Advanced Silver-Catalyzed Alpha-Position C-C Bond Formation for Pharmaceutical Intermediates Manufacturing
The landscape of organic synthesis is constantly evolving towards greener, more atom-economical processes, particularly in the construction of carbon-carbon bonds which form the backbone of complex molecular architectures. A significant breakthrough in this domain is detailed in Chinese Patent CN111606894A, which discloses a novel silver-catalyzed method for forming carbon-carbon bonds at the α-position of ether compounds. This technology represents a paradigm shift from traditional pre-functionalization strategies to direct C-H activation, specifically utilizing a Cross-Dehydrogenative Coupling (CDC) approach. By employing silver trifluoroacetate (AgOCOCF3) as a catalyst and potassium persulfate (K2S2O8) as an oxidant in an aqueous medium, this invention enables the direct coupling of substituted quinolines or pyridines with cyclic ethers. For R&D directors and process chemists seeking reliable pharmaceutical intermediates supplier partners, this methodology offers a compelling alternative to existing technologies, promising high selectivity and operational simplicity while adhering to stringent environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of ether α-positions has been fraught with significant technical and economic challenges that hinder efficient manufacturing. Prior art, such as the work by the MacMillan group in 2015, relied heavily on photoredox catalysis using expensive Iridium complexes activated by visible light sources. While effective, these systems introduce substantial cost burdens due to the precious metal catalyst and require specialized photo-reactors that complicate scale-up efforts. Furthermore, other conventional approaches, like those reported by Singh's group, necessitate harsh reaction conditions, often requiring temperatures as high as 120°C to drive the activation of inert C-H bonds. These high-energy processes not only increase utility costs but also pose safety risks related to thermal runaway and pressure management in large-scale reactors. Additionally, many traditional methods suffer from poor atom economy, generating excessive waste streams that require complex downstream processing to meet the purity specifications demanded by the fine chemical industry.
The Novel Approach
In stark contrast to these legacy methods, the silver-catalyzed protocol described in CN111606894A introduces a remarkably mild and economically viable pathway for ether functionalization. The core innovation lies in the ability to activate the α-C-H bond of ethers at ambient temperature (25°C), effectively eliminating the need for energy-intensive heating or complex photonic equipment. By utilizing a simple combination of AgOCOCF3 and K2S2O8 in a water-containing solvent system, the reaction achieves high conversion rates with excellent regioselectivity. This approach drastically simplifies the reaction setup, allowing standard glass-lined or stainless steel reactors to be used without modification. The use of water as a co-solvent further enhances the green chemistry profile of the process, reducing the reliance on volatile organic solvents and facilitating easier product isolation. For procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing, this transition from precious metal photocatalysts to abundant silver salts represents a tangible opportunity to lower raw material expenditures.
Mechanistic Insights into Silver-Catalyzed Cross-Dehydrogenative Coupling
The mechanistic underpinning of this transformation involves a sophisticated radical generation and propagation cycle driven by the silver catalyst and the persulfate oxidant. Initially, the silver(I) species interacts with the persulfate anion to generate highly reactive sulfate radical anions through a single-electron transfer process. These sulfate radicals are potent hydrogen abstractors capable of selectively removing a hydrogen atom from the α-position of the cyclic ether, thereby generating a nucleophilic α-oxy radical intermediate. This radical species subsequently attacks the electron-deficient heteroaromatic ring of the quinoline or pyridine substrate. The resulting radical adduct undergoes oxidation and deprotonation to restore aromaticity, yielding the final C-C coupled product. The silver catalyst plays a dual role, potentially mediating the initial radical generation and assisting in the re-oxidation steps to close the catalytic cycle. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining precise stoichiometric ratios between the oxidant and the substrate to prevent over-oxidation or catalyst deactivation.
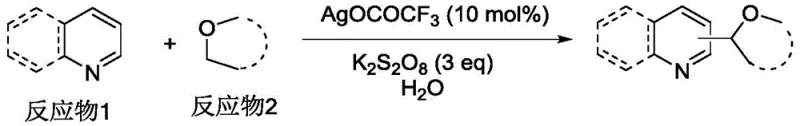
From an impurity control perspective, the high selectivity of this silver-mediated radical pathway is a distinct advantage for producing high-purity OLED material or API precursors. Unlike acid-catalyzed methods that might promote ether cleavage or polymerization side reactions, the radical mechanism is highly specific to the α-C-H bond. The mild reaction temperature further suppresses thermal degradation pathways that often lead to tar formation or complex byproduct mixtures in high-temperature processes. Consequently, the crude reaction profile is typically cleaner, reducing the burden on purification units such as distillation columns or chromatography systems. This inherent selectivity ensures that the final product meets the rigorous quality standards required for downstream applications in drug synthesis and agrochemical production, minimizing the risk of toxic impurities carrying through to the final active ingredient.
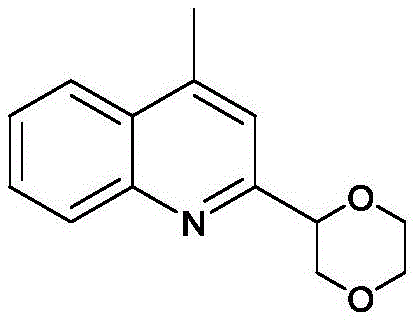
How to Synthesize 4-Methyl-2-(2-1,4-dioxanyl)quinoline Efficiently
To implement this technology in a laboratory or pilot plant setting, operators must adhere to a standardized protocol that maximizes yield while ensuring safety. The process begins with the sequential addition of the heterocyclic substrate, the silver catalyst, and the oxidant into a reactor containing the cyclic ether and water. Maintaining the reaction at a constant 25°C with efficient magnetic stirring is critical to ensure homogeneous mixing and consistent heat transfer throughout the 4 to 10-hour reaction window. Following the reaction completion, a straightforward workup procedure involving aqueous quenching and organic extraction allows for the recovery of the product. Detailed standard operating procedures regarding exact molar ratios, stirring speeds, and purification parameters are essential for reproducibility.
- Charge the reactor with substituted quinoline or pyridine, AgOCOCF3 catalyst (10 mol%), K2S2O8 oxidant (3 eq), cyclic ether, and water.
- Maintain the reaction mixture at 25°C in an oil bath with magnetic stirring and reflux condensation for 4-10 hours.
- Extract the reaction mixture with dichloromethane, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this silver-catalyzed technology offers profound strategic benefits that extend beyond mere technical feasibility. The shift away from exotic photocatalysts and high-energy processes directly translates into a more resilient and cost-effective supply chain. By simplifying the reaction conditions and utilizing widely available reagents, manufacturers can mitigate risks associated with raw material scarcity and price volatility. Furthermore, the environmental compatibility of the process aligns with increasingly strict global regulations on industrial emissions and waste disposal, reducing the compliance burden on manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of expensive Iridium-based photocatalysts and the removal of specialized lighting equipment result in significant capital expenditure savings. Additionally, operating at ambient temperature drastically reduces energy consumption compared to traditional high-temperature methods, leading to lower utility costs per kilogram of product. The simplified workup procedure also reduces solvent usage and processing time, contributing to overall operational efficiency and improved profit margins for high-value intermediates.
- Enhanced Supply Chain Reliability: The reagents required for this process, including silver trifluoroacetate and potassium persulfate, are commodity chemicals with robust global supply chains, unlike niche photocatalysts that may face availability bottlenecks. This accessibility ensures consistent production schedules and reduces the lead time for high-purity pharmaceutical intermediates. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies, enhancing overall plant reliability and uptime.
- Scalability and Environmental Compliance: The use of water as a co-solvent and the absence of hazardous high-pressure or high-temperature conditions make this process inherently safer and easier to scale from gram to ton quantities. The reduced generation of hazardous waste and the potential for solvent recycling align with green chemistry principles, facilitating easier permitting and environmental compliance. This scalability ensures that the technology can meet the growing demand for complex polymer additives and electronic chemicals without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed ether functionalization technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the advantages of using AgOCOCF3 over Iridium photocatalysts?
A: Unlike expensive Iridium-based photocatalysts that require specialized light sources, the silver trifluoroacetate system operates under thermal conditions at ambient temperature, significantly reducing equipment costs and catalyst loading expenses.
Q: Can this method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes water as a co-solvent and operates at mild temperatures (25°C), eliminating the need for extreme heating or cryogenic cooling, which facilitates safer and more energy-efficient scale-up.
Q: What is the substrate scope for this silver-catalyzed reaction?
A: The methodology demonstrates high versatility, successfully functionalizing various substituted quinolines and pyridines with cyclic ethers such as 1,4-dioxane and tetrahydrofuran, yielding products with high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-2-(2-1,4-dioxanyl)quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the silver-mediated C-H activation described in CN111606894A. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle sensitive silver-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We understand that consistency and quality are paramount for our clients in the pharmaceutical and agrochemical sectors, and our dedicated technical team is committed to delivering excellence in every shipment.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets, helping you identify the most efficient route to market. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline and optimize your manufacturing costs.
