Scalable Copper-Catalyzed Synthesis of N-Substituted Tetrahydropyridine Boronate Esters for Pharma
Introduction to Advanced Tetrahydropyridine Boronate Synthesis
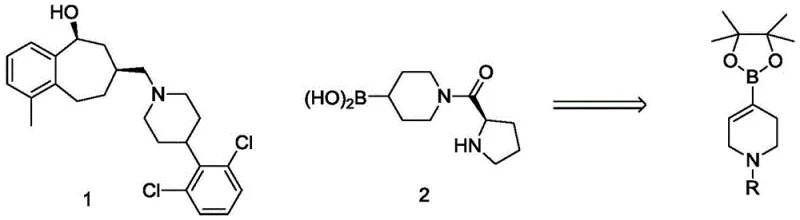
The pharmaceutical industry continuously seeks robust and cost-effective pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical intermediates for bioactive molecules. Patent CN109762007B introduces a groundbreaking process for synthesizing N-substituted-1,2,5,6-tetrahydropyridine-4-boronic acid esters, a structural motif frequently found in potent drug candidates such as opioid receptor antagonists and fatty acid amide hydrolase inhibitors. This innovation addresses long-standing challenges in the field by replacing expensive and hazardous traditional methods with a mild, copper-catalyzed protocol. The significance of this technology lies in its ability to deliver high-purity intermediates through a streamlined one-step reaction, thereby enhancing the efficiency of downstream drug development pipelines. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is crucial for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted-1,2,5,6-tetrahydropyridine-4-boronate esters has relied heavily on two primary methodologies, both of which present significant drawbacks for large-scale manufacturing. The first conventional approach utilizes palladium-catalyzed coupling reactions starting from expensive N-substituted-1,2,5,6-tetrahydropyridine-4-trifluoromethanesulfonates. This method not only incurs high raw material costs due to the triflate precursor but also introduces the complexity of removing trace palladium residues, which is a stringent requirement for pharmaceutical grade materials. The second traditional method involves a two-step sequence starting from N-substituted-4-piperidone, utilizing highly reactive Grignard reagents like isopropyl magnesium chloride-lithium chloride. This pathway necessitates harsh low-temperature conditions to control reactivity, posing safety risks and increasing energy consumption, while often resulting in lower yields and purity profiles that complicate subsequent purification steps.
The Novel Approach
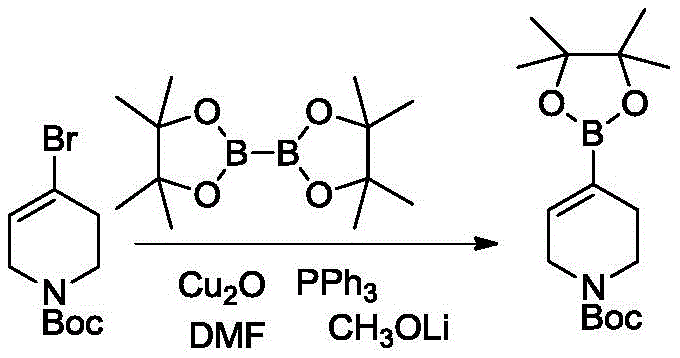
In stark contrast, the novel process disclosed in the patent utilizes a direct copper-catalyzed borylation of N-substituted-1,2,5,6-tetrahydropyridine-4-halides. This method represents a paradigm shift by employing inexpensive cuprous oxide or cuprous halides as catalysts instead of precious metals. The reaction proceeds under remarkably mild conditions, typically between 30-60°C, eliminating the need for cryogenic cooling and reducing operational hazards. By using readily available halide starting materials and diboron reagents, the process shortens the synthetic route to a single step, drastically improving overall atom economy. This streamlined approach not only simplifies the operational workflow but also ensures consistent product quality, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Copper-Catalyzed Borylation
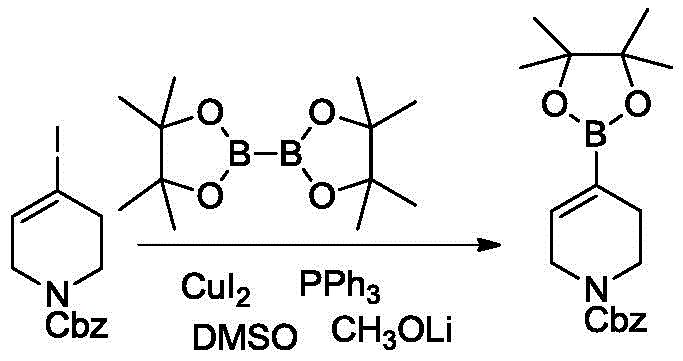
The core of this innovative synthesis lies in the efficient activation of the carbon-halogen bond by the copper catalyst system. The reaction mechanism involves the oxidative addition of the N-substituted-1,2,5,6-tetrahydropyridine-4-halide to the copper(I) species, facilitated by phosphine ligands such as triphenylphosphine or dppp. This forms an organocopper intermediate which subsequently undergoes transmetallation with the diboron reagent, such as pinacol diboron or neopentyl glycol diborate. The presence of an organic base, specifically lithium methoxide or lithium tert-butoxide, plays a critical role in activating the diboron species and facilitating the final reductive elimination step to release the desired boronate ester. This catalytic cycle is highly selective, minimizing side reactions such as homocoupling or dehalogenation, which are common pitfalls in transition metal catalysis.
Furthermore, the choice of solvent and reaction parameters significantly influences the impurity profile and overall yield. The patent specifies the use of polar aprotic solvents like DMF or DMSO, which effectively solubilize the inorganic base and copper catalyst while stabilizing the reactive intermediates. The mild thermal conditions prevent the decomposition of the sensitive tetrahydropyridine ring and the protecting groups (Boc, Cbz, Benzyl), ensuring the structural integrity of the final product. This precise control over reaction dynamics allows for the achievement of purity levels exceeding 99% as determined by HPLC and NMR analysis. Such high purity is essential for reducing the burden on downstream purification processes, thereby enhancing the commercial viability of the synthesis for complex pharmaceutical intermediates.
How to Synthesize N-Substituted Tetrahydropyridine Boronate Efficiently
The practical implementation of this copper-catalyzed borylation requires careful attention to reagent stoichiometry and atmospheric conditions to maximize yield and purity. The process begins with the preparation of a reaction mixture containing the halide substrate, diboron reagent, copper catalyst, ligand, and base in a suitable solvent under an inert nitrogen atmosphere. Maintaining the temperature within the optimal range of 45-55°C is critical for balancing reaction rate and selectivity. Upon completion, a straightforward workup involving filtration through celite to remove copper residues, followed by aqueous washing and solvent evaporation, yields the crude product. Final purification is achieved through a simple slurry process using a mixed solvent system of heptane and ethanol, which effectively removes remaining impurities without the need for column chromatography. For detailed standardized operating procedures, please refer to the guide below.
- Dissolve N-Boc-1,2,5,6-tetrahydropyridine-4-bromide, pinacol diboron, cuprous oxide, triphenylphosphine, and lithium methoxide in DMF under nitrogen protection.
- Stir the reaction mixture at a controlled temperature between 45-55°C until GC assay indicates completion.
- Filter through celite, wash with toluene, dilute, wash with water and brine, evaporate solvent, and slurry in heptane/ethanol to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology offers substantial strategic benefits beyond mere technical feasibility. The shift from palladium to copper catalysts represents a direct and significant reduction in raw material costs, as copper salts are orders of magnitude cheaper than palladium complexes. Additionally, the elimination of cryogenic conditions and the simplification of the synthetic route from two steps to one step drastically reduce energy consumption and processing time. These factors collectively contribute to a more lean and efficient manufacturing process, allowing for better margin management in a competitive market environment. The robustness of the method also implies fewer batch failures and more predictable production schedules, which is vital for maintaining supply continuity.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with economical copper sources directly lowers the bill of materials. Furthermore, the avoidance of specialized low-temperature equipment and the reduction in reaction steps minimize utility costs and labor hours. The high purity of the crude product reduces the need for extensive and costly purification steps like preparative HPLC, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including N-substituted tetrahydropyridine halides and pinacol diboron, are commercially available and stable, reducing the risk of supply disruptions associated with exotic or unstable reagents. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors, facilitating easier technology transfer between manufacturing sites. This flexibility ensures a resilient supply chain capable of meeting fluctuating demand without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and lack of hazardous reagents like Grignard solutions. The reduced use of heavy metals aligns with increasingly stringent environmental regulations regarding metal residues in APIs. The simplified waste stream, devoid of complex organometallic byproducts, eases the burden on waste treatment facilities, supporting sustainable manufacturing practices and reducing the environmental footprint of chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of N-substituted-1,2,5,6-tetrahydropyridine-4-boronic acid esters. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers provided reflect the consensus on the advantages of copper catalysis over traditional methods in terms of cost, safety, and product quality.
Q: Why is the copper-catalyzed method preferred over palladium catalysis for this synthesis?
A: The copper-catalyzed method significantly reduces raw material costs as copper salts are substantially cheaper than palladium catalysts. Furthermore, it eliminates the risk of heavy metal contamination often associated with palladium, simplifying the purification process and ensuring higher product purity suitable for pharmaceutical applications.
Q: What purity levels can be achieved with this novel process?
A: This process consistently achieves HPLC and NMR purity levels exceeding 99%. The mild reaction conditions and efficient workup procedure, involving filtration and slurry purification, effectively remove impurities and residual solvents without requiring complex chromatographic separation.
Q: Is this method scalable for industrial production?
A: Yes, the method is highly scalable. It operates at mild temperatures (30-60°C) avoiding cryogenic conditions, uses readily available reagents, and involves a simple one-step reaction sequence. These factors make it ideal for commercial scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Tetrahydropyridine Boronate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic intermediates positions us as a strategic partner for global pharmaceutical companies seeking reliable and high-quality supply solutions.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
