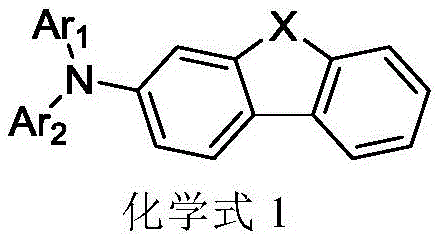
Advanced Synthesis of Tertiary Amines for High-Performance OLED Hole Transport Layers
The rapid evolution of organic electroluminescent devices (OLEDs) has created an insatiable demand for high-performance hole transport materials that offer superior stability and efficiency. Patent CN110885320B discloses a groundbreaking preparation method for tertiary amine compounds, specifically targeting the structural motifs essential for next-generation display technologies. This technical disclosure addresses critical bottlenecks in the synthesis of triarylamine derivatives where nitrogen atoms are connected to 3-substituted dibenzothiophene or dibenzofuran structures. By fundamentally re-engineering the synthetic pathway, this innovation provides a robust framework for producing high-purity OLED materials while simultaneously mitigating significant safety and environmental liabilities associated with traditional manufacturing protocols.
The strategic value of this patent lies in its ability to bypass the reliance on hazardous precursors that have long plagued the fine chemical industry. For R&D directors and process chemists, the shift away from carcinogenic primary amines represents a paradigm shift in risk management and regulatory compliance. The disclosed method ensures that the resulting tertiary amine compounds maintain the rigorous electronic properties required for efficient charge transport, making them ideal candidates for integration into complex multi-layered organic thin film structures. As a reliable OLED material supplier, understanding these mechanistic nuances is paramount for securing a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the prior art, specifically referenced in documents such as JP2018090561A, the synthesis of triarylamine compounds typically relies on the reaction of 3-bromodibenzofuran or 3-bromodibenzothiophene with aryl primary amines. This conventional approach is fraught with substantial disadvantages that hinder both economic viability and operational safety. Firstly, the starting material, 3-bromodibenzofuran or 3-bromodibenzothiophene, is not only prohibitively expensive but also notoriously difficult to prepare, often requiring lengthy multi-step syntheses involving high-risk operations. Furthermore, the aryl primary amine compounds necessary for this coupling, such as 4-aminobiphenyl, 1-naphthylamine, and 2-naphthylamine, are classified as primary carcinogens by the World Health Organization. The handling of these substances necessitates extreme containment measures, driving up operational costs and creating significant liability for manufacturers.
The Novel Approach
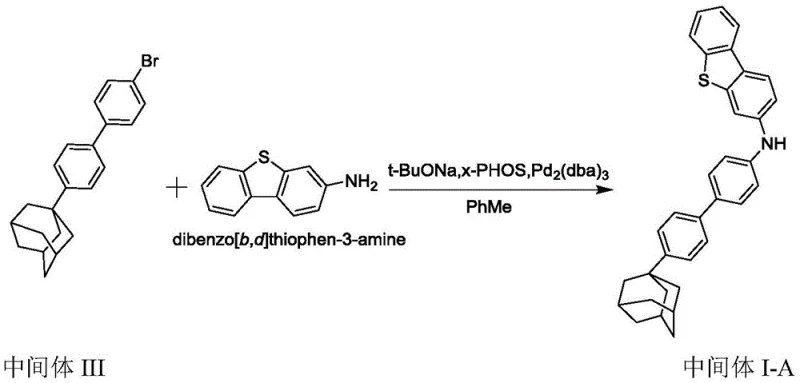
The methodology presented in CN110885320B ingeniously reverses the coupling strategy to circumvent these pitfalls. Instead of reacting a brominated heterocycle with a toxic amine, the process utilizes 3-aminodibenzofuran or 3-aminodibenzothiophene as the core nucleophile, reacting it with safer aryl halides. This strategic inversion eliminates the need for hazardous primary aryl amines entirely. The synthesis proceeds through a sequential carbon-nitrogen coupling protocol, first generating a secondary amine intermediate and subsequently converting it into the target tertiary amine. This approach not only drastically reduces the toxicity profile of the raw materials but also simplifies the overall process flow by avoiding dangerous nitration and hydrogenation steps often required to synthesize the brominated precursors used in older methods.
This novel pathway offers a cleaner, more sustainable route to complex electronic chemicals. By substituting dangerous amines with stable aryl halides, the process inherently lowers the barrier for commercial scale-up. The avoidance of explosive nitration reactions and high-pressure hydrogenation further enhances the safety profile, making this method particularly attractive for facilities aiming to reduce their environmental footprint while maintaining high throughput. For procurement managers, this translates into a more resilient supply chain less susceptible to regulatory shutdowns or safety incidents.
Mechanistic Insights into Palladium-Catalyzed Carbon-Nitrogen Coupling
The core of this innovative synthesis relies on the Buchwald-Hartwig amination reaction, a powerful tool for constructing carbon-nitrogen bonds. In the first stage, the compound of chemical formula 2 reacts with the compound of chemical formula 3 in the presence of a palladium catalyst, a specialized organophosphine ligand, and a strong base. The patent specifies the use of catalysts such as tris(dibenzylideneacetone)dipalladium (Pd2(dba)3) or palladium acetate, paired with bulky, electron-rich ligands like x-Phos or s-Phos. These ligands are critical for facilitating the oxidative addition of the aryl halide and stabilizing the active palladium species, thereby enabling the coupling to proceed efficiently even with sterically hindered substrates. The reaction is typically conducted in toluene at temperatures ranging from 100°C to 105°C under an inert nitrogen atmosphere to prevent catalyst deactivation.
Following the formation of the secondary amine intermediate (chemical formula 4), the process undergoes a second coupling cycle with a different aryl halide (chemical formula 5) to install the final aryl group. The mechanistic precision here allows for the modular assembly of diverse tertiary amine structures by simply varying the aryl halide inputs. Impurity control is managed through careful selection of stoichiometry, with molar ratios optimized to minimize homocoupling side reactions. The patent data indicates that yields for the first coupling step can reach 60% to 80%, while the second step achieves 50% to 75%. Post-reaction workup involves standard extraction and purification techniques, such as column chromatography and recrystallization, ensuring the final product meets the stringent purity specifications (>99.9% HPLC) required for electronic applications.
How to Synthesize Tertiary Amine Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value intermediates in a laboratory or pilot plant setting. The procedure emphasizes the importance of maintaining anhydrous and oxygen-free conditions to preserve catalyst activity throughout the reaction duration. Detailed operational parameters, including specific solvent volumes and heating profiles, are provided to ensure reproducibility. For those seeking to implement this technology, the following guide summarizes the critical operational phases derived directly from the patent examples.
- Perform a carbon-nitrogen coupling reaction between a compound of chemical formula 2 (3-aminodibenzothiophene/furan derivative) and a compound of chemical formula 3 (aryl halide) using a palladium catalyst and base to obtain a secondary amine intermediate (chemical formula 4).
- Conduct a second carbon-nitrogen coupling reaction between the obtained secondary amine (chemical formula 4) and a compound of chemical formula 5 (another aryl halide) under similar catalytic conditions to yield the final tertiary amine compound (chemical formula 1).
- Purify the final reaction mixture through extraction, drying, concentration, and recrystallization or column chromatography to achieve HPLC purity greater than 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the advantages of this patented process extend far beyond mere chemical elegance. The elimination of carcinogenic raw materials fundamentally alters the cost structure of manufacturing by removing the need for expensive safety infrastructure and hazardous waste disposal protocols. Traditional methods relying on toxic amines often incur hidden costs related to regulatory compliance, worker health monitoring, and specialized containment equipment. By switching to safer aryl halide coupling partners, manufacturers can achieve significant cost reduction in electronic chemical manufacturing without compromising on product quality or performance metrics.
- Cost Reduction in Manufacturing: The avoidance of expensive and difficult-to-prepare 3-bromodibenzofuran or 3-bromodibenzothiophene precursors leads to direct material cost savings. Furthermore, the simplified synthetic route reduces the number of unit operations, lowering energy consumption and labor hours per kilogram of product. The use of commercially available and relatively inexpensive aryl halides instead of custom-synthesized toxic amines creates a more favorable economic model for large-scale production.
- Enhanced Supply Chain Reliability: Sourcing hazardous primary amines can be challenging due to strict transportation regulations and limited supplier bases. By utilizing widely available aryl halides and safer amine cores, the supply chain becomes more robust and less prone to disruptions. This reliability is crucial for meeting the tight delivery schedules demanded by the consumer electronics industry, ensuring that production lines for OLED displays remain uninterrupted.
- Scalability and Environmental Compliance: The process operates under mild thermal conditions and avoids high-pressure hydrogenation or exothermic nitration reactions, which are common barriers to scaling fine chemical processes. This inherent safety makes the technology easier to transfer from the lab to multi-ton reactors. Additionally, the reduced generation of hazardous waste aligns with increasingly stringent global environmental standards, future-proofing the manufacturing facility against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived from the specific experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability potential.
Q: How does this synthesis method improve safety compared to traditional routes?
A: This method avoids the use of highly toxic and carcinogenic aryl primary amines such as 4-aminobiphenyl and naphthylamines, which are commonly required in prior art. Instead, it utilizes safer aryl halides as coupling partners, significantly reducing operator health risks and environmental hazards.
Q: What are the typical yields and purity levels achievable with this process?
A: The patent data indicates that the first coupling step can achieve yields between 60% and 80%, while the second coupling step typically yields between 50% and 75%. Final products can be purified to HPLC purity levels exceeding 99.9% through standard recrystallization and chromatography techniques.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process eliminates dangerous operations such as high-pressure hydrogenation and nitration found in conventional methods. The use of mild reaction conditions (100-105°C) and stable reagents facilitates safer and more reliable commercial scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tertiary Amine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced hole transport materials play in the advancement of OLED technology. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless. We are committed to delivering high-purity tertiary amine compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch delivered conforms to the exacting standards required for high-performance electronic applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient methodology. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of next-generation OLED materials.
