Advanced Palladium-Catalyzed Synthesis of Chiral 3-Substituted Indolines for Commercial Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of Chiral 3-Substituted Indolines for Commercial Pharmaceutical Applications
The landscape of chiral heterocycle synthesis is undergoing a significant transformation driven by the demand for more efficient and environmentally benign processes. A pivotal development in this field is documented in Chinese Patent CN115991667A, which discloses a robust method for the palladium-catalyzed asymmetric hydrogenation of 3-substituted unprotected indoles. This technology addresses a longstanding challenge in organic synthesis: the direct conversion of readily available indole precursors into high-value chiral 3-substituted indoline derivatives without the need for cumbersome protecting group strategies. By leveraging a specialized palladium-chiral bisphosphine complex in conjunction with a Brønsted acid promoter, this methodology achieves exceptional enantioselectivity and yield under relatively mild reaction conditions. For R&D directors and process chemists, this represents a critical advancement, as it streamlines the production of key scaffolds found in bioactive molecules such as pentopril and various kinase inhibitors. The ability to access these optically pure compounds directly from unprotected starting materials not only simplifies the synthetic route but also aligns with the principles of green chemistry by reducing waste and energy consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric synthesis of chiral indolines has been fraught with significant technical hurdles that impact both cost and scalability. Traditional approaches often relied heavily on chemical resolution or enzymatic splitting, methods that are inherently inefficient due to a maximum theoretical yield of 50% for the desired enantiomer. Furthermore, earlier catalytic hydrogenation strategies, such as those developed by the Kuwano group using Rhodium/Ph-TRAP systems, typically required the indole nitrogen to be protected with groups like Ac, Ts, or Boc. While effective, this protection-deprotection sequence adds at least two additional synthetic steps, increasing material costs, solvent usage, and processing time. Other attempts using Iridium or Ruthenium catalysts on unprotected substrates have struggled with catalyst poisoning caused by the free nitrogen lone pair, resulting in poor conversion rates or negligible enantioselectivity. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, while supply chain heads face risks associated with complex multi-step processes that are difficult to scale reliably.
The Novel Approach
The methodology presented in patent CN115991667A offers a paradigm shift by enabling the direct asymmetric hydrogenation of 3-substituted unprotected indoles. This novel approach utilizes a palladium precursor, such as palladium trifluoroacetate, coordinated with sophisticated chiral bisphosphine ligands like WingPhos derivatives. Crucially, the system incorporates a Brønsted acid promoter, such as L-camphorsulfonic acid (L-CSA), which plays a dual role: it protonates the indole nitrogen to prevent catalyst deactivation and facilitates a dynamic kinetic resolution process. This allows for the conversion of racemic or prochiral starting materials into single enantiomers with high fidelity. The reaction proceeds efficiently in a mixed solvent system of trifluoroethanol and benzene at moderate temperatures (60°C) and hydrogen pressures (700 psi). For a reliable pharmaceutical intermediate supplier, this translates to a streamlined process that eliminates the need for protecting group manipulation, thereby significantly reducing the overall step count and improving the cost reduction in API manufacturing. The operational simplicity and the use of commercially available catalyst components make this technology highly attractive for immediate industrial adoption.
Mechanistic Insights into Acid-Promoted Pd-Catalyzed Asymmetric Hydrogenation
At the heart of this technological breakthrough lies a sophisticated interplay between the palladium catalyst and the acid promoter. The free nitrogen atom in 3-substituted indoles possesses a lone pair of electrons that acts as a strong Lewis base, which traditionally binds irreversibly to transition metal centers, rendering the catalyst inactive. In this patented system, the Brønsted acid serves as a protective agent by protonating the nitrogen atom in situ, effectively masking its basicity while maintaining the aromatic integrity required for the reaction. The chiral environment is established by the bulky bisphosphine ligand, which creates a specific steric pocket around the palladium center. As the hydrogen molecule is activated on the metal surface, the substrate approaches in a highly oriented manner dictated by the chiral ligand and the ion-pairing interaction with the sulfonate anion of the acid promoter. This precise spatial arrangement ensures that hydrogen addition occurs exclusively from one face of the double bond, leading to the observed high enantiomeric excess. Understanding this mechanism is vital for R&D teams aiming to optimize the process further, as it highlights the importance of acid strength and ligand sterics in controlling the stereochemical outcome.
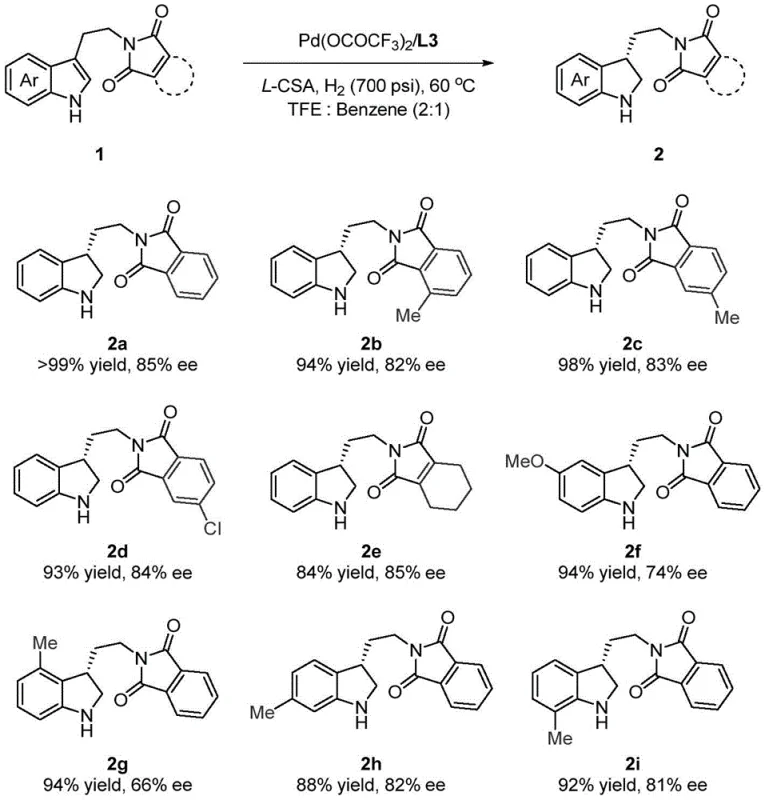
Impurity control is another critical aspect where this mechanism excels, particularly concerning the suppression of over-reduction or isomerization byproducts. The dynamic kinetic resolution component of the reaction ensures that even if the starting material undergoes rapid tautomerization or racemization under the acidic conditions, the catalyst selectively converts the favorable conformer into the desired chiral product. This self-correcting feature minimizes the formation of diastereomers and other structural impurities that are notoriously difficult to separate during downstream purification. The patent data indicates that by fine-tuning the ratio of acid to substrate (optimally 1:1.2), the reaction achieves complete conversion with minimal side reactions. For quality control laboratories, this means the crude product profile is much cleaner, reducing the burden on chromatographic purification steps. The ability to consistently achieve enantiomeric excess values up to 89% across a diverse range of substrates demonstrates the robustness of this catalytic cycle, providing a reliable foundation for the production of high-purity pharmaceutical intermediates that meet stringent regulatory standards.
How to Synthesize 3-Substituted Indolines Efficiently
Implementing this synthesis route requires careful attention to the preparation of the active catalyst species and the maintenance of an inert atmosphere to prevent oxidation of the phosphine ligands. The process begins with the pre-formation of the palladium-ligand complex in a volatile solvent like acetone, which is subsequently removed to ensure the active species is introduced directly into the reaction matrix without dilution effects. The reaction is then conducted in a high-pressure autoclave capable of sustaining hydrogen pressures up to 700 psi, ensuring sufficient driving force for the reduction of the stable aromatic indole ring. Detailed standardized operating procedures regarding the order of addition, specifically mixing the substrate and acid prior to catalyst introduction, are essential to maximize reproducibility. The detailed standardized synthesis steps are outlined below for technical reference.
- Prepare the catalyst by stirring palladium trifluoroacetate and a chiral bisphosphine ligand (e.g., L3) in acetone under nitrogen, followed by vacuum concentration.
- In a glove box, mix the 3-substituted indole substrate and a Brønsted acid promoter (e.g., L-CSA) in a TFE/Benzene solvent mixture.
- Add the catalyst solution, transfer to a high-pressure reactor, and hydrogenate at 60°C under 700 psi H2 pressure for 24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed hydrogenation technology offers substantial strategic benefits for organizations focused on cost efficiency and supply chain resilience. The elimination of nitrogen protecting groups removes entire unit operations from the manufacturing workflow, including the reagents and solvents required for protection and the subsequent harsh conditions needed for deprotection. This simplification directly correlates to a significant reduction in raw material consumption and waste generation, aligning with modern sustainability goals. Furthermore, the use of earth-abundant palladium compared to more exotic metals like iridium or rhodium can lead to lower catalyst costs, although the specific ligand cost must be evaluated. For procurement managers, this translates into a more predictable cost structure and reduced exposure to price volatility associated with rare metal catalysts. The mild reaction conditions also imply lower energy requirements for heating and cooling, contributing to overall operational expenditure savings.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic shortening of the synthetic sequence. By bypassing the protection-deprotection steps, manufacturers save on reagents, solvents, and labor hours associated with these additional transformations. Additionally, the high atom economy of the hydrogenation reaction means that the majority of the starting material mass is incorporated into the final product, minimizing waste disposal costs. The ability to use commercially available catalyst precursors further stabilizes the supply chain against shortages of specialized reagents. Qualitative analysis suggests that the simplified workflow allows for faster batch turnover, effectively increasing plant capacity without the need for capital investment in new equipment.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply continuity. The tolerance for various functional groups, including esters and amides, means that a single catalytic platform can be used to produce a wide library of intermediates, reducing the need for multiple dedicated production lines. The starting materials, 3-substituted indoles, are generally readily available or easily synthesized from commodity chemicals, ensuring a stable upstream supply. This flexibility allows supply chain heads to respond quickly to changes in demand for different API precursors. Moreover, the high selectivity of the reaction reduces the risk of batch failures due to impurity profiles, ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard high-pressure hydrogenation equipment commonly found in fine chemical plants. The solvent system, while requiring careful handling due to benzene content, can potentially be optimized or substituted with greener alternatives in future iterations, though the current TFE/Benzene mix is effective. The reduction in chemical waste steps inherently lowers the environmental footprint of the manufacturing process, aiding in compliance with increasingly strict environmental regulations. The low energy input required for the 60°C reaction temperature further supports sustainability initiatives, making this technology a viable candidate for green chemistry certification and long-term sustainable manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for decision-makers evaluating this process for integration into their production pipelines. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific target molecules.
Q: Why is acid promotion necessary for the hydrogenation of unprotected indoles?
A: The nitrogen atom in unprotected indoles can coordinate strongly with the metal center, poisoning the catalyst. The Brønsted acid promoter protonates the nitrogen, preventing deactivation and enabling dynamic kinetic resolution for high enantioselectivity.
Q: What represents the primary advantage of this method over Rhodium or Iridium systems?
A: This palladium-based system operates under milder conditions with commercially available ligands and avoids the need for N-protecting groups, thereby eliminating tedious deprotection steps and improving overall atom economy.
Q: Can this process tolerate sensitive functional groups like esters and amides?
A: Yes, the patent data demonstrates excellent functional group tolerance, successfully hydrogenating substrates containing ester, amide, and halogen substituents with high yields and enantiomeric excess values up to 89%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Indolines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the palladium-catalyzed asymmetric hydrogenation described in CN115991667A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to multi-ton manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR instrumentation to guarantee that every batch of 3-substituted indolines meets the highest quality standards required by global pharmaceutical regulators.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this unprotected indole route or need specific COA data to verify the enantiomeric purity of our pilot batches, we are ready to assist. Our commitment to innovation and quality makes us the ideal partner for the commercial scale-up of complex pharmaceutical intermediates. Contact us today to request route feasibility assessments and discover how we can support your supply chain with high-performance chiral building blocks.
