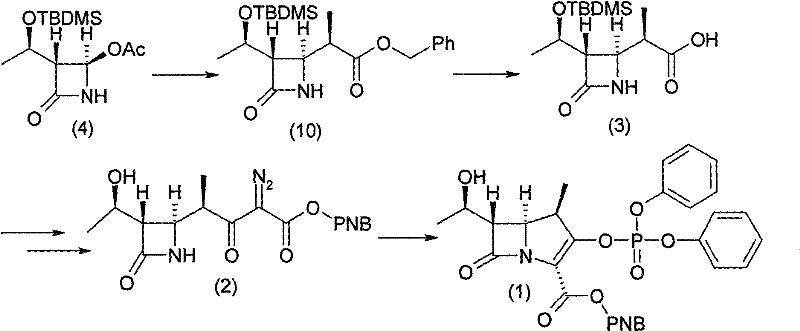
Additionally, alternative methods reported by entities like Meiji Seika Kaisha involve harsh reaction conditions that promote the formation of non-corresponding isomers, further diminishing the utilization ratio of key starting materials like compound (4). Perhaps most critically, academic routes such as those described by Kazuhiko Kondo utilize chiral auxiliaries and Reformatsky reactions that require extremely low temperatures, often around minus 60 degrees Celsius. These cryogenic conditions are not only energy-intensive but also pose significant safety risks due to violent exothermic reactions and the destructive nature of the reagents on the product, making product separation difficult and rendering these methods unsuitable for large-scale commercial manufacturing without substantial modification.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN101723971B employs a highly controlled condensation strategy that fundamentally alters the efficiency profile of the synthesis. The process initiates with the formation of a titanium enolate complex using titanium tetrachloride under the influence of a specific alkali mixed solution, which ensures excellent stereoselectivity with a beta-to-alpha ratio of 98:2. This high selectivity dramatically improves the utilization ratio of the starting material, compound (4), effectively doubling the efficiency compared to routes that produce racemic mixtures. Furthermore, the subsequent hydrolysis step utilizes acetone, a safe and water-soluble solvent, eliminating the explosive safety hazards associated with using tetrahydrofuran (THF) in the presence of peroxides, which was a standard but dangerous practice in traditional technologies.
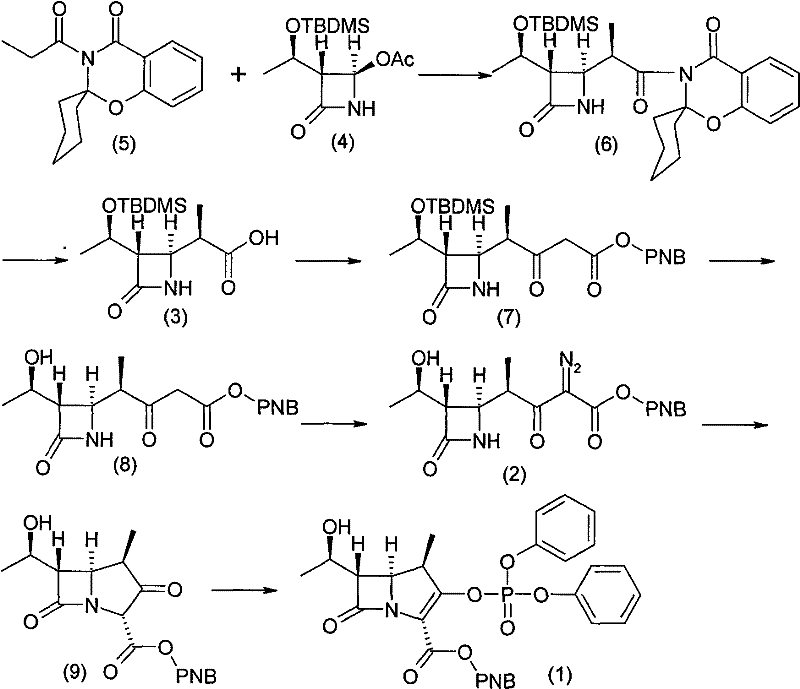
The innovation extends into the functionalization phase, where a 'one-pot' methodology is adopted to synthesize compound (2) from compound (3). This integrated approach combines activation, condensation, deprotection, and diazotization into a seamless sequence, thereby reducing the number of isolation steps and minimizing product damage caused by repeated purification. Instead of relying on difficult-to-control magnesium salts of malonic acid esters, the process uses simple malonic acid mono-p-nitrobenzyl ester, which is easier to handle and significantly lowers raw material costs. The final cyclization and active esterification steps are optimized to yield a white powder solid, compound (1), through membrane filtration and crystallization, ensuring a stable process that is easy to control and suitable for industrialized mass production with minimal three-waste generation.
Mechanistic Insights into Titanium-Mediated Stereoselective Condensation
The cornerstone of this advanced synthesis is the precise control over stereochemistry during the initial coupling of the beta-lactam ring with the side chain. The mechanism involves the generation of an enol titanate network from compound (5) using titanium tetrachloride in the presence of triethylamine and a catalytic amount of 4-dimethylaminopyridine (DMAP). This specific coordination environment creates a rigid transition state that favors the attack of the nucleophile from the beta-face, resulting in the observed 98:2 stereoselectivity. For R&D teams, understanding this ligand-accelerated catalysis is crucial, as the molar ratios of titanium tetrachloride, triethylamine, and the substrates are tightly controlled at 1:1.05:1.1 and 1:1.5 respectively to maintain this high fidelity. Deviation from these stoichiometric balances could lead to the formation of the undesired alpha-isomer, which would compromise the biological activity of the final antibiotic.
Furthermore, the purification mechanism represents a significant departure from standard laboratory practices, focusing on crystallization-driven purity rather than chromatographic separation. In the diazotization step, the use of sodium azide as the nitrene source, combined with methanesulfonyl chloride and a base like sodium bicarbonate, generates the diazo compound in situ. The byproducts of this reaction are designed to be water-soluble or easily extractable, allowing for their removal through simple washing and decolorization with activated carbon. This eliminates the need for silica gel column chromatography, which is a major bottleneck in scaling up fine chemical synthesis due to solvent consumption and batch variability. The direct crystallization of compound (2) from a concentrated solution upon the addition of poor solvents like petroleum ether ensures that the impurity profile remains within stringent specifications, providing a robust foundation for the subsequent rhodium-catalyzed cyclization.
How to Synthesize 1-Beta-Methyl Carbapenem Nucleus Efficiently
The execution of this synthesis requires strict adherence to the temperature profiles and reagent addition rates specified in the patent to ensure safety and yield. The process is divided into three distinct phases: the formation of the chiral intermediate (3), the one-pot conversion to the diazo compound (2), and the final cyclization to the bicyclic nucleus (1). Each phase builds upon the stereochemical integrity established in the previous step, making process control vital for success. The following guide outlines the standardized operational procedure derived from the patent examples, serving as a blueprint for pilot plant and commercial scale-up operations.
- Perform stereoselective condensation between compound (5) and compound (4) using a titanium tetrachloride-amine complex to form compound (6), followed by safe hydrolysis to obtain compound (3).
- Execute a 'one-pot' sequence on compound (3) involving activation, condensation with malonic acid monoester, deprotection, and diazotization using sodium azide to directly crystallize compound (2).
- Cyclize compound (2) into the bicyclic ketone ester using a rhodium catalyst, followed by active esterification with diphenylphosphoryl chloride to yield the final compound (1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound advantages in terms of cost structure and operational reliability. The shift from complex, multi-step purifications to a streamlined 'one-pot' process significantly reduces the consumption of organic solvents and the associated costs of solvent recovery and disposal. By eliminating the reliance on column chromatography, the manufacturing timeline is drastically shortened, allowing for faster batch turnover and improved responsiveness to market demand fluctuations. This efficiency gain is compounded by the use of readily available and inexpensive reagents, such as malonic acid monoester and sodium azide, which replace costly and difficult-to-source specialty chemicals used in legacy routes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substantial simplification of the workflow and the optimization of raw material usage. The high stereoselectivity ensures that nearly all input material is converted into the desired isomer, virtually eliminating the cost penalty associated with discarding unwanted optical isomers. Additionally, the replacement of hazardous solvents like THF with safer alternatives like acetone reduces the need for specialized explosion-proof equipment and lowers insurance and compliance costs. The overall yield improvement to 46 percent means that less starting material is required to produce the same amount of final product, directly lowering the cost of goods sold (COGS) for the API manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are less susceptible to market volatility compared to the specialized chiral auxiliaries and complex thiazolidine derivatives required by older methods. The robustness of the reaction conditions, which operate at moderate temperatures rather than extreme cryogenic levels, reduces the risk of batch failures due to equipment malfunction or temperature excursions. This stability ensures a consistent output of high-purity intermediates, minimizing the risk of production delays that can ripple through the entire pharmaceutical supply chain and affect the availability of finished antibiotic dosages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the reduction in three wastes (wastewater, waste gas, and solid waste) aligns with increasingly stringent global environmental regulations. The ability to perform direct crystallization and membrane filtration simplifies the engineering requirements for scale-up, making it easier to transition from kilogram-scale pilot runs to multi-ton commercial production without significant re-engineering. The lower energy consumption resulting from the avoidance of ultra-low temperature reactions further contributes to a smaller carbon footprint, enhancing the sustainability profile of the manufacturing site and ensuring long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology for industrial production.
Q: How does this patent improve stereoselectivity compared to conventional routes?
A: The process utilizes a specific titanium tetrachloride and amine mixed solution to generate an enol titanate complex, achieving a beta-to-alpha isomer ratio of 98:2, which significantly maximizes raw material utilization compared to older methods that produced equivalent optical isomers.
Q: What safety advantages does this method offer regarding solvents and reagents?
A: The method replaces hazardous tetrahydrofuran (THF) with safer acetone for hydrolysis in the presence of peroxides and substitutes expensive, difficult-to-control tosyl azide with sodium azide for diazotization, drastically reducing explosion risks and operational complexity.
Q: Does this process require column chromatography for purification?
A: No, a key innovation of this patent is the elimination of silica gel column chromatography; the intermediate compound (2) is obtained via direct crystallization after extraction and washing, which simplifies scale-up and reduces solvent waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Beta-Methyl Carbapenem Nucleus Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN101723971B can be translated into reliable supply volumes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-beta-methyl carbapenem nucleus meets the exacting standards required for downstream API synthesis, providing our partners with peace of mind regarding quality and consistency.
We invite pharmaceutical manufacturers and procurement specialists to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the efficiency and profitability of your antibiotic manufacturing operations.
