Revolutionizing Benzopyran Production: A Base-Catalyzed Route for Commercial Scale-Up
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign methodologies. A significant breakthrough in this domain is documented in Chinese patent CN115286608A, which introduces a novel preparation method for benzopyran compounds. These structures, also known as chromans, are pivotal scaffolds in medicinal chemistry, forming the core of biologically active natural products such as Vitamin E, various coumarins, and flavonoid derivatives. The patent details a robust synthetic strategy that utilizes alpha,beta-unsaturated carbonyl compounds and naphthol derivatives as substrates. Unlike conventional approaches that rely heavily on acidic conditions, this invention leverages a basic catalyst, specifically highlighting the efficacy of potassium carbonate, to drive an oxa-Michael addition and cyclization tandem reaction. This shift in catalytic paradigm not only simplifies the operational procedure but also dramatically improves the tolerance for sensitive functional groups, addressing a long-standing pain point in the synthesis of complex pharmaceutical intermediates.
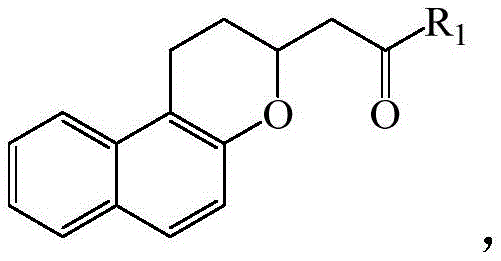
The implications of this technology extend far beyond the laboratory bench. For global supply chains, the ability to synthesize these high-value intermediates using cheap, abundant inorganic bases rather than scarce or toxic Lewis acids represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the mild reaction conditions reported, typically ranging from 60°C to 100°C, suggest a process that is inherently safer and easier to control on a commercial scale, reducing the energy burden and safety risks associated with high-temperature or high-pressure reactors. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative pathways is crucial for maintaining competitiveness and ensuring the continuous availability of critical building blocks for the global healthcare industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzopyran skeleton has been predominantly achieved through the reaction of phenols with alkenes catalyzed by Lewis acids. While effective in simple systems, this traditional methodology suffers from significant drawbacks when applied to complex molecule synthesis. The primary limitation lies in the incompatibility of strong acidic conditions with acid-sensitive functional groups. In the context of modern drug discovery, target molecules frequently possess reactive moieties such as free hydroxyl groups, amines, or other base-labile protecting groups like acetals and tert-butoxycarbonyl (Boc) groups. Exposure to the harsh acidic environments required by traditional Lewis acid catalysts often leads to the decomposition of these sensitive groups or the cleavage of essential protecting groups. Consequently, chemists are forced to employ cumbersome protection-deprotection sequences, which add multiple steps to the synthesis, reduce overall atom economy, and significantly inflate production costs. Additionally, many traditional catalysts involve heavy metals or expensive organometallic complexes, posing challenges for waste disposal and residual metal control in final API products.
The Novel Approach
The methodology disclosed in patent CN115286608A offers a transformative solution by shifting the catalytic regime from acidic to basic. By utilizing alpha,beta-unsaturated carbonyl compounds and naphthol derivatives in the presence of a basic catalyst, the reaction proceeds through a concerted oxa-Michael addition followed by intramolecular cyclization. This base-mediated pathway operates under remarkably mild conditions, effectively bypassing the degradation issues associated with acid catalysis. The use of inexpensive inorganic bases like potassium carbonate eliminates the need for costly transition metal catalysts, thereby simplifying the purification process and reducing the environmental footprint. Moreover, the terminal carbonyl group remaining on the product molecule provides a versatile handle for further structural diversification, allowing for the rapid generation of derivative libraries for biological screening. This approach not only enhances the synthetic efficiency but also broadens the scope of accessible chemical space, enabling the production of benzopyran derivatives that were previously difficult or impossible to synthesize using conventional acid-catalyzed routes.
Mechanistic Insights into Base-Catalyzed Oxa-Michael Addition and Cyclization
The core of this innovation lies in the mechanistic pathway facilitated by the basic catalyst. The reaction initiates with the deprotonation of the naphthol hydroxyl group by the base, generating a nucleophilic phenoxide species. This activated nucleophile then attacks the beta-carbon of the alpha,beta-unsaturated carbonyl compound in a classic Michael addition fashion. This step forms a new carbon-oxygen bond, creating an enolate intermediate. Subsequently, this intermediate undergoes an intramolecular cyclization where the enolate attacks the electrophilic center within the naphthalene ring system, closing the pyran ring to form the stable benzopyran skeleton. The elegance of this tandem process is its ability to form two bonds and a ring in a single operational sequence, maximizing step economy. The choice of catalyst is critical; experimental data within the patent demonstrates that while various bases like DBU, CsF, and t-BuOK can promote the reaction, potassium carbonate provides the optimal balance of basicity and nucleophilicity, driving the equilibrium towards the product with exceptional efficiency.
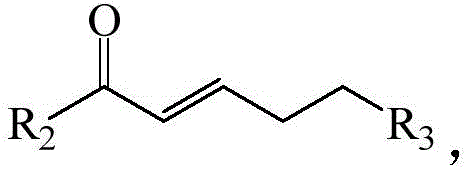
Controlling impurities is another critical aspect where this mechanism excels. In acid-catalyzed reactions, side reactions such as polymerization of the unsaturated ketone or rearrangement of the phenolic substrate are common due to the high reactivity of carbocation intermediates. In contrast, the anionic intermediates generated in this base-catalyzed system are generally more controlled and selective. The mild nature of the base prevents the activation of unintended side pathways, leading to a cleaner reaction profile. Furthermore, the specific selection of the protecting group R3 on the side chain of the unsaturated carbonyl reactant plays a vital role. The patent highlights that when R3 is a dimethyl tert-butyl silyl ether or an acetoxy group, the yields are significantly higher. This suggests that these groups provide the right steric and electronic environment to facilitate the cyclization step without hindering the initial Michael addition, thereby minimizing the formation of oligomeric byproducts and ensuring high purity of the final benzopyran product.
How to Synthesize Benzopyran Compounds Efficiently
Implementing this synthesis requires careful attention to reaction parameters to replicate the high yields reported in the patent. The process begins with the preparation of the alpha,beta-unsaturated carbonyl precursor, which can be synthesized via a Wittig reaction or similar condensation methods as detailed in the patent examples. Once the precursors are ready, the key step involves mixing the unsaturated carbonyl compound with the naphthol derivative and the chosen base in an appropriate solvent. The patent emphasizes the importance of solvent selection, with methanol and N,N-dimethylformamide (DMF) proving superior to non-polar solvents like toluene or dichloroethane. The reaction is typically conducted under an inert atmosphere to prevent oxidation of sensitive intermediates. Following the reaction period, a standard aqueous workup involving dilution with ethyl acetate and washing with brine effectively removes inorganic salts, leaving the organic product ready for purification via column chromatography.
- Prepare the reaction mixture by combining alpha,beta-unsaturated carbonyl compounds, naphthol derivatives, and a basic catalyst such as potassium carbonate in a suitable solvent like methanol or DMF.
- Maintain the reaction under an inert gas atmosphere (nitrogen or argon) and heat the mixture to a temperature between 60°C and 100°C for 8 to 10 hours.
- Upon completion, dilute the system with ethyl acetate, wash with saturated brine, dry the organic phase, and purify via column chromatography to isolate the high-purity benzopyran product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-catalyzed synthesis offers tangible strategic benefits that go beyond mere technical feasibility. The shift away from specialized Lewis acids and precious metal catalysts towards commodity chemicals like potassium carbonate fundamentally alters the cost structure of the manufacturing process. This transition reduces dependency on volatile supply markets for rare earth metals or complex organocatalysts, ensuring a more stable and predictable supply chain. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The replacement of expensive Lewis acid catalysts with inexpensive potassium carbonate results in a drastic reduction in raw material costs. Potassium carbonate is a bulk commodity chemical with a stable global supply, unlike specialized organometallic catalysts which can be subject to price fluctuations and supply shortages. Furthermore, the elimination of acid-sensitive protecting groups reduces the number of synthetic steps required, directly lowering labor, solvent, and energy consumption per kilogram of product. The mild reaction conditions also reduce the energy load on heating and cooling systems, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience. The reagents involved, including naphthol derivatives and alpha,beta-unsaturated ketones, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The tolerance of the reaction to various functional groups means that a wider range of starting materials can be sourced without requiring custom synthesis of highly protected precursors. This flexibility allows procurement teams to negotiate better terms with suppliers and switch sources quickly if disruptions occur, ensuring continuous production flow and preventing delays in delivering critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process is highly favorable for scale-up. The absence of heavy metals simplifies waste treatment protocols and reduces the cost of hazardous waste disposal. The use of common solvents like methanol and DMF, which have well-established recycling infrastructure, further supports green chemistry initiatives. The high atom economy of the tandem reaction minimizes waste generation, aligning with increasingly stringent environmental regulations. This compliance ease facilitates smoother regulatory approvals for new manufacturing sites and reduces the administrative burden associated with environmental reporting, making it an ideal candidate for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and background analysis of patent CN115286608A. These insights are intended to clarify the operational advantages and scope of the technology for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is a base-catalyzed method preferred over traditional Lewis acid catalysis for benzopyran synthesis?
A: Traditional Lewis acid methods often require harsh acidic conditions that can degrade acid-sensitive functional groups like hydroxyl or amino groups commonly found in drug molecules. The base-catalyzed approach described in patent CN115286608A operates under mild alkaline conditions, preserving these sensitive groups without the need for complex protection-deprotection strategies.
Q: What represents the optimal catalyst and solvent system for maximizing yield in this process?
A: Experimental data indicates that potassium carbonate (K2CO3) serves as the most effective catalyst, achieving yields up to 91%. When paired with polar solvents such as methanol or N,N-dimethylformamide (DMF), the reaction efficiency and synthesis speed are significantly enhanced compared to non-polar alternatives.
Q: Can this synthetic route be adapted for large-scale commercial production?
A: Yes, the process utilizes inexpensive, commercially available reagents like potassium carbonate and standard solvents, avoiding expensive precious metal catalysts. The mild reaction temperatures (60-100°C) and straightforward workup procedures make it highly suitable for scaling from laboratory grams to multi-ton industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Supplier
The technological advancements described in patent CN115286608A underscore the immense potential of base-catalyzed tandem reactions in modern organic synthesis. At NINGBO INNO PHARMCHEM, we recognize the value of such innovations in driving efficiency and quality in the production of complex fine chemicals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, ensuring stringent purity specifications and rigorous QC labs to validate every batch against the highest industry standards. We are committed to translating laboratory breakthroughs into reliable, commercial-grade supply solutions.
We invite you to explore how this advanced benzopyran synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to bring high-quality, cost-effective benzopyran intermediates to your development pipeline, ensuring your projects move forward with speed and confidence.
