Advanced Synthetic Route for Lobeline: Cost-Effective Manufacturing for Pharmaceutical Intermediates
Introduction to Advanced Lobeline Synthesis Technology
Lobeline, chemically known as 2-[(2R,6S)-6-[(2S)-2-hydroxy-2-phenylethyl]-1-methylpiperidine]-1-acetophenone, is a critical alkaloid utilized primarily as a respiratory stimulant and smoking cessation aid. Its therapeutic efficacy relies heavily on its specific stereochemical configuration, making the asymmetric synthesis of this molecule a significant challenge in organic chemistry. The patent CN109232387B discloses a groundbreaking synthetic method that addresses the longstanding inefficiencies in Lobeline production. Unlike previous methodologies that struggled with low yields and complex purification, this invention offers a streamlined 10-step route that achieves high chiral purity and excellent overall yield.
The core innovation lies in the strategic use of S-tert-butyl sulfinamide as a chiral auxiliary, which effectively controls the stereochemistry of the key carbon centers without the need for expensive transition metal catalysts. This approach not only enhances the optical purity of the final product but also drastically simplifies the operational requirements for manufacturing. By shifting away from enzymatic resolutions or high-pressure hydrogenation, this technology represents a paradigm shift towards more robust and economically viable pharmaceutical intermediate production.
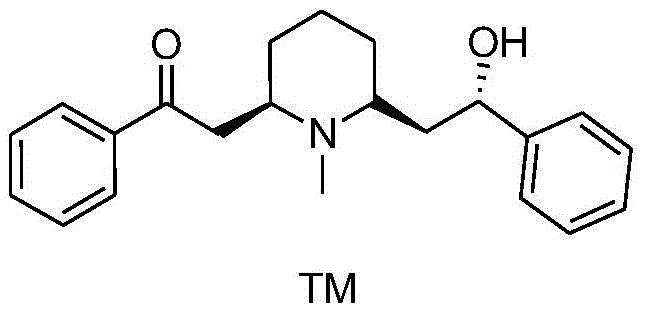
For procurement managers and supply chain directors, the implications of this technology are profound. The reliance on readily available starting materials such as 3,4-dihydropyran and ethanethiol ensures a stable supply chain, while the avoidance of precious metal catalysts reduces both raw material costs and the regulatory burden associated with heavy metal residue limits in APIs. This synthesis route exemplifies the kind of process intensification that modern fine chemical manufacturers seek to implement to remain competitive in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Lobeline has been plagued by significant technical and economic hurdles. Early routes attempted to construct the four chiral centers one by one, resulting in excessively long reaction sequences with cumulative yield losses that made industrial application impractical. As asymmetric chemistry evolved, methods employing chiral catalysts or enzymes were introduced, yet these brought their own set of complications. For instance, non-enzymatic asymmetric acylation using (S)-BTM requires extended reaction times of up to two days, followed by tedious oxidation and deacylation steps.
Furthermore, alternative approaches utilizing asymmetric reduction with bidentate phosphine ligands necessitate high-pressure conditions ranging from 15 to 25 bar. Such high-pressure hydrogenation processes demand specialized reactor equipment, rigorous safety protocols, and substantial capital investment, all of which inflate the cost of goods sold (COGS). The combination of harsh reaction conditions, expensive chiral ligands, and low overall yields has traditionally created a bottleneck in the reliable supply of high-purity Lobeline for the pharmaceutical industry.
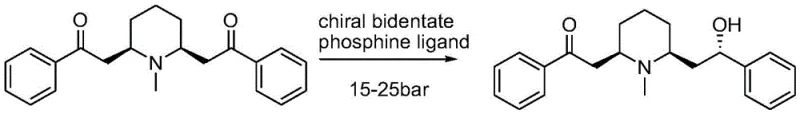
The Novel Approach
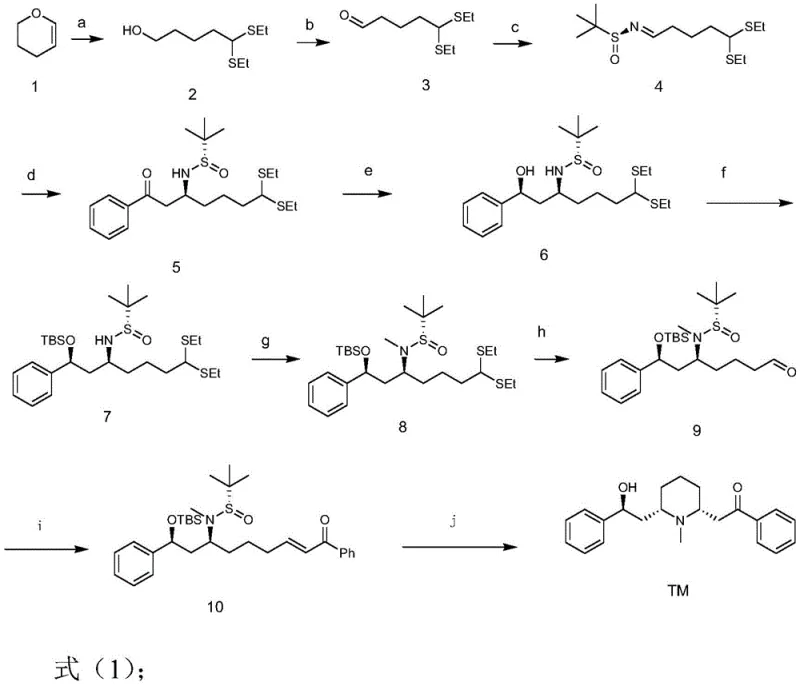
The synthetic method disclosed in patent CN109232387B offers a decisive solution to these challenges by employing a convergent strategy centered on chiral sulfinimine chemistry. The route begins with the ring-opening of 3,4-dihydropyran, followed by oxidation and condensation with S-tert-butyl sulfinamide to generate a chiral imine intermediate. This intermediate serves as a powerful electrophile that directs the subsequent addition of acetophenone enolates with high diastereoselectivity, effectively setting the crucial stereocenters early in the synthesis.
This novel approach eliminates the need for high-pressure equipment and expensive chiral catalysts, relying instead on stoichiometric chiral auxiliaries that are cost-effective and easy to handle. The reaction conditions are notably mild, typically proceeding at atmospheric pressure and moderate temperatures, which significantly lowers the barrier to entry for manufacturing partners. The entire process, from raw materials to the final cyclized product, is designed for operational simplicity, featuring straightforward workup procedures such as aqueous washes and crystallization rather than complex chromatographic separations.
Mechanistic Insights into Sulfinamide-Mediated Asymmetric Synthesis
The success of this synthetic route hinges on the precise control of stereochemistry during the carbon-carbon bond-forming steps. The mechanism initiates with the formation of a chiral N-tert-butanesulfinyl imine from the aldehyde precursor. The bulky tert-butyl group on the sulfur atom creates a steric environment that shields one face of the imine double bond. When the acetophenone enolate, generated using a strong base like potassium tert-butoxide at low temperatures (below -50°C), attacks the imine, it is forced to approach from the less hindered face. This facial selectivity ensures the formation of the desired (S)-configuration at the benzylic position with high fidelity.
Following the addition, the subsequent reduction step utilizes a specific borane-based reducing agent to convert the ketone moiety into a secondary alcohol while maintaining the integrity of the existing chiral centers. The use of this specific reducing environment prevents epimerization and ensures that the stereochemical information encoded by the sulfinamide auxiliary is faithfully transferred to the final carbon skeleton. The auxiliary is eventually removed during the final acidic hydrolysis and cyclization step, where the amine nitrogen attacks the ketone to close the piperidine ring, simultaneously cleaving the sulfur-nitrogen bond and releasing the free amine of Lobeline.
Impurity control is inherently built into this mechanism through the crystallinity of the intermediates. The sulfinamide intermediates often possess distinct physical properties that allow for purification via recrystallization before proceeding to the next step. This "purge capability" is critical for pharmaceutical manufacturing, as it prevents the carryover of diastereomeric impurities that could be difficult to separate in the final API. By establishing the correct stereochemistry early and maintaining it through robust protection and deprotection strategies (such as TBS protection), the process minimizes the formation of hard-to-remove isomers, thereby ensuring a final product with optical purity exceeding 99%.
How to Synthesize Lobeline Efficiently
The synthesis of Lobeline via this patented route involves a logical sequence of ten chemical transformations that convert simple feedstocks into the complex alkaloid structure. The process is characterized by its modularity, allowing for quality control checkpoints at key intermediates such as the chiral sulfinimine and the protected amino-alcohol. Detailed standard operating procedures for each step, including specific molar ratios, temperature profiles, and quenching protocols, are essential for reproducing the high yields reported in the patent examples.
- Perform ring-opening of 3,4-dihydropyran with ethanethiol and Lewis acid, followed by oxidation to aldehyde and condensation with S-tert-butyl sulfinamide to form the chiral sulfinimine.
- Execute nucleophilic addition of acetophenone enolate to the sulfinimine, followed by stereoselective reduction and protection of the resulting hydroxyl group.
- Complete the piperidine ring formation via methylation, elimination, Wittig olefination, and final acidic hydrolysis/cyclization to yield Lobeline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like 3,4-dihydropyran and ethanethiol, manufacturers can mitigate the risks associated with sourcing specialized or single-source reagents. This diversification of the supply base enhances resilience against market volatility and ensures continuous production capability, which is paramount for meeting the demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and high-pressure hydrogenation equipment translates directly into lower capital expenditure (CAPEX) and operational expenditure (OPEX). Traditional routes often require precious metal ligands that are not only costly to purchase but also require expensive recovery or disposal processes to meet regulatory limits on heavy metals. By replacing these with stoichiometric sulfinamide auxiliaries and atmospheric pressure reactions, the process significantly reduces the cost per kilogram of the active pharmaceutical ingredient. Furthermore, the high yields observed at each step minimize waste generation, leading to substantial savings in raw material consumption and waste treatment costs.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and reduced downtime. Processes that operate under mild conditions are less prone to runaway reactions or equipment failures, ensuring a predictable production schedule. Additionally, the use of common solvents and reagents means that procurement teams are not held hostage by the lead times of exotic chemicals. This reliability allows for more accurate forecasting and inventory management, reducing the need for safety stock and freeing up working capital for other strategic investments within the organization.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior to legacy methods. The avoidance of high-pressure hydrogenation removes a major safety hazard from the plant floor, simplifying insurance and compliance requirements. Moreover, the atom economy of the route is improved by reducing the number of protection/deprotection cycles and avoiding stoichiometric oxidants that generate heavy metal waste. This aligns with the growing industry demand for green chemistry practices, making the manufacturing process more sustainable and easier to permit in jurisdictions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lobeline synthesis technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: How does this new synthesis method improve upon traditional Lobeline production?
A: Traditional methods often rely on expensive chiral catalysts, enzymatic resolution which is time-consuming, or high-pressure hydrogenation (15-25 bar) which poses safety risks. This novel route utilizes S-tert-butyl sulfinamide for efficient chiral induction under mild atmospheric conditions, significantly simplifying the process and reducing equipment costs.
Q: What are the key raw materials required for this synthesis?
A: The synthesis starts from commodity chemicals such as 3,4-dihydropyran and ethanethiol. Key reagents include S-tert-butyl sulfinamide for chirality control, acetophenone for the side chain, and standard protecting group reagents like TBSCl. These materials are widely available and cost-effective compared to specialized chiral catalysts.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids harsh conditions like high pressure and cryogenic temperatures below -50°C are only used for specific addition steps which are manageable in industrial reactors. The high yields reported in patent examples (e.g., >80% overall) and simple workup procedures indicate strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lobeline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates like Lobeline. Our team of expert process chemists has extensively evaluated the technology disclosed in CN109232387B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific low-temperature and moisture-sensitive steps required by this sulfinamide-mediated route, ensuring that stringent purity specifications are met consistently.
We invite global pharmaceutical companies and contract research organizations to collaborate with us on the commercialization of this advanced Lobeline synthesis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can optimize your supply chain for this vital respiratory stimulant intermediate.
