Advanced Metal-Free Synthesis of Bromobenzene Para-Amination Compounds for Commercial Scale-Up
Advanced Metal-Free Synthesis of Bromobenzene Para-Amination Compounds for Commercial Scale-Up
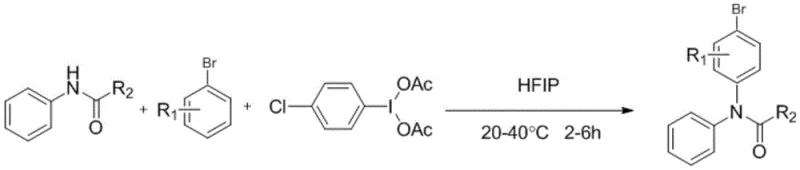
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. Patent CN113336665B introduces a groundbreaking preparation method for bromobenzene para-amination compounds, utilizing a hypervalent iodine reagent to mediate the critical C-N bond formation. This technology represents a paradigm shift away from traditional transition-metal catalysis, offering a robust pathway for synthesizing complex arylamide structures that are pivotal in the development of active pharmaceutical ingredients (APIs) and agrochemical agents. By leveraging the unique oxidative properties of p-chloroiodobenzenediethyl ester in a hexafluoroisopropanol (HFIP) medium, this process achieves high regioselectivity and yield under exceptionally mild thermal conditions. For R&D directors and procurement strategists, this innovation signals a new era of cost-effective and environmentally compliant manufacturing for high-value nitrogen-containing heterocycles and intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-aryl amide scaffolds has heavily relied on transition-metal catalyzed cross-coupling reactions, such as the ubiquitous Suzuki-Miyaura or Buchwald-Hartwig protocols. While effective, these conventional methods impose severe constraints on industrial scalability and product purity profiles. The reliance on palladium or copper catalysts introduces significant downstream processing challenges, particularly the rigorous removal of trace heavy metals to meet stringent pharmacopeial limits for drug substances. Furthermore, these catalytic systems often require expensive ligands, inert atmospheres, and elevated temperatures, which collectively drive up the operational expenditure (OpEx) and carbon footprint of the manufacturing process. The generation of metal-contaminated waste streams also complicates environmental compliance, necessitating costly waste treatment protocols that erode profit margins in high-volume production scenarios.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113336665B circumvents these metallurgical bottlenecks by employing a metal-free hypervalent iodine mediation strategy. This novel approach utilizes p-chloroiodobenzenediethyl ester as a potent oxidant to activate the amide nitrogen, facilitating direct para-selective amination of the bromobenzene ring. The reaction proceeds smoothly in hexafluoroisopropanol (HFIP), a solvent known for its ability to stabilize cationic intermediates through strong hydrogen bonding networks. This synergy allows the transformation to occur at ambient to slightly elevated temperatures ranging from 20°C to 40°C, drastically reducing energy consumption compared to traditional reflux conditions. The result is a streamlined synthetic route that delivers high-purity arylamide products with excellent yields, effectively bypassing the need for complex metal scavenging steps and enabling a more sustainable manufacturing lifecycle.
Mechanistic Insights into Hypervalent Iodine Mediated C-H Amination
The core of this technological advancement lies in the unique reactivity of the hypervalent iodine(III) species, which acts as both an oxidant and a transient directing group. In the presence of HFIP, the p-chloroiodobenzenediethyl ester undergoes ligand exchange to generate a highly electrophilic iodine center capable of activating the nucleophilic nitrogen of the substituted acetanilide. This activation creates an N-iodo intermediate that serves as the active aminating agent. The electron-rich bromobenzene ring then attacks this activated nitrogen species in an electrophilic aromatic substitution mechanism. The specific solvation environment provided by HFIP is critical, as it stabilizes the developing positive charge in the transition state, thereby lowering the activation energy barrier and ensuring rapid reaction kinetics within the 2-6 hour window specified in the patent data.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. The concerted nature of the electrophilic substitution minimizes the formation of homocoupling byproducts or over-oxidized species that often plague radical chemistry. Moreover, the absence of transition metals eliminates the risk of metal-induced side reactions, such as dehalogenation of the bromobenzene substrate, which is a common issue in palladium-catalyzed systems. The high regioselectivity for the para-position is governed by the steric and electronic properties of the bromobenzene substrate, ensuring that the resulting N-(4-bromophenyl)-N-phenylacetamide derivatives are formed with minimal isomeric contamination. This inherent selectivity simplifies the purification workflow, often allowing for high-purity isolation via standard crystallization or chromatography techniques without extensive recycling of mother liquors.
How to Synthesize N-(4-Bromophenyl)-N-phenylacetamide Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing sequences to maximize yield and safety. The process begins with the sequential addition of substituted acetanilide, the hypervalent iodine reagent, and the bromobenzene substrate into a reactor containing hexafluoroisopropanol. Maintaining the reaction temperature strictly between 20°C and 40°C is crucial to prevent decomposition of the hypervalent iodine species while ensuring sufficient kinetic energy for the transformation. Following the reaction period of 2-6 hours, the mixture is quenched with water, and the product is extracted using organic solvents like ethyl acetate. Detailed standardized operating procedures for scaling this route from gram to kilogram quantities are outlined in the technical guide below.
- Charge the reactor with substituted acetanilide, p-chloroiodobenzenediethyl ester, bromobenzene, and hexafluoroisopropanol (HFIP) in the specified molar ratios.
- Maintain the reaction mixture at a mild temperature range of 20°C to 40°C with continuous stirring for a duration of 2 to 6 hours.
- Quench the reaction with water, extract the organic phase with ethyl acetate, and purify the crude product via column chromatography to obtain the target arylamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hypervalent iodine-mediated route presents a compelling value proposition centered on cost optimization and supply security. By eliminating the dependency on precious metal catalysts like palladium, manufacturers can insulate their production costs from the volatile fluctuations of the global precious metals market. The simplified workup procedure, which avoids the need for specialized metal scavenger resins or complex filtration steps, translates directly into reduced cycle times and lower utility costs per kilogram of product. Furthermore, the use of commercially available and stable starting materials ensures a robust supply chain that is less susceptible to disruptions compared to routes relying on sensitive organometallic reagents or air-sensitive catalysts.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of transition metal catalysts from the process flow. Traditional methods incur substantial costs not only for the purchase of palladium or copper salts but also for the downstream removal and disposal of these toxic metals to meet regulatory standards. By switching to a hypervalent iodine system, facilities can eliminate the capital expenditure associated with metal recovery units and reduce the recurring cost of scavenger materials. Additionally, the mild reaction temperatures (20-40°C) significantly lower steam and cooling requirements, contributing to a leaner energy profile and substantial overall cost savings in large-scale API manufacturing.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various substituted acetanilides and bromobenzene derivatives, are commodity chemicals with well-established global supply chains. Unlike specialized ligands or unstable organoboron reagents used in cross-coupling, these substrates are readily available from multiple vendors, reducing the risk of single-source bottlenecks. The stability of the hypervalent iodine reagent also allows for bulk purchasing and storage without the need for cryogenic conditions, further enhancing inventory management flexibility and ensuring consistent production schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology mitigates those concerns through its inherent design. The reaction operates at near-ambient temperatures and pressures, removing the hazards associated with high-pressure hydrogenation or exothermic metal-catalyzed couplings. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and reduces the toxicity of solid waste streams. This aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations, facilitating smoother permitting processes for new manufacturing lines and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hypervalent iodine-mediated synthesis. These insights are derived directly from the experimental data and beneficial effects described in patent CN113336665B, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: How does this hypervalent iodine method compare to traditional Suzuki coupling for arylamide synthesis?
A: Unlike traditional Suzuki coupling which relies on expensive and toxic palladium or copper catalysts, this method utilizes a hypervalent iodine reagent. This eliminates the risk of heavy metal contamination in the final API, significantly simplifying the purification process and reducing the environmental burden associated with metal waste disposal.
Q: What are the specific reaction conditions required for this para-amination process?
A: The process operates under remarkably mild conditions, requiring temperatures between 20°C and 40°C. The reaction utilizes hexafluoroisopropanol (HFIP) as a solvent, which facilitates the activation of the hypervalent iodine species, allowing the transformation to proceed efficiently within 2 to 6 hours without the need for extreme heat or pressure.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its operational simplicity and safety profile. The absence of pyrophoric catalysts and the use of moderate temperatures make it suitable for large-scale batch reactors. Furthermore, the workup procedure involving standard aqueous quenching and organic extraction is easily adaptable to industrial separation units.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(4-Bromophenyl)-N-phenylacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic technologies in modernizing the pharmaceutical supply chain. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113336665B are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of arylamide intermediate meets the exacting standards required for downstream drug substance synthesis. We are committed to delivering high-quality chemical solutions that empower our partners to bring life-saving therapies to market faster and more efficiently.
We invite forward-thinking R&D and procurement teams to collaborate with us on optimizing their synthetic routes for arylamide production. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your specific project needs, identifying opportunities to reduce material costs and improve process efficiency. We encourage you to contact our technical procurement team today to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your proprietary molecules. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing.
