Scalable Production of High-Purity Benzofuranone via Solid Acid Catalysis for Global Agrochemical Supply Chains
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of critical intermediates, with benzofuranone standing out as a pivotal building block for the widely used fungicide azoxystrobin. Patent CN102040572A introduces a transformative production method that shifts the paradigm from traditional corrosive liquid acid catalysis to a more sustainable and efficient solid acid catalytic system. This technological breakthrough utilizes commercial cationic resins to facilitate the cyclization of o-hydroxyphenylacetic acid, achieving superior reaction control while mitigating the severe equipment corrosion and environmental burdens associated with conventional sulfuric or p-toluenesulfonic acid processes. By leveraging azeotropic dehydration techniques in conjunction with macroreticular ion exchange resins, this method delivers benzofuranone with exceptional purity levels exceeding 96%, addressing the stringent quality requirements of modern pharmaceutical and agrochemical supply chains. The implications of this patent extend beyond mere chemical transformation, offering a robust framework for cost-effective manufacturing that aligns with increasingly rigorous global environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzofuranone has relied heavily on the use of strong liquid acid catalysts such as concentrated sulfuric acid, p-toluenesulfonic acid, or phosphorus-based dehydrating agents, which present significant operational and economic challenges for large-scale manufacturers. These traditional protocols often necessitate harsh reaction conditions that induce severe corrosion on stainless steel reactors and piping, leading to substantial capital expenditure on specialized corrosion-resistant equipment and frequent maintenance downtime. Furthermore, the post-reaction workup in these liquid acid systems is notoriously cumbersome, requiring extensive aqueous washing steps to neutralize and remove residual acid, which generates vast quantities of acidic wastewater that demand complex and costly treatment before discharge. The difficulty in separating the homogeneous liquid catalyst from the product mixture also results in product loss and complicates the purification process, often leading to lower overall yields and inconsistent batch-to-batch quality that can jeopardize downstream synthesis of sensitive active ingredients like azoxystrobin.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent employs a heterogeneous cationic resin catalyst that fundamentally alters the reaction landscape by combining high catalytic activity with exceptional ease of separation. This solid acid system operates effectively within a moderate temperature range of 60-150°C using common organic solvents like toluene or chlorobenzene to drive the equilibrium forward through continuous azeotropic removal of water. The heterogeneity of the catalyst allows for a simple filtration or centrifugation step to recover the solid resin, which can be regenerated and reused multiple times, thereby drastically reducing raw material consumption and waste generation. This streamlined process not only eliminates the need for corrosive acid neutralization and extensive washing but also ensures that the final product is obtained with high concentration and minimal impurities, representing a significant leap forward in green chemistry and process efficiency for fine chemical manufacturing.
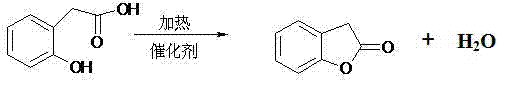
Mechanistic Insights into Cationic Resin-Catalyzed Cyclization
The core of this innovative synthesis lies in the unique mechanistic pathway facilitated by the sulfonic acid groups anchored on the macroreticular polymer matrix of the cationic resin, which act as solid proton donors to activate the carboxylic acid moiety of the substrate. Upon heating, the protons from the resin's sulfonic acid sites protonate the carbonyl oxygen of the o-hydroxyphenylacetic acid, increasing the electrophilicity of the carbonyl carbon and rendering it susceptible to nucleophilic attack by the adjacent phenolic hydroxyl group. This intramolecular nucleophilic acyl substitution proceeds through a tetrahedral intermediate, which subsequently collapses to eliminate a molecule of water, forming the stable five-membered lactone ring characteristic of benzofuranone. The porous structure of the macroreticular resin provides a high surface area for this interaction, ensuring efficient mass transfer while the hydrophobic nature of the polymer backbone helps to repel the generated water, shifting the equilibrium towards product formation in accordance with Le Chatelier's principle during the azeotropic distillation process.
From an impurity control perspective, the use of a solid acid catalyst offers distinct advantages over liquid acids by minimizing side reactions such as polymerization, charring, or over-oxidation that are commonly observed under the aggressive conditions of homogeneous acid catalysis. The mild acidity and controlled microenvironment within the resin pores prevent the degradation of the sensitive phenolic substrate, resulting in a cleaner reaction profile with fewer by-products that could otherwise complicate downstream purification or affect the efficacy of the final agrochemical product. Additionally, the ability to precisely control the catalyst loading between 0.1% and 20% allows process chemists to fine-tune the reaction kinetics, balancing reaction rate with selectivity to ensure consistent high-quality output. This level of control is critical for maintaining the stringent purity specifications required for intermediates destined for biological applications, where trace impurities can have profound effects on the safety and performance of the final active pharmaceutical or agrochemical ingredient.
How to Synthesize Benzofuranone Efficiently
The synthesis of benzofuranone via this resin-catalyzed route represents a highly practical and scalable methodology that can be readily implemented in existing multipurpose chemical plants with minimal modification to standard infrastructure. The process begins with the charging of o-hydroxyphenylacetic acid and a selected solvent into a reactor equipped with a water separator, followed by the addition of the dried cationic resin catalyst to initiate the cyclization upon heating to reflux. As the reaction progresses, water is continuously removed as an azeotrope with the solvent, driving the conversion to completion typically within a few hours, after which the mixture is cooled and the solid catalyst is filtered off for regeneration. Detailed standardized synthetic steps see the guide below.
- Charge the reactor with o-hydroxyphenylacetic acid, a suitable solvent (such as toluene or chlorobenzene), and a catalytic amount of macroreticular cationic exchange resin.
- Heat the mixture to reflux (60-150°C) to facilitate azeotropic dehydration, removing water continuously via a Dean-Stark trap until reaction completion.
- Cool the reaction mixture, filter off the solid resin catalyst for reuse, and distill off the solvent to isolate high-purity benzofuranone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cationic resin-catalyzed process translates directly into tangible operational efficiencies and risk mitigation strategies that enhance the overall resilience of the supply chain. By eliminating the reliance on corrosive liquid acids, manufacturers can significantly extend the lifespan of their production assets, reducing the frequency of equipment replacement and the associated capital expenditures that often inflate the cost of goods sold in traditional chemical manufacturing. The simplified workup procedure, which bypasses the need for extensive aqueous washing and neutralization, leads to a drastic reduction in utility consumption, particularly water and energy, while simultaneously lowering the volume of hazardous waste that requires disposal, thereby aligning production with sustainability goals and reducing regulatory compliance costs.
- Cost Reduction in Manufacturing: The implementation of a reusable solid catalyst system fundamentally alters the cost structure of benzofuranone production by removing the recurring expense of purchasing stoichiometric or excess amounts of liquid acid catalysts for every batch. Since the cationic resin can be recovered via simple filtration and reused multiple times without significant loss of activity, the effective cost per kilogram of catalyst consumed is negligible compared to traditional methods, leading to substantial long-term savings. Furthermore, the elimination of corrosion-related maintenance and the reduction in wastewater treatment fees contribute to a leaner operating model, allowing suppliers to offer more competitive pricing structures without compromising on margin or quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by reducing the complexity of the manufacturing process, which in turn minimizes the risk of production delays caused by equipment failure or waste disposal bottlenecks. The use of commercially available and stable cationic resins ensures a consistent supply of catalytic material, avoiding the volatility often associated with the sourcing of specialized liquid reagents. Additionally, the high yield and purity achieved in a single pass reduce the need for complex recrystallization or distillation steps, shortening the overall cycle time from raw material intake to finished goods readiness and enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more straightforward due to the absence of highly corrosive reagents and the exothermic risks associated with mixing strong liquid acids. The green chemistry attributes of this method, specifically the reduction in hazardous waste and the potential for solvent recycling, position manufacturers favorably against tightening environmental regulations, ensuring long-term operational continuity without the threat of shutdowns due to non-compliance. This environmental stewardship not only safeguards the license to operate but also appeals to downstream customers who are increasingly prioritizing sustainable sourcing practices in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of benzofuranone using the cationic resin catalytic method described in patent CN102040572A. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation to provide clarity on process feasibility and advantages. Understanding these details is crucial for technical teams evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using cationic resin over liquid acids for benzofuranone synthesis?
A: Using cationic resin eliminates equipment corrosion caused by strong liquid acids like sulfuric acid, simplifies product purification by avoiding aqueous washing steps, and allows for catalyst recovery and reuse, significantly reducing waste generation.
Q: What is the typical purity and yield achievable with this resin-catalyzed method?
A: According to patent data, this method consistently achieves product content above 96% with high yields (often exceeding 97%), making it suitable for demanding agrochemical applications like azoxystrobin production.
Q: Can the cationic resin catalyst be recycled in this process?
A: Yes, the solid nature of the cationic resin allows it to be easily separated from the reaction mixture via filtration or centrifugation, enabling multiple reuse cycles which lowers overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like benzofuranone play in the global agrochemical and pharmaceutical industries, and we are committed to delivering this essential building block with unmatched consistency and reliability. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzofuranone meets the exacting standards necessary for the synthesis of high-value end products like azoxystrobin.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs and drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized production methods can reduce your overall input costs. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your supply chain requirements.
