Revolutionizing Asymmetric Piperazine Production: A Green Aqueous Phase Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to synthesize complex asymmetric intermediates, particularly piperazine derivatives which serve as critical scaffolds in drug discovery and agrochemical formulations. A groundbreaking approach detailed in patent CN112939894A introduces a novel aqueous phase preparation method for 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine, addressing long-standing challenges in regioselectivity and cost-efficiency. This technology leverages a sophisticated single-end-group protection strategy using simple inorganic or organic acids in a water-based medium, effectively bypassing the need for expensive protecting groups like BOC or cumbersome salt formations. By shifting the paradigm from organic solvent-heavy processes to a greener aqueous system, this innovation not only enhances the environmental profile of the synthesis but also drastically simplifies the downstream purification workflow. For R&D teams and procurement strategists alike, this represents a significant leap forward in the manufacturing of high-purity asymmetric piperazines, offering a robust pathway to reduce production costs while maintaining stringent quality standards required for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of asymmetric hydroxyalkyl piperazines has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Conventional routes often rely on the use of N-BOC protected piperazine or pre-synthesized mono-substituted starting materials like N-(2-hydroxyethyl)piperazine, both of which command premium prices in the global chemical market. The reliance on BOC anhydride introduces additional synthetic steps for protection and deprotection, generating substantial chemical waste and increasing the overall process mass intensity. Furthermore, existing methods frequently struggle with selectivity issues, where the symmetrical nature of the piperazine ring leads to the formation of unwanted di-substituted byproducts, thereby lowering the yield of the desired asymmetric target. These inefficiencies necessitate complex purification protocols, often involving multiple chromatographic separations or distillations, which drive up operational expenditures and extend lead times for final API intermediates. Additionally, many legacy processes utilize volatile organic solvents that pose safety risks and require specialized containment infrastructure, further complicating the supply chain logistics for hazardous material handling.
The Novel Approach
In stark contrast, the methodology disclosed in CN112939894A revolutionizes the synthesis landscape by employing a direct, aqueous-phase single-end-group protection technique that utilizes commodity-grade raw materials. Instead of expensive organic protecting agents, this process uses readily available inorganic acids such as sulfuric acid or organic acids like citric acid to temporarily mask one reactive nitrogen site on the piperazine ring. This strategic protonation creates a mono-ammonium salt intermediate that directs the subsequent alkylation exclusively to the free amine, effectively suppressing the formation of symmetrical di-substituted impurities. The use of water as the primary reaction medium eliminates the need for costly anhydrous conditions and reduces the fire hazard associated with organic solvents, making the process inherently safer for operators and the environment. Moreover, the deprotection step is elegantly integrated into the workup procedure by simply adjusting the pH, allowing for the seamless transition to the second alkylation step without isolating unstable intermediates. This streamlined approach not only boosts the overall yield of the target 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine to impressive levels but also significantly lowers the barrier to entry for commercial manufacturing.
Mechanistic Insights into Aqueous Phase Single-End-Group Protection
The core brilliance of this synthesis lies in its precise control over the nucleophilicity of the piperazine nitrogen atoms through acid-base chemistry. Initially, piperazine is dissolved in water and treated with a stoichiometric amount of acid, resulting in the formation of a piperazine mono-ammonium salt. In this state, the protonated nitrogen loses its nucleophilic character due to the positive charge, while the remaining unprotonated nitrogen retains its ability to attack electrophiles. When the first epoxide, such as propylene oxide, is introduced, it undergoes a ring-opening reaction exclusively at the free amine site, forming a stable N-monosubstituted intermediate. This selective mono-alkylation is the critical determinant of success, as it prevents the statistical distribution of products that typically plagues symmetric diamine reactions. The reaction conditions, including temperature and pH, are meticulously optimized to maintain the integrity of the mono-protected species while facilitating rapid conversion of the epoxide. Following the first alkylation, the reaction mixture is treated with a base to raise the pH to between 9 and 14, which deprotonates the ammonium salt and regenerates the secondary amine functionality. This regenerated amine is then free to react with the second epoxide, ethylene oxide, completing the asymmetric functionalization of the piperazine ring.
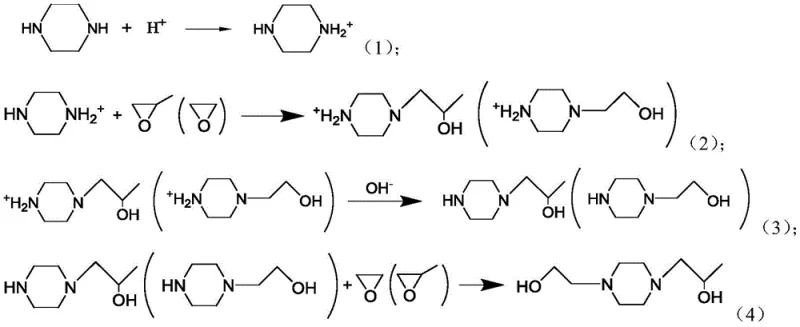
Beyond the primary reaction pathway, the mechanism also incorporates robust impurity control measures that are vital for pharmaceutical grade outputs. The aqueous environment plays a dual role by acting as both a solvent and a heat sink, absorbing the exothermic heat generated during the epoxide ring-opening reactions, which helps prevent thermal runaway and side reactions. The choice of acid protecting agent is also pivotal; polybasic acids like citric acid can offer buffering capacity that stabilizes the pH during the reaction, further enhancing selectivity. Furthermore, the separation strategy leverages the differential solubility of the intermediates and byproducts in the aqueous phase versus organic extractants. By adjusting the pH to alkaline conditions, the desired product remains in the aqueous phase while certain organic impurities or unreacted starting materials can be partitioned out or filtered off. This inherent purification capability reduces the load on final distillation columns, ensuring that the final 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine meets high purity specifications with minimal residual solvents or heavy metal contaminants. The result is a chemically elegant process that maximizes atom economy while minimizing waste generation.
How to Synthesize 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine Efficiently
The practical implementation of this synthesis route involves a sequence of well-defined unit operations that are compatible with standard chemical processing equipment. The process begins with the dissolution of piperazine in water followed by the controlled addition of the acid protecting agent under reflux conditions to ensure complete salt formation. Once the mono-ammonium salt is generated, the first epoxide is added dropwise to manage the exotherm, followed by a holding period to ensure full conversion. The subsequent deprotection and pH adjustment step is critical for activating the second nitrogen site, after which the second epoxide is introduced to finalize the molecular architecture.
- Dissolve piperazine in water and add a single-end-group protective agent (inorganic or organic acid) to form a mono-ammonium salt intermediate.
- React the protected intermediate with the first epoxide (propylene oxide or ethylene oxide) under controlled temperature to achieve mono-substitution.
- Adjust pH to 9-14 for deprotection, separate the water phase, and react with the second epoxide to form the final asymmetric product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this aqueous phase technology offers transformative benefits that directly impact the bottom line and operational resilience. The most immediate advantage is the drastic reduction in raw material costs, as the process replaces expensive specialty reagents like BOC-anhydride or pre-functionalized piperazines with bulk commodity chemicals such as sulfuric acid, citric acid, and industrial grade epoxides. These inputs are widely available from multiple global suppliers, mitigating the risk of supply disruptions and providing significant leverage in price negotiations. Furthermore, the elimination of complex protection-deprotection sequences reduces the number of processing steps, which translates to lower energy consumption, reduced labor hours, and decreased usage of auxiliary solvents. This simplification of the manufacturing workflow allows for faster batch turnover times, enabling suppliers to respond more agilely to fluctuating market demands without the need for excessive inventory buffers. The reliance on water as a solvent also alleviates the regulatory and logistical burdens associated with the storage and disposal of volatile organic compounds, streamlining compliance with environmental health and safety regulations.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of high-cost reagents with low-cost acids and the minimization of solvent usage. By avoiding the purchase of protected amine starting materials, manufacturers can achieve substantial savings on the bill of materials, which is often the largest component of production costs. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, leading to higher overall yields and reduced waste disposal fees. The simplified workup procedure, which relies on pH adjustment and filtration rather than complex extractions or chromatography, further lowers the operational expenditure associated with purification. These cumulative efficiencies create a highly competitive cost structure that allows for better margin management in the face of raw material price volatility.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the key raw materials—piperazine, ethylene oxide, and propylene oxide—are produced on a massive industrial scale for various sectors including polymers and surfactants. This broad demand base ensures a consistent supply flow and reduces the likelihood of shortages that can plague niche fine chemical intermediates. The robustness of the aqueous chemistry also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in supplier selection. Consequently, procurement teams can diversify their vendor base without compromising on product quality, thereby building a more resilient supply chain capable of withstanding geopolitical or logistical shocks. The reduced dependency on specialized catalysts or exotic solvents further decouples production from fragile supply lines.
- Scalability and Environmental Compliance: From a scale-up perspective, the use of water as a reaction medium presents significant engineering advantages, particularly regarding heat transfer and safety. Exothermic reactions involving epoxides are much easier to control in water due to its high heat capacity, reducing the risk of thermal runaways during large-batch production. This safety profile facilitates smoother technology transfer from pilot plants to multi-ton commercial reactors. Environmentally, the process aligns with green chemistry principles by reducing the emission of VOCs and generating less hazardous waste, which simplifies permitting and lowers the cost of environmental compliance. The ability to recycle the aqueous phase or treat it with standard wastewater protocols makes this method highly attractive for facilities aiming to reduce their carbon footprint and meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous phase synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: What is the primary advantage of the aqueous phase protection method over traditional BOC protection?
A: The aqueous phase method utilizes inexpensive inorganic or organic acids instead of costly BOC anhydride, significantly reducing raw material costs and simplifying the deprotection process without requiring complex organic solvents.
Q: How does this process control the formation of symmetrical byproducts?
A: By protonating one nitrogen atom of the piperazine ring to form a mono-ammonium salt, the reactivity of that specific site is temporarily suppressed, ensuring that the first alkylation occurs selectively at the unprotonated nitrogen.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process uses water as the primary solvent and commodity chemicals like piperazine and epoxides, making it inherently safer and easier to scale compared to processes requiring anhydrous conditions or exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical supply chain. Our team of expert chemists has thoroughly analyzed the potential of the aqueous phase single-end-group protection method and is fully equipped to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant scale is seamless and efficient. Our state-of-the-art facilities are designed to handle reactive chemistries safely, with rigorous QC labs dedicated to maintaining stringent purity specifications for every batch of 1-(2-hydroxyethyl)-4-(2-hydroxypropyl)piperazine we produce. We understand that consistency is key for our partners, and our robust quality management systems guarantee that our intermediates meet the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your manufacturing costs with our reliable, high-quality piperazine derivatives.
