Advanced Michael Addition Strategy for Scalable Benzhydryl Quinuclidinone Production
Advanced Michael Addition Strategy for Scalable Benzhydryl Quinuclidinone Production
The pharmaceutical landscape for veterinary medicine is constantly evolving, driven by the need for more efficient and environmentally sustainable synthesis routes for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN108822101B, which discloses a robust method for synthesizing benzhydryl quinuclidinone via a copper-catalyzed Michael addition. This compound serves as a pivotal intermediate in the manufacture of Maropitant, a potent neurokinin-1 (NK1) receptor antagonist widely recognized under the trade name Cerenia for its antiemetic properties in large animals. The structural integrity and purity of this quinuclidinone derivative are paramount, as they directly influence the efficacy and safety profile of the final veterinary drug. By leveraging a specific Grignard reagent addition strategy in the presence of a cuprous catalyst, this technology addresses long-standing challenges regarding yield optimization and solvent safety that have plagued previous synthetic attempts.
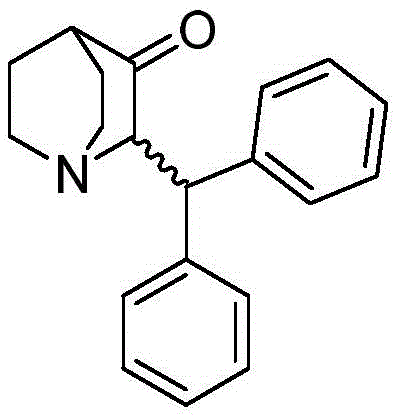
The significance of this patent extends beyond mere academic interest; it represents a tangible solution for industrial manufacturers seeking to streamline their supply chains for veterinary intermediates. The traditional reliance on hazardous solvents and the acceptance of suboptimal yields have created bottlenecks in production capacity and increased regulatory burdens. The methodology outlined in CN108822101B utilizes readily available starting materials, specifically (Z)-2-benzylidenequinuclidin-3-one, and subjects them to a controlled conjugate addition. This approach not only enhances the atom economy of the process but also ensures that the resulting product meets stringent purity specifications, often exceeding 99% as determined by HPLC analysis. For stakeholders in the fine chemical industry, understanding the nuances of this transformation is essential for evaluating potential partnerships and sourcing strategies for high-value heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzhydryl quinuclidinone has been fraught with significant technical and environmental drawbacks that hindered its widespread commercial adoption. Early methodologies, such as those disclosed in US3560510, relied heavily on the use of benzene as a reaction solvent. While benzene is an effective solvent for certain organic transformations, its classification as a known human carcinogen imposes severe restrictions on its use in modern Good Manufacturing Practice (GMP) environments. Furthermore, these legacy processes operated without the benefit of transition metal catalysis, resulting in poor reaction control and mediocre yields hovering around 51.8%. The lack of selectivity often led to complex mixtures that required extensive and costly purification steps, thereby inflating the overall cost of goods sold (COGS) and generating substantial chemical waste.
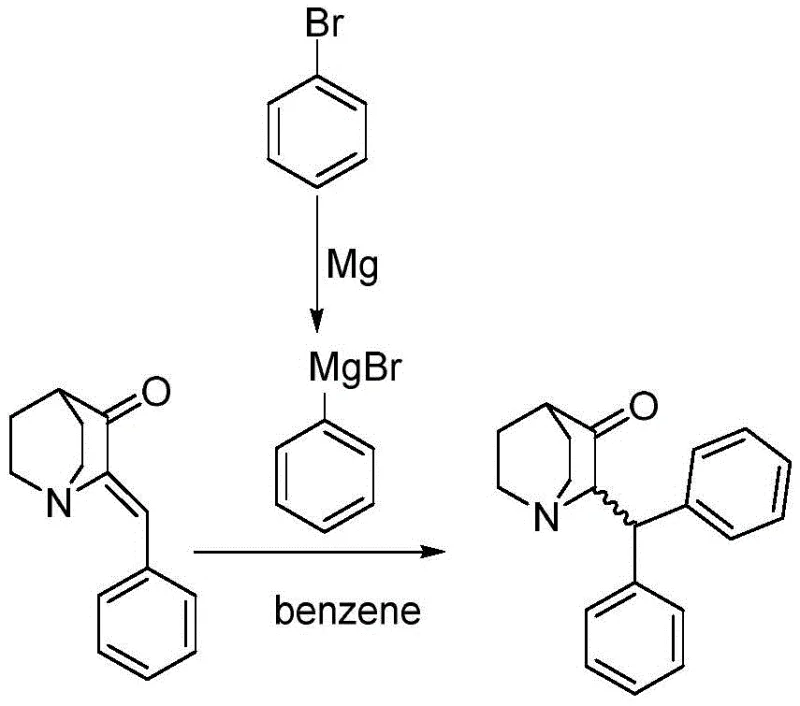
In addition to solvent toxicity, other reported methods suffered from fundamental chemoselectivity issues. For instance, research published in the European Journal of Medicinal Chemistry highlighted attempts using cuprous iodide in benzene, which paradoxically resulted in yields as low as 3%. The primary failure mode in these uncatalyzed or poorly catalyzed systems was the preferential formation of alcohol byproducts via 1,2-addition rather than the desired 1,4-conjugate addition. This side reaction not only consumed valuable raw materials but also generated impurities that were structurally similar to the target molecule, making separation via standard column chromatography difficult and impractical for large-scale operations. The cumulative effect of these limitations was a synthesis route that was neither economically viable nor environmentally sustainable for the demands of the modern veterinary pharmaceutical market.
The Novel Approach
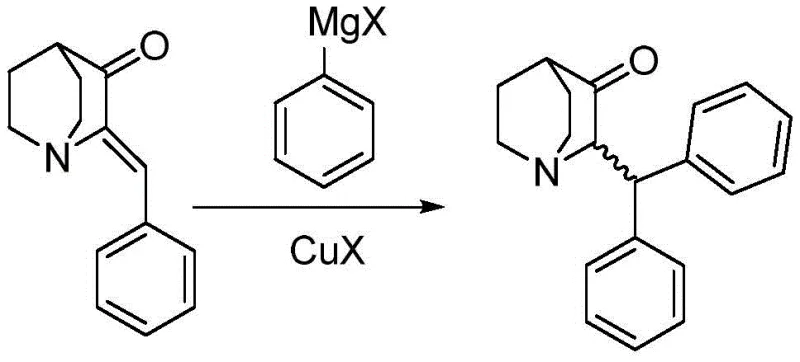
The innovative process described in CN108822101B fundamentally alters the reaction landscape by introducing a specific cuprous catalyst system that directs the Grignard reagent exclusively toward the desired Michael addition pathway. By employing catalysts such as cuprous bromide, cuprous chloride, or cuprous iodide in molar ratios ranging from 1:0.01 to 1:0.30 relative to the substrate, the reaction achieves exceptional regioselectivity. This catalytic intervention effectively suppresses the formation of the troublesome alcohol byproducts, channeling the reaction flux toward the formation of the 2-(benzhydryl)quinuclidin-3-one skeleton with remarkable efficiency. Under optimized conditions, this method consistently delivers yields reaching up to 85%, a substantial improvement over the historical benchmarks. Moreover, the process eliminates the need for carcinogenic benzene, substituting it with safer, industrially friendly solvents like toluene, tetrahydrofuran (THF), or methyl tert-butyl ether (MTBE).
Operational simplicity is another hallmark of this novel approach, making it highly attractive for contract development and manufacturing organizations (CDMOs). The reaction proceeds under mild thermal conditions, typically between room temperature and 50°C, over a duration of 8 to 24 hours. This thermal leniency reduces energy consumption and minimizes the risk of thermal runaway incidents, which are critical considerations for process safety management. The workup procedure is equally straightforward, involving a standard aqueous quench with ammonium chloride followed by extraction and crystallization from absolute ethanol. This streamlined downstream processing ensures that the final product is obtained with a purity greater than 99%, ready for subsequent coupling reactions in the Maropitant synthesis sequence without the need for complex chromatographic purification. Such operational robustness is a key enabler for reliable supply chain continuity.
Mechanistic Insights into Copper-Catalyzed Conjugate Addition
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic role of the cuprous species in mediating the Grignard reaction. In the absence of a copper catalyst, phenylmagnesium halides typically exhibit hard nucleophilic character, favoring direct attack at the carbonyl carbon (1,2-addition) of the alpha,beta-unsaturated ketone substrate. This kinetic preference leads to the formation of tertiary alcohols, which constitute the major impurities observed in prior art methods. However, the introduction of a cuprous salt (CuX) facilitates the transmetallation of the Grignard reagent to form an organocuprate species in situ. Organocuprates are softer nucleophiles compared to their Grignard counterparts, and they possess a distinct thermodynamic preference for conjugate addition (1,4-addition) to the beta-carbon of the enone system.
This mechanistic switch is the cornerstone of the high selectivity observed in the patent. The organocuprate intermediate adds to the beta-position of the (Z)-2-benzylidenequinuclidin-3-one, generating an enolate intermediate that subsequently protonates upon aqueous workup to yield the saturated ketone product. The careful control of the catalyst loading, specifically within the range of 1:0.01 to 1:0.30, ensures that there is sufficient catalytic turnover to drive the reaction to completion without introducing excessive amounts of metal contaminants that would require rigorous removal later. Furthermore, the choice of solvent plays a crucial role in stabilizing these organometallic intermediates; ethers like THF coordinate with the magnesium and copper centers, maintaining the solubility and reactivity of the catalytic species throughout the 8 to 24-hour reaction window. This precise orchestration of reagents and conditions effectively shuts down the competing 1,2-addition pathway, ensuring an impurity profile that is easily managed through simple crystallization.
How to Synthesize 2-(Benzhydryl)quinuclidin-3-one Efficiently
The practical implementation of this synthesis requires strict adherence to the optimized parameters regarding reagent stoichiometry and temperature control to maximize yield and purity. The process begins with the preparation of the electrophile solution, followed by the generation of the active nucleophilic species under inert atmosphere. Detailed standardized operating procedures for this transformation are provided below to ensure reproducibility and safety during scale-up.
- Prepare Solution A by dissolving (Z)-2-benzylidenequinuclidin-3-one in a reaction solvent such as tetrahydrofuran or toluene.
- Mix a phenylmagnesium halide Grignard reagent with a cuprous catalyst (e.g., CuBr) under nitrogen protection at low temperature.
- Add Solution A dropwise to the catalyst mixture, allow reaction to proceed at room temperature to 50°C, then quench and crystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN108822101B offers compelling strategic advantages that extend well beyond the laboratory bench. The shift away from hazardous solvents and the dramatic improvement in reaction yield translate directly into tangible economic and logistical benefits. By replacing benzene with common solvents like toluene or THF, manufacturers can significantly reduce the costs associated with specialized hazardous waste disposal and regulatory compliance monitoring. This simplification of the environmental health and safety (EHS) profile allows for faster permitting and more flexible production scheduling, which is critical for meeting the fluctuating demands of the global veterinary pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive and complex purification steps, such as column chromatography, results in substantial cost savings. The high intrinsic purity of the crude product achieved through this catalytic method means that a simple crystallization step is sufficient to meet quality standards. Furthermore, the use of inexpensive cuprous salts as catalysts, rather than precious metals or stoichiometric reagents, lowers the raw material input costs. The high yield of 85% ensures that less starting material is wasted, optimizing the overall atom economy and reducing the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commoditized raw materials enhances supply security. Both the starting material, (Z)-2-benzylidenequinuclidin-3-one, and the Grignard reagents are commercially available in bulk quantities from multiple suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, provides manufacturing flexibility. This resilience ensures that production can continue uninterrupted even if specific grades of solvents face temporary shortages, thereby guaranteeing consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, having been demonstrated effectively from gram to kilogram scales in the patent examples. The absence of malodorous sulfur-containing complexes, which were present in some prior art methods, improves the working environment and reduces the burden on scrubber systems. The use of non-carcinogenic solvents aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing process against tighter legislative controls. This compliance advantage minimizes the risk of production shutdowns due to regulatory non-compliance, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of benzhydryl quinuclidinone. These answers are derived directly from the experimental data and claims presented in the patent literature to provide accurate guidance for technical decision-makers.
Q: What are the key advantages of the copper-catalyzed method over prior art?
A: The copper-catalyzed method described in CN108822101B achieves yields up to 85% and purity greater than 99%, significantly outperforming older methods that suffered from low yields (around 51%) or required carcinogenic benzene solvents.
Q: Why is this intermediate critical for veterinary pharmaceuticals?
A: Benzhydryl quinuclidinone is the essential core structure for Maropitant (Cerenia), a widely used NK1 receptor antagonist for preventing vomiting in dogs and cats, making its efficient synthesis vital for the veterinary supply chain.
Q: Does this process eliminate hazardous solvents like benzene?
A: Yes, the novel process replaces carcinogenic benzene with safer, industrially common solvents such as toluene, tetrahydrofuran, or methyl tert-butyl ether, greatly improving environmental compliance and operator safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzhydryl Quinuclidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving veterinary medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering benzhydryl quinuclidinone with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for GMP API synthesis. Our facility is equipped to handle the specific solvent and safety requirements of this copper-catalyzed process, guaranteeing a supply that is both consistent and compliant.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their Maropitant programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market.
