Advanced Manufacturing of 1-Cyclopropylnaphthalene: A Strategic Upgrade for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks efficient pathways to access complex heterocyclic and carbocyclic scaffolds essential for next-generation therapeutics. Patent CN103664471A introduces a transformative methodology for the synthesis of 1-cyclopropylnaphthalene, a critical building block in the manufacture of novel allylamine antifungal agents. This innovation addresses the longstanding economic and technical bottlenecks associated with producing cyclopropyl-substituted aromatic compounds. By leveraging a modified Simmons-Smith type cyclopropanation using organomagnesium reagents, the disclosed process converts inexpensive 1-vinylnaphthalene directly into the target molecule with exceptional efficiency. For R&D directors and procurement strategists, this represents a pivotal shift away from reliance on precious metal catalysis and high-cost halogenated starting materials, paving the way for more sustainable and cost-effective supply chains in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-cyclopropylnaphthalene has been constrained by two primary methodologies, both of which present significant drawbacks for large-scale manufacturing. The first approach typically involves a Suzuki-Miyaura cross-coupling reaction between 1-bromonaphthalene and cyclopropylboronic acid. While chemically effective, this route suffers from the prohibitive cost of boronic acid derivatives and the necessity for expensive palladium catalysts, which require rigorous and costly removal steps to meet pharmaceutical purity standards. The second conventional method utilizes naphthalene derivatives coupled with dihalomethanes under the catalysis of rare-earth metals like lanthanum. This technique is equally problematic due to the scarcity and high price of rare-earth catalysts, alongside the operational complexities associated with handling air-sensitive lanthanide species. These factors collectively inflate the production cost and complicate the supply chain reliability for this vital intermediate.
The Novel Approach
In stark contrast, the methodology outlined in CN103664471A offers a streamlined, one-pot solution that circumvents these traditional hurdles. By employing 1-vinylnaphthalene—a commodity chemical with significantly lower market pricing than its brominated counterparts—as the starting substrate, the process fundamentally alters the cost structure of the synthesis. The reaction utilizes dihalomethane and tert-butylmagnesium chloride to generate a reactive carbenoid species in situ, which subsequently cyclopropanates the vinyl double bond. This approach not only eliminates the need for transition metal catalysts entirely but also operates under mild thermal conditions, specifically utilizing an ice bath to control exothermicity. The result is a robust, operationally simple protocol that yields high-purity product after a straightforward recrystallization, making it ideally suited for industrial adoption.
Mechanistic Insights into Organomagnesium-Mediated Cyclopropanation
The core of this technological breakthrough lies in the generation and reactivity of the organomagnesium carbenoid intermediate. Unlike the classic zinc-based Simmons-Smith reaction, this patent utilizes a Grignard reagent, specifically tert-butylmagnesium chloride, to activate the dihalomethane. Upon mixing the dihalomethane with the Grignard reagent, a halogen-metal exchange occurs, generating a highly electrophilic methylene transfer agent. This species then coordinates with the electron-rich double bond of the 1-vinylnaphthalene substrate. The reaction proceeds through a concerted mechanism where the methylene group is transferred to the alkene, forming the strained three-membered cyclopropane ring while retaining the stereochemical integrity of the substituents. This mechanistic pathway is highly advantageous as it avoids the formation of radical intermediates that often lead to polymerization or side reactions in vinyl systems.
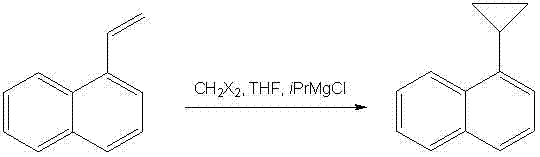
Furthermore, the control of impurities in this system is inherently superior due to the specificity of the carbenoid attack. In traditional cross-coupling methods, homocoupling of the aryl halide or protodeboronation of the boronic acid can generate difficult-to-remove impurities that compromise the quality of the final API. In this organomagnesium-mediated process, the primary side reactions are limited to the decomposition of excess Grignard reagent upon quenching, which forms volatile hydrocarbons that are easily removed during workup. The use of an ice bath ensures that the reaction temperature remains low enough to prevent thermal degradation of the sensitive cyclopropane ring or the polymerization of the vinyl starting material. Consequently, the crude product obtained prior to recrystallization already possesses high chemical purity, simplifying downstream processing and reducing solvent consumption.
How to Synthesize 1-Cyclopropylnaphthalene Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and addition rates to maximize yield and safety. The process begins with the preparation of the Grignard solution under an inert atmosphere, followed by the controlled addition of the vinyl substrate to manage heat evolution. Subsequent addition of the dihalomethane triggers the cyclopropanation event.
- Under nitrogen protection and ice bath cooling, slowly add 1-vinylnaphthalene to a solution of tert-butylmagnesium chloride (3M) maintaining a molar ratio between 1: 0.3 and 1:2.
- Upon completion of addition, introduce dihalomethane (1-6 mol) and additional tert-butylmagnesium chloride (0.3-2 mol) to the reaction mixture.
- Stir the reaction for 1-6 hours, quench with saturated ammonium chloride, extract with ethyl acetate, and purify the crude product via toluene recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple unit cost savings. The shift from specialized, high-cost reagents to commodity chemicals creates a more resilient supply chain less susceptible to market volatility. By removing the dependency on palladium and rare-earth metals, manufacturers eliminate the risks associated with the geopolitical supply constraints of these critical minerals. Additionally, the simplified workup procedure, which avoids complex chromatographic purifications or heavy metal scavenging steps, translates directly into reduced manufacturing cycle times and lower waste disposal costs. This efficiency gain allows for faster response to market demand fluctuations and improved overall asset utilization within the production facility.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic reduction in raw material expenses. Replacing 1-bromonaphthalene and cyclopropylboronic acid with 1-vinylnaphthalene and dihalomethane leverages the economies of scale associated with bulk petrochemical derivatives. Furthermore, the elimination of precious metal catalysts removes a significant line item from the bill of materials, while also saving on the costs of catalyst recovery or disposal. The high yield reported in the patent examples implies that less starting material is wasted, further enhancing the overall process mass intensity and contributing to substantial cost savings in the final product pricing.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical intermediates, and this process secures the supply chain by utilizing widely available reagents. 1-Vinylnaphthalene and dihalomethanes are produced by multiple global suppliers, reducing the risk of single-source bottlenecks. In contrast, specialized boronic acids or rare-earth catalysts often have limited supplier bases, making them vulnerable to disruptions. The robustness of the reaction conditions, which do not require ultra-low temperatures or high-pressure equipment, also means that the process can be easily transferred between different manufacturing sites without significant requalification efforts, ensuring consistent supply availability.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with modern green chemistry principles. The absence of toxic heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with metal residue limits in pharmaceutical products. The use of standard solvents like THF and ethyl acetate facilitates solvent recovery and recycling, minimizing the environmental footprint of the manufacturing process. The mild reaction conditions reduce energy consumption compared to high-temperature reflux processes, and the one-pot nature of the synthesis minimizes the number of unit operations, thereby reducing the potential for safety incidents and improving the overall scalability of the production line.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding the feasibility, quality, and operational aspects of the patented method.
Q: What are the primary advantages of this synthesis method over traditional Suzuki coupling?
A: Unlike traditional methods requiring expensive 1-bromonaphthalene and cyclopropylboronic acid with palladium catalysts, this patent utilizes low-cost 1-vinylnaphthalene and eliminates the need for precious metal catalysts, drastically reducing raw material and purification costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (ice bath) and uses common solvents like THF and ethyl acetate. The one-pot nature and absence of sensitive rare-earth catalysts make it highly robust for commercial scale-up.
Q: What purity levels can be achieved with this method?
A: According to the patent examples, the method consistently achieves high purity levels exceeding 99% (HPLC) after simple toluene recrystallization, meeting stringent requirements for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Cyclopropylnaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antifungal medications. Our technical team has thoroughly analyzed the pathway described in CN103664471A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to fruition. We are committed to delivering 1-cyclopropylnaphthalene with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the exacting standards demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions of this organomagnesium-mediated synthesis safely and efficiently.
We invite potential partners to engage with us to explore how this optimized synthetic route can enhance your supply chain resilience and cost structure. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development pipeline.
