Advanced Synthesis of Dexmedetomidine Degradation Impurities for Pharmaceutical Quality Control
Advanced Synthesis of Dexmedetomidine Degradation Impurities for Pharmaceutical Quality Control
The pharmaceutical industry faces stringent regulatory requirements regarding the identification and quantification of degradation products in active pharmaceutical ingredients (APIs). Patent CN114436965A addresses a critical gap in the quality control of Dexmedetomidine hydrochloride, a potent sedative agent, by providing a robust method for synthesizing its primary degradation impurities, specifically Impurity A and Impurity B. As a leading reliable pharmaceutical intermediates supplier, understanding the nuances of this patent is essential for ensuring the safety and efficacy of the final drug product. The invention details a novel synthetic pathway that bypasses the limitations of previous methods, offering a streamlined approach to generating high-purity reference standards needed for rigorous analytical validation.
This technological breakthrough is particularly significant for R&D teams focused on stability studies and regulatory filings. The ability to access authentic reference materials allows for precise monitoring of drug degradation during storage and transport. By leveraging the specific reaction conditions outlined in the patent, manufacturers can produce these critical impurities with high selectivity, ensuring that the impurity profile of the bulk drug is accurately characterized. This capability not only supports compliance with international pharmacopoeia standards but also enhances the overall reliability of the supply chain for this vital medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the preparation of Dexmedetomidine degradation impurities relied heavily on direct oxidation or complex total synthesis routes, both of which presented significant operational challenges. The oxidation method, as illustrated in existing literature, involves treating Dexmedetomidine directly to induce degradation; however, this approach suffers from extremely low yields and poor selectivity. 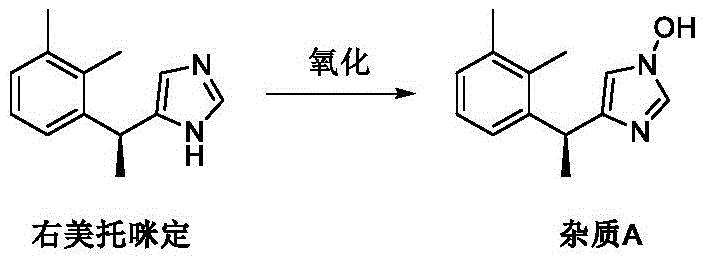
Furthermore, the oxidation process often leads to the simultaneous formation of multiple byproducts, including isomers and over-oxidized compounds, which are structurally similar and notoriously difficult to separate via standard chromatographic techniques. On the other hand, total synthesis routes reported previously involve multi-step sequences exceeding eight steps, resulting in cumulative yield losses and high production costs. These conventional methods fail to provide a practical solution for the large-scale preparation of single, high-purity impurities required for quantitative analysis.
The Novel Approach
The methodology disclosed in patent CN114436965A introduces a divergent synthetic strategy that significantly simplifies the production of both Impurity A and Impurity B. Instead of degrading the final API, this approach constructs the impurity molecules from a common ketone precursor (Compound SM) through a controlled substitution and cyclization sequence. 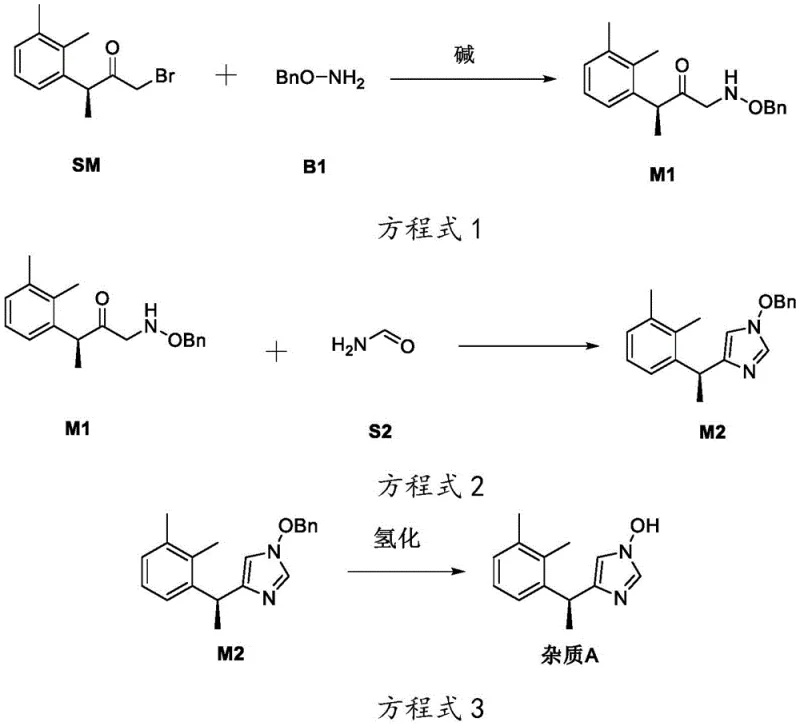
By modulating the reaction conditions, specifically the pH environment during the initial substitution step, the process can be directed towards either the alkaline pathway to produce Impurity A or the acidic pathway to produce Impurity B. This switchable mechanism eliminates the need for complex separation of isomers post-synthesis, as the desired isomer is generated preferentially. The route reduces the step count drastically compared to total synthesis, utilizing readily available reagents like formamide and O-benzylhydroxylamine, thereby achieving cost reduction in API manufacturing while maintaining exceptional purity levels suitable for reference standards.
Mechanistic Insights into pH-Controlled Regioselective Substitution
The core innovation of this patent lies in the precise control of regioselectivity during the nucleophilic substitution reaction between the bromo-ketone precursor (SM) and O-benzylhydroxylamine (B1). Under alkaline conditions, the reaction favors the formation of the O-alkylated intermediate M1, which serves as the specific precursor for Impurity A. Conversely, under acidic conditions, the reaction pathway shifts to favor the formation of the isomeric intermediate M3, which leads exclusively to Impurity B. This pH-dependent divergence is a sophisticated example of reaction engineering that allows for the targeted synthesis of specific isomers without requiring chiral resolution or extensive purification at the early stages.
Following the substitution, the intermediates undergo a cyclization reaction with formamide to construct the imidazole ring, a critical structural motif in the Dexmedetomidine scaffold. The final step involves catalytic hydrogenation to remove the benzyl protecting group, revealing the hydroxyl functionality characteristic of the degradation impurities. 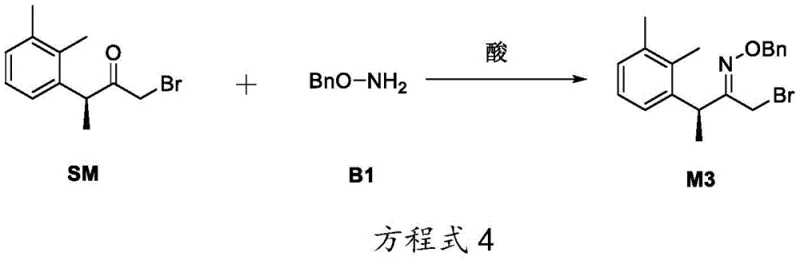
This mechanistic understanding is vital for high-purity pharmaceutical intermediates production, as it highlights the importance of strict process parameter control. The use of mild conditions and common solvents ensures that the process is not only chemically efficient but also environmentally benign. The ability to toggle between two distinct product outcomes using the same starting materials demonstrates a high level of process flexibility, which is a key attribute for contract development and manufacturing organizations (CDMOs) aiming to optimize their production workflows.
How to Synthesize Dexmedetomidine Impurities Efficiently
The synthesis protocol outlined in the patent offers a clear roadmap for producing these critical reference materials. The process begins with the preparation of the key intermediates M1 or M3, followed by ring closure and final deprotection. Each step has been optimized to maximize yield and minimize impurity carryover. For detailed operational parameters, including specific solvent volumes, temperature profiles, and stoichiometric ratios, please refer to the standardized synthesis guide below which encapsulates the core technical steps derived from the patent examples.
- Perform a substitution reaction between compound SM and O-benzylhydroxylamine under controlled pH conditions (alkaline for Impurity A precursor, acidic for Impurity B precursor) to generate intermediates M1 or M3.
- Execute a cyclization reaction by reacting the intermediate (M1 or M3) with formamide to construct the imidazole ring, yielding protected intermediates M2 or M4.
- Conduct a catalytic hydrogenation using Pd/C to remove the benzyl protecting group, followed by purification to isolate the final high-purity degradation impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route presents substantial strategic benefits beyond mere technical feasibility. The shift from low-yield oxidation or lengthy total synthesis to this streamlined three-step process translates directly into improved cost efficiency and supply security. By reducing the number of unit operations and eliminating the need for expensive chiral separation technologies, the overall cost of goods sold (COGS) for these reference standards is significantly lowered. This economic efficiency allows for more competitive pricing models for downstream pharmaceutical clients who require these materials for routine quality assurance testing.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the initial steps and the use of commodity chemicals like formamide drastically reduce raw material costs. Furthermore, the high selectivity of the reaction minimizes waste generation and reduces the burden on downstream purification processes, leading to substantial cost savings in solvent consumption and waste disposal. The simplified workflow also reduces labor hours and equipment occupancy time, enhancing overall plant throughput.
- Enhanced Supply Chain Reliability: Relying on a robust synthetic route that starts from stable, commercially available ketone precursors mitigates the risk of supply disruptions associated with sourcing complex natural products or unstable intermediates. The ability to produce both Impurity A and Impurity B from a single common starting material simplifies inventory management and reduces the complexity of the supply chain. This flexibility ensures that manufacturers can respond quickly to fluctuating demand for specific reference standards without maintaining excessive stock levels of diverse raw materials.
- Scalability and Environmental Compliance: The process avoids the use of hazardous oxidants and extreme reaction conditions, making it inherently safer and easier to scale from laboratory to commercial production. The workup procedures involve standard aqueous extractions and crystallizations, which are well-understood unit operations in the fine chemical industry. This alignment with green chemistry principles not only facilitates regulatory approval but also supports corporate sustainability goals by minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this method for industrial purposes.
Q: How does this new method improve upon traditional oxidation routes for Impurity A?
A: Traditional oxidation of Dexmedetomidine yields very low amounts of Impurity A and generates difficult-to-separate byproducts like Impurity B and over-oxidized compounds. This new method uses a directed synthesis approach starting from a ketone precursor, allowing for high yield and superior purity without complex separation challenges.
Q: Can both Impurity A and Impurity B be produced using the same starting materials?
A: Yes, the patent describes a divergent synthesis strategy where the same starting material (Compound SM) is used. By simply adjusting the reaction environment from alkaline to acidic during the initial substitution step, manufacturers can selectively produce the specific isomer required, streamlining inventory management.
Q: Is this synthesis route scalable for commercial reference standard production?
A: The process utilizes common reagents like formamide and standard catalytic hydrogenation, avoiding exotic catalysts or extreme conditions. The workup involves standard extraction and crystallization techniques, making it highly suitable for scale-up from gram to kilogram quantities for industrial quality control needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexmedetomidine Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality reference standards play in the pharmaceutical lifecycle. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Dexmedetomidine impurities meets the highest industry standards, supporting your regulatory submissions and quality control initiatives with confidence.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for commercial scale-up of complex pharmaceutical intermediates.
