Advanced Gas-Phase Methylation Strategy for High-Purity N-(6-Chloro-3-Picolyl)-Methylamine Production
The global demand for high-efficiency neonicotinoid insecticides continues to drive innovation in the synthesis of their key precursors. A pivotal advancement in this domain is detailed in patent CN102827070A, which discloses an improved process for synthesizing N-(6-chloro-3-picolyl)-methylamine, a critical intermediate for producing imidacloprid and acetamiprid. This technology represents a significant departure from traditional aqueous-based methodologies by utilizing methylamine gas directly in a toluene solvent system. For R&D directors and process engineers, this shift addresses fundamental challenges regarding waste management and product quality. The patent outlines a robust protocol that not only enhances the purity of the final amine but also drastically simplifies the downstream purification workflow. By transitioning from a 30% monomethylamine aqueous solution to anhydrous methylamine gas, the process eliminates the massive water burden typically associated with this alkylation, thereby aligning modern manufacturing with stringent environmental compliance standards while simultaneously boosting yield metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-(6-chloro-3-picolyl)-methylamine has relied heavily on the use of 30% monomethylamine aqueous solutions. While this method is chemically feasible, it introduces substantial inefficiencies that plague large-scale operations. The primary drawback is the excessive generation of wastewater; since the reaction produces hydrochloric acid as a byproduct, which dissolves in the water phase, neutralization and disposal become costly and environmentally taxing. Furthermore, the presence of water promotes the hydrolysis of the chloromethyl group on the pyridine ring, leading to the formation of alcohol impurities that are difficult to separate. This side reaction not only consumes valuable raw materials but also caps the achievable purity of the final product, often limiting it to around 96%. Additionally, the volumetric inefficiency of carrying 70% water in the reagent means that reactor capacity is underutilized, forcing manufacturers to run more batches to meet production targets, which inflates operational expenditures and energy consumption.
The Novel Approach
The innovative process described in the patent data fundamentally re-engineers the reaction environment by replacing the aqueous amine source with pure methylamine gas dissolved in toluene. This anhydrous approach effectively suppresses hydrolytic side reactions, ensuring that the nucleophilic substitution proceeds with high selectivity towards the desired amine product. By operating in a non-polar organic solvent like toluene, the resulting methylamine hydrochloride salt precipitates or remains distinct, facilitating easier separation. The patent highlights that this modification allows for a significant increase in the loading capacity of the reaction kettle; specifically, the removal of water enables a roughly 30% increase in the amount of methylamine that can be processed per batch in the same vessel. This translates directly to enhanced throughput without capital investment in new hardware. Moreover, the absence of bulk water simplifies the work-up procedure, as the organic layer containing the product can be separated more cleanly, leading to a refined product purity that consistently exceeds 97.5%, with some batches reaching as high as 98.5%.
Mechanistic Insights into Gas-Phase Nucleophilic Substitution
The core chemical transformation involves a nucleophilic substitution where the lone pair of electrons on the nitrogen atom of methylamine attacks the electrophilic carbon of the chloromethyl group on the 2-chloro-5-chloromethylpyridine (2-chloro-5-PMC) molecule. In the conventional aqueous system, water molecules compete with methylamine as nucleophiles, attacking the chloromethyl group to form the corresponding alcohol byproduct. However, in the gas-phase toluene system described in CN102827070A, the concentration of water is negligible, effectively shutting down this competitive pathway. The reaction is exothermic, necessitating precise thermal management to prevent runaway conditions and further degradation. The patent specifies a controlled temperature window of -5°C to 5°C during the initial gas dissolution and maintains sub-zero conditions (-5°C to -2°C) during the addition of the halide. This low-temperature regime is critical for kinetic control, ensuring that the activation energy barrier for the desired substitution is met while keeping the energy available for decomposition pathways insufficient.
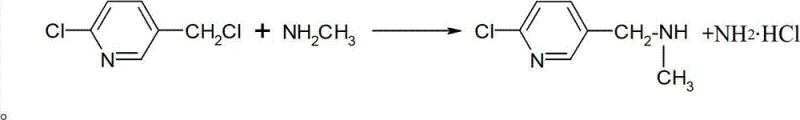
Impurity control in this mechanism is largely dictated by the stoichiometry and the phase behavior of the byproducts. As the reaction proceeds, methylamine hydrochloride is generated. In the toluene medium, this salt has limited solubility, which helps drive the equilibrium forward according to Le Chatelier's principle. The patent describes a two-stage addition process where a portion of the methylamine gas is charged first, followed by the dropwise addition of the 2-chloro-5-PMC solution, and then a second charge of gas. This staged approach ensures that the concentration of the free amine remains high relative to the halide throughout the reaction, minimizing the risk of dialkylation or polymerization side reactions. The final purity specifications are rigorously maintained by monitoring the residual 2-chloro-5-PMC content, ensuring it drops below 0.2% before the reaction is deemed complete, thus guaranteeing a clean profile for downstream pesticide synthesis.
How to Synthesize N-(6-chloro-3-picolyl)-methylamine Efficiently
Implementing this improved synthesis route requires careful attention to the sequence of reagent addition and thermal regulation to maximize safety and yield. The process begins with the preparation of the reaction vessel, where toluene is charged and cooled prior to the introduction of the hazardous methylamine gas. The subsequent dropwise addition of the pyridine derivative must be managed to control the exotherm, followed by a secondary gas charge to ensure complete conversion of the starting material. The work-up involves a straightforward aqueous wash to remove inorganic salts, followed by solvent recovery.
- Charge toluene into the reactor, cool to -5°C to 5°C, and dissolve methylamine gas.
- Dropwise add 2-chloro-5-chloromethylpyridine (2-chloro-5-PMC) toluene solution while maintaining low temperature.
- Wash the reaction mixture with water, separate layers, and distill off toluene to obtain the refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this gas-phase methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most immediate impact is seen in the reduction of raw material costs and waste disposal fees. By eliminating the transport and handling of large volumes of water inherent in aqueous amine solutions, the logistics footprint is significantly compressed. The patent explicitly notes that the switch to methylamine gas reduces transportation costs due to the higher effective concentration of the active reagent. Furthermore, the drastic reduction in wastewater generation alleviates the burden on effluent treatment plants, which is a critical factor in regions with strict environmental regulations. This leads to substantial operational cost savings, as the energy and chemicals required for wastewater neutralization and treatment are minimized.
- Cost Reduction in Manufacturing: The elimination of water from the reaction matrix directly correlates to a reduction in utility consumption. Without the need to heat or distill large quantities of water during the work-up and solvent recovery phases, steam and cooling water usage are optimized. Additionally, the higher purity of the crude product reduces the need for extensive recrystallization or chromatographic purification steps, which are often the most expensive parts of fine chemical manufacturing. The ability to load more reactant mass into the same reactor volume—improving equipment efficiency by over 30%—means that fixed costs such as labor, depreciation, and overhead are spread over a larger output, effectively lowering the cost per kilogram of the produced intermediate.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the production capacity of suppliers who rely on outdated, inefficient technologies. This novel process enhances supply reliability by increasing the throughput of existing manufacturing assets without the need for new capital expenditure on larger reactors. The simplified process flow also reduces the cycle time per batch, allowing for more flexible scheduling and faster response to market demand fluctuations. Moreover, the use of methylamine gas, a commodity chemical, ensures a stable supply of raw materials, avoiding the potential supply chain disruptions associated with specialized aqueous formulations.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste issues, but this technology is inherently greener. The reduction in pollutant discharge makes it easier for manufacturing sites to maintain compliance with evolving environmental laws, reducing the risk of regulatory shutdowns. The process is designed for industrial production, utilizing standard glass-lined steel reactors and distillation equipment, which facilitates seamless scale-up from pilot plant to commercial tonnage. The robust nature of the reaction conditions ensures consistent quality across different batch sizes, providing supply chain partners with confidence in the continuity and stability of the product supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what buyers and technical teams can expect when adopting this route. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific supply chain.
Q: Why is methylamine gas preferred over aqueous methylamine in this synthesis?
A: Using methylamine gas eliminates the introduction of water into the reaction system, which significantly reduces wastewater generation and prevents the hydrolysis of raw materials, leading to higher product purity (>97.5%) and reduced raw material consumption.
Q: What are the critical temperature controls for this reaction?
A: The reaction requires strict temperature control, initially cooling to -5°C to 5°C for gas dissolution, maintaining -5°C to -2°C during the first addition, and -2°C to 0°C during the second addition to minimize side reactions and ensure safety.
Q: How does this process impact equipment efficiency?
A: By removing water from the system, the process allows for a higher loading capacity of reactants in the same reactor volume, improving equipment utilization efficiency by over 30% compared to traditional aqueous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(6-chloro-3-picolyl)-methylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final pesticide depends heavily on the purity of its precursors. Our technical team has extensively analyzed the process described in CN102827070A and possesses the expertise to execute this gas-phase methylation with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this laboratory-proven method are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of N-(6-chloro-3-picolyl)-methylamine meets the high standards required for neonicotinoid synthesis.
We invite you to collaborate with us to optimize your supply chain for agrochemical intermediates. By leveraging our technical capabilities, you can achieve significant cost reduction in pesticide intermediate manufacturing while securing a sustainable supply source. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can support your long-term business goals.
