Revolutionizing Letrozole Intermediate Production: A Deep Dive into High-Efficiency Pd-Cu Catalytic Coupling
The pharmaceutical industry is constantly seeking more efficient pathways to produce critical oncology medications, and the synthesis of letrozole, a potent third-generation aromatase inhibitor, remains a focal point for process optimization. Patent CN113072462A introduces a groundbreaking preparation method for a key letrozole intermediate that fundamentally shifts the paradigm from multi-step, hazardous sequences to a streamlined, one-step catalytic coupling. This innovation addresses the longstanding challenges of low overall yields and toxic reagent usage that have plagued previous synthetic strategies. By leveraging a sophisticated palladium and copper dual-catalyst system, this technology enables the direct construction of the diphenylmethane backbone from readily available carboxylic acid and halide precursors. For R&D directors and procurement specialists alike, this represents a significant opportunity to enhance the economic viability and environmental sustainability of letrozole manufacturing. The ability to achieve yields exceeding 90% in a single transformation drastically reduces the material throughput required, directly impacting the cost of goods sold (COGS) and simplifying the supply chain logistics for this high-value pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of letrozole has been fraught with chemical inefficiencies and safety hazards that complicate large-scale production. Traditional Route 1, for instance, relies on the bromination of p-methyl benzonitrile followed by triazole substitution, a sequence that often generates difficult-to-separate isomers and requires harsh acidic conditions for purification. Other pathways, such as Route 2, utilize n-butyllithium at cryogenic temperatures of -60°C, presenting severe safety risks and imposing heavy energy burdens on the manufacturing infrastructure. Furthermore, Route 3 involves diazotization reactions employing cuprous cyanide, a highly toxic reagent that creates substantial wastewater treatment challenges and regulatory compliance hurdles. Even the more industrially favored Route 6, which starts from 4,4'-dicyanobenzophenone, faces bottlenecks due to the difficulty and cost associated with synthesizing the benzophenone starting material itself. These legacy methods collectively suffer from long reaction sequences, cumulative yield losses, and the generation of significant chemical waste, making them increasingly untenable in a modern, green-chemistry-focused manufacturing environment.
The Novel Approach
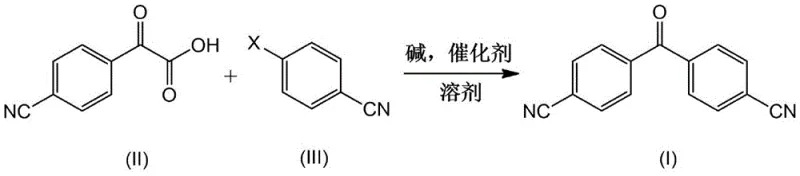
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN113072462A offers a remarkably concise and robust alternative. This novel approach utilizes 2-(4-cyanophenyl)formylcarboxylic acid and 4-halobenzonitrile as the primary building blocks, reacting them in a single pot under the influence of a Pd-Cu catalytic system. This decarboxylative cross-coupling strategy elegantly bypasses the need for pre-functionalized benzophenones or toxic cyanide salts. The reaction proceeds smoothly in common polar aprotic solvents like DMF or dioxane at moderate temperatures ranging from 80°C to 140°C. By eliminating multiple isolation steps and avoiding extreme conditions, this route not only simplifies the operational workflow but also significantly enhances the overall atom economy. The result is a high-purity intermediate that serves as a superior precursor for the final letrozole API, ensuring that downstream processing is both efficient and reliable. This technological leap provides a clear competitive advantage for manufacturers looking to optimize their production lines for cost and safety.
Mechanistic Insights into Pd-Cu Dual Catalytic Decarboxylative Coupling
The core of this innovative synthesis lies in the synergistic interaction between the palladium and copper catalysts, which facilitates a complex decarboxylative cross-coupling mechanism. The reaction likely initiates with the oxidative addition of the aryl halide to the palladium center, forming an aryl-palladium species. Simultaneously, the copper catalyst assists in the activation of the carboxylic acid substrate, promoting decarboxylation to generate a nucleophilic organometallic intermediate. This transmetallation step is critical, as it transfers the organic fragment from copper to palladium, setting the stage for the final reductive elimination that forms the carbon-carbon bond. The presence of nitrogen ligands, such as 1,10-phenanthroline or bipyridine, plays a pivotal role in stabilizing the metal centers and accelerating the catalytic turnover. This dual-catalyst design effectively overcomes the kinetic barriers typically associated with coupling unactivated carboxylic acids, allowing the reaction to proceed with high efficiency and selectivity. Understanding this mechanistic nuance is vital for process chemists aiming to fine-tune reaction parameters for maximum throughput.
From an impurity control perspective, this mechanism offers distinct advantages over traditional electrophilic aromatic substitutions or radical pathways. The specificity of the transition metal catalysis minimizes the formation of regioisomers and homocoupling by-products that often plague non-catalytic methods. The patent data highlights that the crude product can be easily purified to exceptional standards, with HPLC analysis confirming purity levels of 99.9% after simple recrystallization. This high level of chemical fidelity is crucial for pharmaceutical applications, where strict limits on genotoxic impurities and residual metals must be maintained. The robust nature of the catalytic cycle ensures consistent batch-to-batch reproducibility, a key metric for quality assurance teams. By controlling the stoichiometry of the base and the choice of ligand, manufacturers can further suppress side reactions, ensuring that the final impurity profile meets the rigorous specifications required for global regulatory submissions.
How to Synthesize Letrozole Intermediate Efficiently
Implementing this synthesis requires careful attention to the selection of catalysts, bases, and solvents to maximize yield and minimize costs. The patent outlines a versatile protocol where 2-(4-cyanophenyl)formylcarboxylic acid and 4-halobenzonitrile are combined in a molar ratio of approximately 1:1, though slight excesses of the halide can drive conversion. The reaction medium typically employs polar aprotic solvents such as N,N-dimethylformamide (DMF), 1,4-dioxane, or dimethyl sulfoxide (DMSO), which facilitate the solubility of the ionic intermediates. Essential to the process is the use of a strong base, with cesium carbonate, potassium phosphate, or sodium tert-butoxide proving highly effective in deprotonating the acid and activating the catalytic cycle. The detailed standardized synthesis steps for optimizing this reaction are provided in the guide below.
- Charge a reactor with 2-(4-cyanophenyl)formylcarboxylic acid and 4-halobenzonitrile in a polar aprotic solvent such as DMF or dioxane.
- Add a base system (e.g., cesium carbonate or potassium phosphate) along with a Pd/Cu catalyst pair and nitrogen ligands under inert atmosphere.
- Heat the mixture to 80-140°C for 5-24 hours, then isolate the product via aqueous workup and recrystallization to achieve >90% yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain; by reducing a multi-step sequence to a single transformation, the number of raw materials required is significantly lowered, thereby reducing the risk of supply disruptions associated with sourcing multiple specialized intermediates. Furthermore, the elimination of hazardous reagents like cuprous cyanide and cryogenic reagents like n-butyllithium removes the need for specialized containment equipment and expensive waste disposal protocols. This shift not only lowers the capital expenditure required for plant infrastructure but also reduces the ongoing operational costs related to environmental compliance and worker safety. The high yield reported in the patent examples suggests a substantial reduction in raw material consumption per kilogram of finished product, directly enhancing the gross margin potential for manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the consolidation of multiple reaction steps into one, which inherently reduces labor, energy, and solvent consumption. By avoiding the low-yielding steps characteristic of older routes, the overall material throughput is optimized, meaning less starting material is wasted to reach the same output volume. Additionally, the use of earth-abundant copper alongside palladium allows for potential catalyst loading optimizations that can further drive down reagent costs. The simplified workup procedure, involving basic aqueous washes and recrystallization, minimizes the need for complex chromatographic separations, which are often the most expensive part of fine chemical purification. These factors combine to create a leaner, more cost-effective manufacturing model that is highly resilient to market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzonitriles and simple carboxylic acids ensures a stable and diverse supply base, mitigating the risk of single-source dependency. Unlike routes that depend on custom-synthesized ketones or unstable organolithium reagents, the starting materials for this process are widely available from multiple global vendors. This abundance fosters competitive pricing and ensures continuity of supply even during periods of market volatility. Moreover, the robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or safety incidents, providing a predictable delivery timeline for downstream API manufacturers. This reliability is paramount for maintaining the inventory levels required to support global pharmaceutical distribution networks.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns perfectly with the principles of green chemistry by minimizing waste generation and avoiding toxic heavy metals. The absence of cuprous cyanide eliminates the generation of cyanide-containing wastewater, a major liability in chemical manufacturing. The high atom economy of the decarboxylative coupling ensures that the majority of the reactant mass ends up in the final product, reducing the volume of organic waste that requires incineration or treatment. Scalability is further supported by the use of standard heating and stirring equipment, allowing for seamless transfer from pilot plants to multi-ton reactors. This ease of scale-up ensures that manufacturers can rapidly respond to increased market demand for letrozole without the need for extensive process re-engineering or new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. The answers reflect the balance between theoretical potential and practical operational realities observed in the laboratory examples.
Q: How does this new route compare to the traditional benzophenone reduction method in terms of safety?
A: Unlike traditional routes that may involve hazardous reagents like cuprous cyanide or extreme cryogenic conditions with n-butyllithium, this novel method utilizes a safer Pd-Cu catalytic system at moderate temperatures, significantly reducing operational risks and waste treatment costs.
Q: What is the expected purity profile of the intermediate produced via this method?
A: The patent data indicates that the resulting intermediate can be purified to extremely high standards, with HPLC analysis showing purity levels reaching 99.9% after simple recrystallization, which is critical for meeting stringent regulatory requirements for API manufacturing.
Q: Is the catalyst system scalable for industrial production volumes?
A: Yes, the process avoids complex multi-step sequences and uses commercially available solvents and bases. The robust nature of the Pd-Cu dual catalytic cycle allows for straightforward scale-up from laboratory grams to multi-ton commercial batches without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Letrozole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology for the global oncology market. As a leading CDMO partner, we possess the technical expertise and infrastructure necessary to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is smooth and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of letrozole intermediate meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific solvent and catalyst requirements of this Pd-Cu catalyzed process safely and effectively.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and sustainable route for their letrozole supply needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how this technology can optimize your supply chain. Together, we can ensure a reliable, high-quality supply of this critical cancer medication intermediate, driving better health outcomes while maximizing operational efficiency.
