Sustainable Regeneration of N,N'-dicyclohexylcarbodiimide for High-Purity Pharmaceutical Synthesis
Sustainable Regeneration of N,N'-dicyclohexylcarbodiimide for High-Purity Pharmaceutical Synthesis
The efficient management of reagents in peptide synthesis and fine chemical manufacturing is a critical challenge for modern R&D departments, particularly concerning the ubiquitous dehydrating agent N,N'-dicyclohexylcarbodiimide (DCC). Patent CN101928237A introduces a transformative regeneration method that addresses the accumulation of N,N'-dicyclohexylurea (DCU), a stoichiometric byproduct formed during DCC-mediated couplings. This technology enables the conversion of waste DCU back into active DCC through an oxidative dehydration process, achieving regeneration rates between 60% and 78% with product purity exceeding 99.5%. By closing the loop on reagent consumption, this process not only mitigates environmental impact but also offers a robust pathway for cost reduction in pharmaceutical intermediates manufacturing. The structural integrity of the regenerated product is confirmed to be identical to virgin DCC, ensuring reliability in downstream applications.
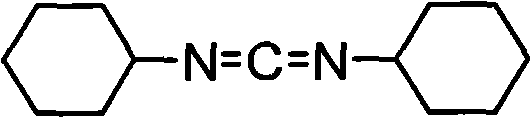
For procurement managers and supply chain heads, the implications of this technology extend beyond mere waste reduction; it represents a strategic shift towards circular economy principles within the supply chain. The ability to recycle a major byproduct reduces the dependency on fresh raw material sourcing, thereby insulating production schedules from volatile market fluctuations. Furthermore, the process utilizes common industrial solvents and oxidizing agents, eliminating the need for exotic or highly regulated catalysts that often complicate logistics. As a reliable pharmaceutical intermediates supplier, understanding these underlying process efficiencies allows us to offer more competitive pricing structures and guaranteed continuity of supply for high-volume clients engaged in large-scale peptide production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial synthesis of N,N'-dicyclohexylcarbodiimide has relied heavily on the thiourea route, which involves the reaction of dicyclohexylamine with carbon disulfide to form a thiourea derivative, followed by desulfurization. While effective, this conventional pathway presents significant drawbacks, particularly regarding environmental safety and operational complexity. One common variation utilizes red precipitate (mercuric oxide) as the desulfurizing agent, introducing toxic heavy metals into the process stream which necessitates rigorous and costly waste treatment protocols to meet environmental compliance standards. Another variation employs hypochlorite oxidation, which, while avoiding mercury, can suffer from issues related to selectivity and the formation of chlorinated byproducts that are difficult to separate. These legacy methods often result in lower overall atom economy and generate substantial hazardous waste, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates where green chemistry metrics are increasingly scrutinized by regulatory bodies and corporate sustainability officers.
The Novel Approach
In stark contrast, the regeneration method disclosed in the patent data utilizes N,N'-dicyclohexylurea (DCU) directly as the starting material, effectively turning a waste liability into a valuable asset. This novel approach bypasses the initial synthesis of the urea backbone entirely, focusing instead on the oxidative dehydration of the existing DCU molecule. By employing oxidizing agents such as phosphorus oxychloride, toluene sulfonyl chloride, or vanadium pentoxide in mild organic solvents, the process achieves high conversion efficiency under relatively gentle thermal conditions (40-42°C). This shift eliminates the need for handling toxic carbon disulfide or heavy metal desulfurizers, drastically simplifying the safety profile of the manufacturing facility. The result is a streamlined workflow that not only enhances the high-purity N,N'-dicyclohexylcarbodiimide output but also aligns perfectly with modern green chemistry principles, offering a sustainable alternative for the global fine chemical industry.
Mechanistic Insights into Oxidative Dehydration Regeneration
The core chemical transformation in this process is the oxidative dehydration of the urea linkage (-NH-CO-NH-) to the carbodiimide functionality (-N=C=N-). Mechanistically, this involves the activation of the urea oxygen by the oxidizing agent, typically a phosphoryl or sulfonyl chloride, which acts as a potent dehydrating species. In the presence of an organic solvent such as methylene dichloride or tetrahydrofuran, the oxidizing agent attacks the carbonyl oxygen of the DCU, forming a reactive intermediate that facilitates the elimination of water (in the form of the oxidant's hydrolyzed byproduct, e.g., metaphosphoric acid or sulfonic acid). This elimination step is crucial and is driven by the thermodynamic stability of the resulting carbodiimide double bond system. The reaction is conducted at a controlled temperature range of 40-42°C, which is sufficient to overcome the activation energy barrier for dehydration without promoting thermal decomposition or side reactions that could compromise the integrity of the cyclohexyl rings. This precise thermal control is a key factor in maintaining the high selectivity observed in the patent examples.
Impurity control is rigorously managed through a multi-stage workup procedure designed to remove both organic and inorganic contaminants. Following the reaction, the mixture is quenched into an alkaline aqueous solution, such as sodium hydroxide or sodium carbonate, which serves a dual purpose: it neutralizes the acidic byproducts generated during oxidation (like HCl or sulfonic acids) and hydrolyzes any unreacted oxidizing agent. The use of an alkaline wash in the pre-treatment stage of the raw DCU also ensures that water-soluble impurities carried over from previous peptide coupling steps are removed before regeneration begins. This comprehensive purification strategy ensures that the final organic phase, upon concentration and crystallization, yields DCC with a purity greater than 99.5%. Such high purity is essential for reducing lead time for high-purity pharmaceutical intermediates in downstream synthesis, as it minimizes the need for additional recrystallization steps that would otherwise delay production timelines.
How to Synthesize N,N'-dicyclohexylcarbodiimide Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for implementing this regeneration technology at scale. The process begins with the meticulous pre-treatment of the DCU feedstock, ensuring that the starting material is free from moisture and soluble contaminants that could interfere with the oxidative step. The reaction itself is straightforward, requiring standard stirred-tank reactor equipment capable of maintaining precise temperature control and handling corrosive oxidizing agents safely. Detailed standardized synthetic steps see the guide below, which breaks down the specific mass ratios and operational parameters required to achieve the reported 60-78% regeneration rates. By adhering to these optimized conditions, manufacturers can consistently produce DCC that meets the stringent quality specifications required for GMP-grade pharmaceutical applications.
- Pre-treatment: Wash N,N'-dicyclohexylurea (DCU) with alkaline aqueous solution to remove water-soluble impurities, followed by centrifugal dehydration and drying.
- Regenerative Reaction: Dissolve dried DCU in an organic solvent (e.g., methylene dichloride) and react with an oxidizing agent (e.g., phosphorus oxychloride) at 40-42°C for 0.15 to 2 hours.
- Purification: Quench the reaction mixture into alkaline solution, separate the organic phase, and concentrate via crystallization to obtain DCC with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this regeneration technology offers compelling economic and logistical advantages that directly impact the bottom line. By converting a low-value byproduct (DCU) back into a high-value reagent (DCC), the effective cost of goods sold for the active ingredient is significantly reduced, as the raw material input is essentially recycled rather than purchased anew. This circular model decouples production costs from the volatility of raw amine markets, providing a more stable pricing structure for long-term contracts. Furthermore, the simplified reaction conditions eliminate the need for specialized equipment required for handling toxic gases like hydrogen sulfide or carbon disulfide, thereby reducing capital expenditure and maintenance costs associated with corrosion and safety systems.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the valorization of waste DCU. In traditional peptide synthesis, DCU is often discarded as solid waste, incurring disposal fees and representing a total loss of the expensive dicyclohexylamine moiety. By regenerating DCC, the effective utilization of the amine precursor is maximized, leading to substantial cost savings on raw materials. Additionally, the process avoids the use of expensive and toxic heavy metal catalysts like mercury oxide, which not only lowers reagent costs but also eliminates the significant expense associated with heavy metal removal and hazardous waste disposal. The mild reaction temperatures further contribute to energy efficiency, reducing the utility load required for heating and cooling compared to more vigorous traditional synthesis routes.
- Enhanced Supply Chain Reliability: The reliance on readily available and commodity-grade chemicals such as phosphorus oxychloride, toluene sulfonyl chloride, and common organic solvents ensures a robust and resilient supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, the reagents for this regeneration process are produced globally in large volumes, minimizing the risk of supply disruptions. The ability to source DCU from internal waste streams of peptide manufacturing facilities also creates a localized supply loop, reducing transportation dependencies and enhancing the overall agility of the supply network. This reliability is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: From a scalability perspective, the process is highly amenable to large-scale production due to its homogeneous nature and the use of standard unit operations like liquid-liquid extraction and crystallization. The absence of heterogeneous catalysis or complex filtration steps simplifies the scale-up trajectory from pilot plant to commercial tonnage. Environmentally, the process aligns with increasingly strict global regulations regarding waste discharge and heavy metal usage. By eliminating mercury and reducing the volume of solid chemical waste, facilities can more easily maintain compliance with environmental permits, avoiding potential fines and reputational damage. This 'green' credential is increasingly becoming a prerequisite for supplying major multinational pharmaceutical companies committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the regeneration of N,N'-dicyclohexylcarbodiimide. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on the feasibility and advantages of this method for industrial application. Understanding these details is vital for technical teams evaluating the integration of recycled reagents into their GMP manufacturing workflows.
Q: What is the primary advantage of regenerating DCC from DCU compared to traditional synthesis?
A: The regeneration method converts the waste byproduct N,N'-dicyclohexylurea (DCU), which accumulates during peptide coupling, back into active DCC. This circular approach significantly reduces raw material costs and environmental waste compared to the traditional thiourea route which often involves toxic heavy metals.
Q: What purity levels can be achieved with this regeneration process?
A: According to patent CN101928237A, the regenerated N,N'-dicyclohexylcarbodiimide achieves a content of over 99.5% as determined by gas chromatography, making it suitable for sensitive pharmaceutical applications like amide and polypeptide synthesis.
Q: Is this process scalable for industrial production?
A: Yes, the process operates under mild conditions (40-42°C) using readily available solvents and oxidizing agents. The absence of complex catalytic cycles and the use of standard separation techniques like phase separation and crystallization facilitate easy scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-dicyclohexylcarbodiimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality coupling reagents play in the success of peptide and pharmaceutical synthesis. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require kilogram quantities for R&D or metric tons for commercial manufacturing, our supply remains uninterrupted. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N,N'-dicyclohexylcarbodiimide meets the exacting standards required for sensitive chemical transformations. Our commitment to process innovation allows us to leverage technologies like the DCU regeneration method to offer competitive value without compromising on quality.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your project goals efficiently and reliably.
