Advanced One-Step Cyclization Strategy for High-Purity Azilsartan Manufacturing
Introduction to Next-Generation Azilsartan Synthesis
The global pharmaceutical landscape is increasingly driven by the urgent need for effective cardiovascular therapeutics, particularly as the prevalence of hypertension continues to rise among aging populations worldwide. Azilsartan, a potent angiotensin II receptor blocker (ARB), has emerged as a critical treatment option due to its superior efficacy and duration of action compared to earlier generation ARBs. However, the commercial viability of this life-saving medication has historically been constrained by complex, hazardous, and low-yielding synthetic routes that struggle to meet modern green chemistry standards. A groundbreaking advancement in this field is detailed in patent CN115703769A, which discloses a novel preparation method that fundamentally reimagines the construction of the oxadiazole ring, a key pharmacophore in the Azilsartan structure. This patent introduces a streamlined approach utilizing ammonium carbonate as a dual-purpose reagent, effectively replacing dangerous and expensive traditional cyclization agents. By shifting from multi-step sequences involving unstable intermediates to a direct, one-pot cyclization strategy, this technology offers a pathway to significantly higher purity and yield, addressing the critical supply chain bottlenecks that have previously limited the widespread availability of cost-effective Azilsartan.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Azilsartan has relied on precarious chemical transformations that introduce substantial operational risks and cost inefficiencies. Traditional routes typically involve the condensation of a nitrile-containing precursor with hydroxylamine or its salts at elevated temperatures to form an amidoxime intermediate, a step notorious for the thermal instability and potential explosion hazards associated with hydroxylamine derivatives. Following this, the introduction of the carbonyl group to close the oxadiazole ring often necessitates the use of highly toxic reagents such as ethyl chloroformate or carbonyl diimidazole (CDI). As illustrated in prior art pathways, these reagents not only carry genotoxicity warnings that complicate regulatory approval but also generate stoichiometric amounts of hazardous waste. Furthermore, the use of ethyl chloroformate can lead to side reactions with alcohol solvents, creating difficult-to-separate impurities that compromise the safety profile of the final drug substance. These legacy methods typically achieve a total yield of only 40-55% over three steps, representing a significant loss of valuable starting materials and inflating the cost of goods sold (COGS) for manufacturers.
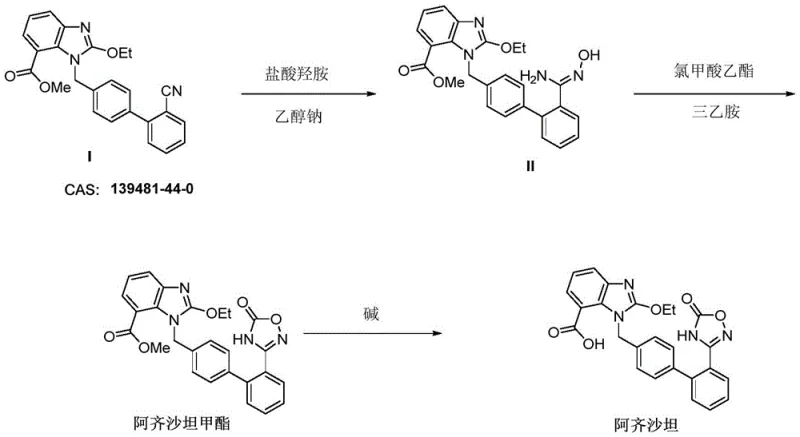
The Novel Approach
In stark contrast to these hazardous legacy processes, the methodology disclosed in patent CN115703769A presents a paradigm shift by employing ammonium carbonate as a safe, inexpensive, and efficient cyclization agent. This innovative route allows for the direct conversion of the nitrile precursor (Compound I) into the Azilsartan ester intermediate in a single step, bypassing the isolation of unstable amidoxime intermediates entirely. The reaction proceeds smoothly in polar aprotic solvents such as dimethyl sulfoxide (DMSO) or N-methylpyrrolidone (NMP) under moderate pressure and temperature conditions, typically around 100-110°C. This one-pot transformation not only eliminates the explosion risks associated with hydroxylamine but also removes the need for genotoxic carbonylating agents, thereby drastically simplifying the impurity profile. The byproducts of the ammonium carbonate decomposition are merely ammonia and carbon dioxide, which are easily vented from the reactor, leaving behind a clean reaction mixture that facilitates straightforward product isolation. This streamlined approach represents a significant leap forward in process safety and environmental compliance, aligning perfectly with the principles of sustainable pharmaceutical manufacturing.
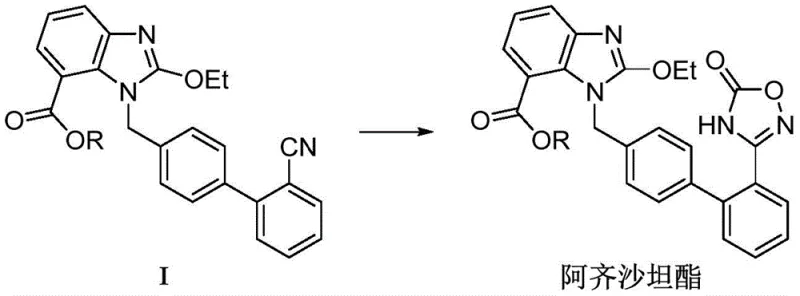
Mechanistic Insights into Ammonium Carbonate-Mediated Cyclization
The chemical elegance of this new process lies in the unique reactivity of ammonium carbonate, which serves as a source of both nitrogen and carbon for the construction of the 1,2,4-oxadiazole-5-one ring system. Under the heated conditions within a closed autoclave system, ammonium carbonate decomposes to release ammonia and carbon dioxide in situ. The ammonia acts as a nucleophile, attacking the electrophilic carbon of the nitrile group on the biphenyl scaffold to form an amidine-like intermediate. Subsequently, the intramolecular cyclization occurs through the interaction of this nitrogen species with the adjacent ester carbonyl or activated carboxyl functionality, facilitated by the carbon dioxide species acting as the carbonyl source. This mechanism avoids the high-energy transition states required in traditional hydroxylamine condensations, resulting in a more controlled and selective reaction pathway. The use of polar aprotic solvents is critical here, as they stabilize the charged transition states and ensure the solubility of the ionic ammonium carbonate species, thereby driving the equilibrium towards the desired cyclic product. This mechanistic efficiency is reflected in the high conversion rates observed, where HPLC monitoring confirms the complete consumption of the starting material Compound I within relatively short reaction times.
From an impurity control perspective, this mechanism offers distinct advantages by inherently suppressing the formation of genotoxic byproducts. In conventional routes using ethyl chloroformate, there is a persistent risk of alkylating impurities or residual chloroformate adducts that require rigorous purging to meet ICH M7 guidelines. The ammonium carbonate route, being metal-free and halogen-free, generates a much cleaner crude product profile. The primary impurities observed are typically structural analogs related to incomplete cyclization or hydrolysis, which are far easier to remove via standard recrystallization techniques than the covalent adducts formed by reactive acylating agents. Furthermore, the mild basicity of the ammonium carbonate system prevents the degradation of the sensitive biphenyl-tetrazole or biphenyl-methyl scaffold that can occur under the harsh acidic or strongly basic conditions of older methods. This preservation of molecular integrity ensures that the final API maintains high optical and chemical purity, which is essential for consistent therapeutic performance and regulatory acceptance in global markets.
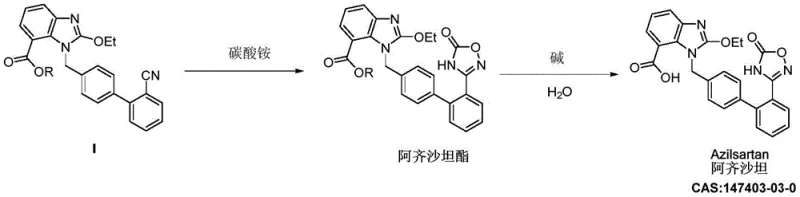
How to Synthesize Azilsartan Efficiently
The implementation of this patented synthesis route requires precise control over reaction parameters to maximize yield and purity while ensuring operational safety. The process begins with the charging of Compound I and ammonium carbonate into a pressure-rated reactor, followed by the addition of a suitable solvent system. The reaction is then heated to the optimal temperature range under an inert atmosphere to prevent oxidation. Once the cyclization is complete, the system is carefully cooled, and the gaseous byproducts are vented before the product is precipitated. The resulting ester intermediate is then subjected to a hydrolysis step to yield the final free acid form of Azilsartan. For a detailed breakdown of the specific molar ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Cyclization Reaction: React Compound I (nitrile precursor) with ammonium carbonate in a polar aprotic solvent like DMSO or NMP at 100-110°C under pressure.
- Isolation: Cool the reaction mixture, release gases, and precipitate the azilsartan ester intermediate using an ice-water mixture.
- Hydrolysis and Purification: Hydrolyze the ester intermediate with alkali (NaOH/KOH) in water, adjust pH to precipitate crude Azilsartan, and recrystallize for final purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ammonium carbonate-based synthesis route offers compelling strategic advantages that extend beyond simple technical metrics. The elimination of hazardous reagents like hydroxylamine and ethyl chloroformate translates directly into reduced costs associated with specialized storage, handling, and waste disposal. Traditional processes often require dedicated explosion-proof facilities and complex scrubbing systems to manage toxic off-gases, representing a significant capital expenditure (CAPEX) and operational expenditure (OPEX) burden. By switching to a benign reagent like ammonium carbonate, manufacturers can utilize standard glass-lined reactors and simplify their environmental health and safety (EHS) protocols, leading to substantial cost reduction in pharmaceutical intermediate manufacturing. Additionally, the high atom economy of this one-step cyclization minimizes raw material waste, ensuring that a greater proportion of the input mass is converted into valuable product, which further drives down the unit cost of production.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic coupling reagents such as carbonyl diimidazole (CDI) and ethyl chloroformate with commodity-grade ammonium carbonate results in a drastic reduction in raw material costs. CDI is a high-value specialty chemical with a volatile price point, whereas ammonium carbonate is a widely available bulk chemical with a stable and low cost. Furthermore, the simplification of the synthetic sequence from three steps to essentially two (cyclization followed by hydrolysis) reduces labor hours, energy consumption, and solvent usage. The removal of the need for extensive purification steps to remove genotoxic impurities also lowers the cost of quality control and analytical testing, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Relying on hazardous reagents often introduces supply chain fragility, as the availability of chemicals like hydroxylamine can be restricted due to regulatory controls or transportation limitations. Ammonium carbonate, being a non-regulated, stable solid, is readily sourced from multiple global suppliers, ensuring a robust and continuous supply of key starting materials. This diversification of the supply base mitigates the risk of production stoppages caused by raw material shortages. Moreover, the improved stability of the reaction intermediates allows for more flexible scheduling and potential campaign manufacturing, enabling suppliers to build inventory buffers and respond more agilely to fluctuations in market demand for Azilsartan APIs.
- Scalability and Environmental Compliance: The process conditions described in the patent, operating at moderate temperatures and pressures, are highly amenable to scale-up from pilot plant to commercial multi-ton production. The use of common industrial solvents like DMSO and NMP, which have established recovery and recycling protocols, facilitates the implementation of closed-loop solvent systems that minimize environmental discharge. The generation of benign gaseous byproducts (ammonia and carbon dioxide) simplifies废气 treatment requirements compared to the complex waste streams generated by chloroformate-based routes. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the manufacturing process against increasingly stringent global environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Azilsartan synthesis method. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's feasibility. Understanding these details is crucial for R&D teams assessing process transfer and for procurement teams evaluating supplier capabilities.
Q: What are the safety advantages of using ammonium carbonate over hydroxylamine in Azilsartan synthesis?
A: Traditional methods utilize hydroxylamine, which poses significant explosion risks at high temperatures and requires strict handling protocols. The patented method replaces this with ammonium carbonate, a stable, non-explosive solid that decomposes into harmless gases (ammonia and carbon dioxide), drastically improving process safety and reducing the need for specialized explosion-proof infrastructure.
Q: How does this new route impact the impurity profile of the final API?
A: By eliminating genotoxic reagents such as ethyl chloroformate and avoiding unstable intermediates associated with hydroxylamine condensation, this route significantly reduces the formation of difficult-to-remove toxic impurities. The result is a cleaner reaction profile that simplifies downstream purification and ensures the final Azilsartan meets stringent regulatory purity specifications without complex chromatographic steps.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for scalability. It utilizes common industrial solvents like DMSO or NMP and operates at moderate temperatures (100-110°C) and pressures (0.6-0.8 MPa) achievable in standard glass-lined or stainless steel autoclaves. The high atom economy and simplified one-step cyclization reduce waste generation, making it economically and environmentally viable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azilsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is paramount for the sustainable growth of the pharmaceutical industry. Our technical team has thoroughly analyzed the potential of the ammonium carbonate cyclization method disclosed in patent CN115703769A and is well-positioned to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with the necessary pressure-rated reactors and advanced purification systems to handle this chemistry with precision, guaranteeing stringent purity specifications and consistent batch-to-batch quality for your Azilsartan requirements.
We invite you to collaborate with us to optimize your supply chain for this critical antihypertensive agent. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener synthesis route for your specific volume needs. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss detailed route feasibility assessments tailored to your project timelines. Let us help you secure a reliable, cost-effective, and compliant supply of high-purity Azilsartan for the global market.
