Industrial Scale Oxidation of 11-Hydroxyl Steroids for Cortisone Acetate Precursors
Introduction to Advanced Steroid Oxidation Technology
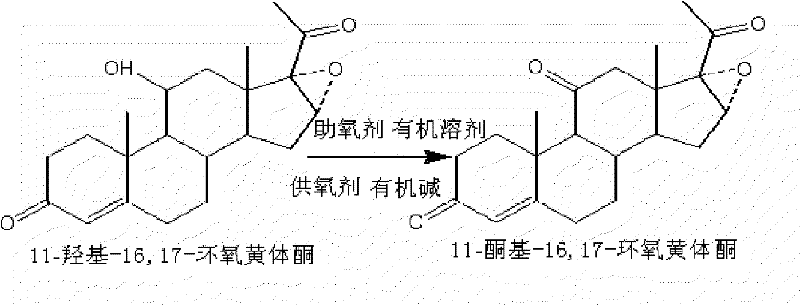
The pharmaceutical industry continuously seeks more efficient and environmentally sustainable pathways for synthesizing critical hormonal intermediates. A pivotal advancement in this domain is detailed in patent CN101544682B, which outlines a superior industrial production method for 11-keto-16,17-epoxyprogesterone. This compound serves as an essential precursor in the synthesis of cortisone acetate, a vital anti-inflammatory medication. The disclosed technology replaces hazardous traditional oxidation agents with a sophisticated system utilizing dimethyl sulfoxide (DMSO) and organic bases. By shifting away from heavy metal oxidants, this process not only enhances the chemical integrity of the final product but also aligns with modern green chemistry principles. For global procurement teams and R&D directors, understanding this transition is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the regulatory burdens associated with toxic waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 11-keto-16,17-epoxyprogesterone relied heavily on oxidation using chromium trioxide. While chemically effective, this legacy approach presents severe drawbacks for modern manufacturing environments. The primary concern is the generation of substantial volumes of chromate wastewater, which contains hexavalent chromium, a known carcinogen and persistent environmental pollutant. Managing this hazardous waste requires expensive treatment facilities and rigorous compliance monitoring, significantly inflating the operational expenditure. Furthermore, the chemical efficiency of the chromium method is suboptimal, typically achieving a yield of only 96 percent and a product content of merely 97 percent. These lower metrics necessitate additional purification steps, increasing solvent consumption and processing time, which ultimately compromises the cost reduction in API manufacturing efforts for downstream users.
The Novel Approach
The innovative method introduced in the patent data offers a transformative solution by employing a non-metallic oxidation system. This process utilizes 11-hydroxyl-16,17-epoxyprogesterone as the starting material, reacting it with an oxygen supply agent, an organic base, and an organic solvent. Specifically, the use of phenyl dichlorophosphate as the activating agent in conjunction with DMSO allows for a highly selective oxidation of the 11-hydroxyl group to the corresponding ketone. This approach eliminates the formation of heavy metal byproducts entirely, resulting in a cleaner reaction profile. The operational parameters are mild, with reactions conducted between -20 and 20 degrees Celsius, ensuring safety and ease of control. Consequently, this method achieves yields and purities greater than or equal to 99 percent, establishing a new benchmark for quality in the production of complex steroid intermediates.
Mechanistic Insights into DMSO-Mediated Oxidation
The core of this technological breakthrough lies in the activation of dimethyl sulfoxide (DMSO) to facilitate the oxidation of the secondary alcohol at the C11 position. In this mechanism, the organic base, such as triethylamine or pyridine, plays a critical role in deprotonating intermediates and driving the reaction forward. The phenyl dichlorophosphate acts as an electrophilic activator, reacting with DMSO to form a highly reactive sulfonium species. This activated sulfur complex then attacks the hydroxyl group of the steroid substrate, forming an alkoxysulfonium salt. Subsequent elimination, mediated by the base, releases the desired ketone functionality while generating dimethyl sulfide as a byproduct. This pathway avoids the radical mechanisms often associated with metal oxidants, thereby preserving the sensitive epoxide ring at the 16,17-position and preventing unwanted side reactions that could degrade the steroid backbone.
From an impurity control perspective, this mechanism offers distinct advantages over chromium-based oxidation. Metal-catalyzed reactions often leave trace residues of chromium in the final product, which are difficult to remove and strictly regulated in pharmaceutical applications due to toxicity concerns. The DMSO-based organic oxidation generates byproducts that are either volatile or easily removed during the aqueous workup and solvent evaporation steps. The absence of transition metals simplifies the purification process, allowing manufacturers to achieve the reported purity of greater than or equal to 99 percent with fewer crystallization cycles. This high level of chemical fidelity is essential for R&D directors who require consistent batch-to-batch reproducibility for clinical and commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 11-Keto-16,17-Epoxyprogesterone Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the efficiency of the DMSO activation. The process begins by dissolving the starting steroid in a suitable organic solvent such as dichloromethane, chloroform, or ethyl acetate. The ratio of reactants is critical, with the patent specifying a mass ratio of the steroid to DMSO to organic base to solvent of approximately 1:(0.5 to 5):(1 to 5):(8 to 15). Maintaining the reaction temperature between -20 and 20 degrees Celsius during the addition of the oxygen supply agent is vital to prevent thermal degradation of the activated intermediates. Following the reaction period of 1 to 20 hours, the mixture is processed to isolate the high-purity ketone product. The detailed standardized synthesis steps are provided in the guide below.
- Mix 11-hydroxyl-16,17-epoxyprogesterone with DMSO, an organic base like triethylamine, and a solvent such as dichloromethane, then cool to between -20 and 20 degrees Celsius.
- Add phenyl dichlorophosphate as the oxygen supply agent to the mixture and maintain the temperature between -20 and 20 degrees Celsius for 1 to 20 hours.
- Upon completion, evaporate the organic solvent under reduced pressure and wash the residue to isolate the final product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chromium-free oxidation technology translates into tangible strategic benefits beyond mere chemical yield. The elimination of hexavalent chromium from the process flow removes a significant regulatory liability and reduces the complexity of waste management logistics. Facilities no longer need to invest in specialized heavy metal scrubbing systems or pay premium fees for hazardous waste disposal, leading to substantial cost savings in operational overhead. Furthermore, the use of commodity chemicals like DMSO and triethylamine ensures a stable and resilient supply chain, as these reagents are widely available globally and are not subject to the same geopolitical restrictions as certain specialty metal catalysts. This reliability is crucial for maintaining continuous production schedules for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The shift to this organic oxidation system drastically simplifies the downstream processing requirements. Because the reaction does not generate heavy metal sludge, the extensive washing and chelation steps required to meet residual metal specifications are rendered unnecessary. This reduction in unit operations decreases solvent usage, energy consumption for heating and cooling, and labor hours per batch. Additionally, the higher yield of greater than or equal to 99 percent means that less raw material is wasted, directly improving the material cost efficiency. These factors combine to lower the overall cost of goods sold, making the final cortisone acetate intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on a process that utilizes common organic solvents and bases mitigates the risk of supply disruptions. Unlike specialized oxidants that may have limited suppliers or long lead times, reagents like dichloromethane and pyridine are standard inventory items for most fine chemical manufacturers. This availability ensures that production can be scaled up rapidly to meet surges in demand without waiting for critical raw material deliveries. Moreover, the robustness of the reaction conditions, which tolerate a temperature range from -20 to 20 degrees Celsius, reduces the risk of batch failures due to minor equipment fluctuations, thereby guaranteeing a consistent flow of materials to downstream customers.
- Scalability and Environmental Compliance: The patent explicitly confirms that this method has been successfully applied in industrial production, demonstrating its readiness for commercial scale-up. The absence of toxic chromium waste aligns perfectly with increasingly stringent environmental regulations worldwide, such as REACH in Europe and various EPA guidelines in the US. By adopting this green chemistry approach, manufacturers future-proof their operations against tightening emission standards. The simplified waste stream, consisting primarily of organic solvents and salts, is easier to treat and recycle, supporting sustainability goals and enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 11-keto-16,17-epoxyprogesterone. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent literature. They provide clarity on why this specific oxidation method is preferred for modern pharmaceutical manufacturing and how it impacts quality and compliance.
Q: What are the environmental advantages of this new oxidation method compared to traditional chromium trioxide processes?
A: The traditional method utilizes chromium trioxide, which generates significant amounts of toxic chromate wastewater requiring complex and costly disposal procedures. In contrast, the novel method described in patent CN101544682B employs dimethyl sulfoxide (DMSO) and phenyl dichlorophosphate, resulting in a process that produces no heavy metal waste and eliminates the severe environmental pollution associated with hexavalent chromium.
Q: What yield and purity levels can be achieved using this industrial production technique?
A: According to the patent data, this optimized oxidation route consistently achieves a reaction yield and product purity of greater than or equal to 99 percent. This represents a significant improvement over the conventional chromium-based method, which typically yields only 96 percent with a purity of roughly 97 percent, thereby reducing the need for extensive downstream purification.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states that the method has been applied in industrial production. The use of common organic solvents like dichloromethane or ethyl acetate, combined with mild reaction temperatures ranging from -20 to 20 degrees Celsius, ensures that the process is robust, safe, and easily scalable for the commercial manufacture of complex steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-Keto-16,17-Epoxyprogesterone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of life-saving hormones. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 11-keto-16,17-epoxyprogesterone meets the highest industry standards. Our commitment to green chemistry aligns with the advanced oxidation methods described, allowing us to offer products that are not only pure but also produced with minimal environmental impact.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can understand the economic benefits of switching to our optimized supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your development timelines are met with the highest quality materials available in the market.
