Revolutionizing Aminoacetylation: A Green Catalytic Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Aminoacetylation: A Green Catalytic Route for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more efficient catalytic systems, a trend vividly exemplified by the innovations disclosed in patent CN102432487A. This pivotal intellectual property introduces a novel method for catalyzing aminoacetylation reactions utilizing a Lewis basic ionic liquid, specifically [HDBU]OAc, derived from 1,8-diazabicyclo(5.4.0)undec-7-ene (DBU). For R&D directors and process chemists seeking robust pathways for API intermediate production, this technology represents a significant departure from conventional stoichiometric base usage. The core breakthrough lies in the transformation of DBU, a potent organic superbase known for its unpleasant odor and handling difficulties, into a stable, room-temperature ionic liquid that serves as a highly efficient, recyclable catalyst. By leveraging this advanced catalytic species, manufacturers can achieve acetylation of both aliphatic and aromatic amines under remarkably mild conditions, typically ranging from 40°C to 80°C, without the need for volatile organic solvents. This solvent-free approach not only aligns with stringent environmental regulations but also drastically reduces the E-factor of the process, making it an attractive candidate for large-scale commercial adoption in the competitive pharmaceutical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acetylation of amines has relied heavily on traditional catalysts such as pyridine, 4-dimethylaminopyridine (DMAP), or various strong acids and Lewis acids like transition metal halides. While these reagents are effective in laboratory settings, they present substantial drawbacks when scaled for industrial manufacturing. Pyridine and its derivatives are notorious for their toxicity, pungent odors, and the difficulty associated with their complete removal from the final product, often necessitating energy-intensive distillation or complex chromatographic purification steps. Furthermore, the use of transition metal catalysts introduces the risk of heavy metal contamination, a critical failure point for pharmaceutical intermediates subject to rigorous ICH Q3D guidelines. These conventional processes frequently require harsh reaction conditions, extended reaction times, and the use of large volumes of organic solvents, all of which contribute to elevated operational costs and significant environmental burdens. The reliance on stoichiometric amounts of base also generates substantial salt waste, complicating waste stream management and increasing the overall cost of goods sold (COGS) for high-volume production runs.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN102432487A utilizes the Lewis basic ionic liquid [HDBU]OAc to facilitate aminoacetylation with exceptional efficiency and selectivity. This innovative approach enables the reaction to proceed under solvent-free conditions, eliminating the need for hazardous volatile organic compounds and simplifying the reactor setup. The reaction profile is remarkably fast, often completing within 0.3 to 1.5 hours at moderate temperatures around 50°C, which significantly lowers energy consumption compared to refluxing conditions required by older protocols. 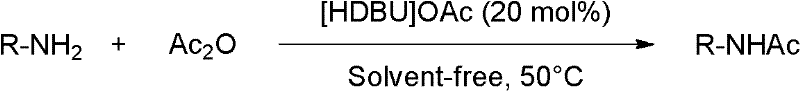 As illustrated in the general reaction scheme, the process accommodates a wide variety of substrates, converting simple amines into their corresponding N-acetyl derivatives with yields frequently exceeding 90% and purities reaching up to 99%. The workup procedure is equally streamlined; post-reaction, the mixture can be diluted with ether, filtered, and washed with simple aqueous solutions like saturated sodium bicarbonate, avoiding the need for specialized scavengers or complex extraction protocols. This simplicity translates directly into reduced processing time and lower labor costs, positioning this method as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing.
As illustrated in the general reaction scheme, the process accommodates a wide variety of substrates, converting simple amines into their corresponding N-acetyl derivatives with yields frequently exceeding 90% and purities reaching up to 99%. The workup procedure is equally streamlined; post-reaction, the mixture can be diluted with ether, filtered, and washed with simple aqueous solutions like saturated sodium bicarbonate, avoiding the need for specialized scavengers or complex extraction protocols. This simplicity translates directly into reduced processing time and lower labor costs, positioning this method as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lewis Base Ionic Liquid Catalysis
The efficacy of [HDBU]OAc stems from its unique dual nature as both a Brønsted base and a Lewis base within an ionic liquid matrix. The catalyst is synthesized by the neutralization of DBU with acetic acid, forming a stable salt that retains the nucleophilic character of the amidine nitrogen while gaining the solubility and stability properties of an ionic liquid. 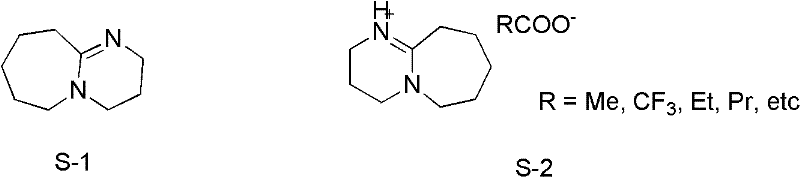 In the catalytic cycle, the basic nitrogen of the [HDBU] cation activates the acetic anhydride by coordinating with the carbonyl oxygen, thereby increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the amine substrate, facilitating rapid acetyl transfer. Unlike heterogeneous catalysts that suffer from mass transfer limitations, this homogeneous ionic liquid system ensures intimate contact between the catalyst and reactants, leading to uniform reaction kinetics. Furthermore, the ionic nature of the catalyst prevents it from volatilizing during the reaction, ensuring that it remains in the reaction vessel and can potentially be recovered or at least does not contaminate the distillate. The preparation of the catalyst itself is straightforward, involving the dropwise addition of glacial acetic acid to DBU at low temperatures (0-5°C) followed by vacuum drying, as shown in the structural evolution.
In the catalytic cycle, the basic nitrogen of the [HDBU] cation activates the acetic anhydride by coordinating with the carbonyl oxygen, thereby increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the amine substrate, facilitating rapid acetyl transfer. Unlike heterogeneous catalysts that suffer from mass transfer limitations, this homogeneous ionic liquid system ensures intimate contact between the catalyst and reactants, leading to uniform reaction kinetics. Furthermore, the ionic nature of the catalyst prevents it from volatilizing during the reaction, ensuring that it remains in the reaction vessel and can potentially be recovered or at least does not contaminate the distillate. The preparation of the catalyst itself is straightforward, involving the dropwise addition of glacial acetic acid to DBU at low temperatures (0-5°C) followed by vacuum drying, as shown in the structural evolution. 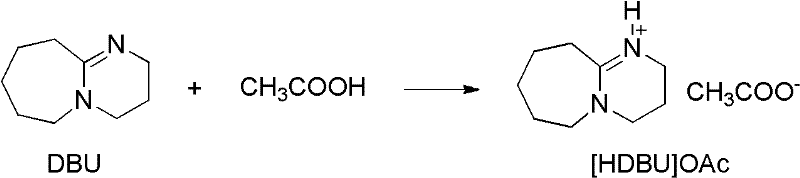 This controlled synthesis ensures high purity of the catalyst, which is crucial for minimizing side reactions and impurity formation in the final API intermediate.
This controlled synthesis ensures high purity of the catalyst, which is crucial for minimizing side reactions and impurity formation in the final API intermediate.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal catalysis. Since the catalyst is metal-free, there is zero risk of leaching toxic metals like palladium, copper, or nickel into the product stream, a common concern that often requires expensive purification resins to rectify. The mild reaction conditions (40-80°C) also suppress thermal degradation pathways that can lead to polymerization or decomposition of sensitive functional groups on the amine substrate. For instance, substrates containing electron-withdrawing groups like nitro or trifluoromethyl groups, which might be sensitive to harsh acidic or basic conditions, are tolerated well under this protocol. The absence of water sensitivity in the catalyst system further enhances its robustness, allowing for operations without stringent anhydrous conditions that are typically required for Lewis acid catalysts. This tolerance simplifies the raw material specifications and reduces the cost of drying solvents or reagents, contributing to a more resilient and forgiving manufacturing process that maintains high purity specifications consistently.
How to Synthesize N-Acetyl Amines Efficiently
The implementation of this catalytic system into a standard operating procedure is designed for ease of adoption in existing pilot and production facilities. The protocol eliminates the complexity associated with handling moisture-sensitive reagents or toxic volatile bases, making it accessible for operators with standard chemical training. The process begins with the precise measurement of the amine substrate and acetic anhydride, followed by the addition of the pre-synthesized [HDBU]OAc catalyst at a loading of 10-30 mol%. The reaction mixture is then heated gently, typically to 50°C, and monitored via TLC or GC until conversion is complete, which usually occurs within an hour. Detailed standardized synthesis steps see the guide below.
- Prepare the Lewis basic ionic liquid [HDBU]OAc by neutralizing DBU with glacial acetic acid at 0-5°C, followed by vacuum drying.
- Mix the amine substrate (aliphatic or aromatic) with acetic anhydride (1.0-2.0 equiv) and add 10-30 mol% of the [HDBU]OAc catalyst.
- Heat the mixture to 40-80°C under solvent-free conditions for 0.3-1.5 hours, then perform aqueous workup and extraction to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this [HDBU]OAc catalyzed process offers tangible strategic benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; by replacing expensive, specialized catalysts like DMAP or precious metal complexes with a readily available organic base derivative, companies can mitigate supply risks and stabilize input costs. The solvent-free nature of the reaction means that facilities do not need to procure, store, or dispose of large volumes of flammable organic solvents, which significantly reduces insurance premiums and regulatory compliance burdens associated with VOC emissions. Moreover, the elimination of heavy metals from the process flow removes the need for costly metal scavenger resins and the associated validation testing for residual metals, streamlining the quality control workflow. This holistic reduction in process complexity translates to a shorter overall cycle time from raw material intake to finished goods, enhancing the agility of the supply chain to respond to market demands for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this ionic liquid catalysis is profound, driven primarily by the elimination of solvent costs and the reduction in waste disposal fees. Traditional acetylation processes often generate significant volumes of saline waste due to the neutralization of stoichiometric acids or bases, whereas this catalytic method operates with high atom economy. The ability to run the reaction at lower temperatures (40-80°C) compared to traditional reflux conditions results in substantial energy savings on heating and cooling utilities over the course of annual production. Additionally, the simplified workup procedure, which relies on basic filtration and washing rather than column chromatography or distillation, reduces labor hours and equipment occupancy time. These factors combine to lower the variable cost per kilogram of the intermediate, providing a competitive margin advantage in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Reliability in the supply of key intermediates is paramount for preventing production stoppages in downstream API synthesis. The catalyst [HDBU]OAc is synthesized from DBU and acetic acid, both of which are commodity chemicals with robust, global supply chains, reducing the risk of single-source bottlenecks. The stability of the ionic liquid catalyst allows for easier storage and handling compared to hygroscopic or air-sensitive alternatives, ensuring consistent performance batch after batch. Furthermore, the broad substrate scope demonstrated in the patent, covering various substituted anilines and aliphatic amines, means that a single catalytic platform can be used for multiple product lines. This versatility allows manufacturers to consolidate their catalyst inventory and simplify their procurement strategies, fostering a more resilient and adaptable supply network capable of weathering market fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden challenges, but this solvent-free methodology is inherently scalable due to the absence of solvent volume constraints. Reactors can be filled to higher capacities since the reaction mass consists primarily of reactants rather than diluents, improving throughput per batch. From an environmental standpoint, the process aligns perfectly with Green Chemistry principles by minimizing waste generation and avoiding toxic reagents. The absence of heavy metals and volatile amines in the waste stream simplifies effluent treatment, reducing the load on wastewater treatment plants and ensuring compliance with increasingly strict environmental regulations. This 'green' credential is not just a regulatory checkbox but a valuable asset for companies aiming to meet corporate sustainability goals and appeal to environmentally conscious partners in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this Lewis basic ionic liquid catalysis technology. These insights are derived directly from the experimental data and embodiments detailed in the patent documentation, providing a reliable foundation for process evaluation. Understanding these nuances is critical for technical teams assessing the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What is the optimal catalyst loading for this aminoacetylation reaction?
A: According to patent CN102432487A, the molar ratio of the Lewis base ionic liquid [HDBU]OAc to the substrate should be between 10% and 30%. This loading ensures high conversion rates while maintaining economic efficiency compared to stoichiometric bases.
Q: Does this method support both aliphatic and aromatic amines?
A: Yes, the technology demonstrates broad substrate scope. It is effective for various aliphatic amines such as benzylamine and cyclohexylamine, as well as substituted aromatic amines including nitro-, methoxy-, and halogen-substituted anilines, yielding products with 96%-99% purity.
Q: How does this process compare to traditional DMAP catalysis regarding purification?
A: Unlike traditional methods using DMAP or pyridine which often require complex chromatographic purification to remove toxic residues, this ionic liquid method allows for simple aqueous washing and filtration. The catalyst system avoids heavy metal contamination, significantly simplifying the downstream processing workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetyl Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the [HDBU]OAc system in driving efficiency and quality in chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale methods are successfully translated into robust industrial processes. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical industry, consistency is key, and our dedicated process development teams work tirelessly to optimize reaction parameters, ensuring that the high yields and purity levels reported in the patent are consistently achieved at scale.
We invite forward-thinking partners to collaborate with us to leverage this green acetylation technology for your specific project needs. Whether you require custom synthesis of complex intermediates or scale-up of existing routes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out to us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that balance technical performance with commercial viability. Together, we can build a more sustainable and efficient supply chain for the next generation of life-saving medicines.
