Optimizing Tigecycline Intermediate Production via Novel Regioselective Nitration and Reductive Methylation
Optimizing Tigecycline Intermediate Production via Novel Regioselective Nitration and Reductive Methylation
The global demand for next-generation antibiotics continues to drive innovation in pharmaceutical intermediate synthesis, particularly for complex molecules like Tigecycline. Patent CN101955443B, filed in 2012, discloses a groundbreaking method for preparing key tigecycline intermediates that fundamentally alters the traditional synthetic landscape. This technology addresses critical bottlenecks in the supply chain by eliminating the dependency on expensive minocycline precursors and solving long-standing regioselectivity issues in nitration reactions. For R&D directors and procurement specialists, understanding this pathway is essential for securing a stable supply of high-purity glycylcycline antibiotics.
Tigecycline, chemically known as [4S-(4α,12aα)]-4,7-bis(dimethylamino)-9-[(tert-butylamino)acetamido]-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-2-tetracenecarboxamide, represents a vital class of broad-spectrum antibiotics effective against resistant bacteria including MRSA. The structural complexity of this molecule requires precise functionalization at the 7 and 9 positions of the tetracycline core. The patent outlines a superior route that begins with 6-demethyl-6-deoxytetracycline, offering a more direct and economically viable entry point compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
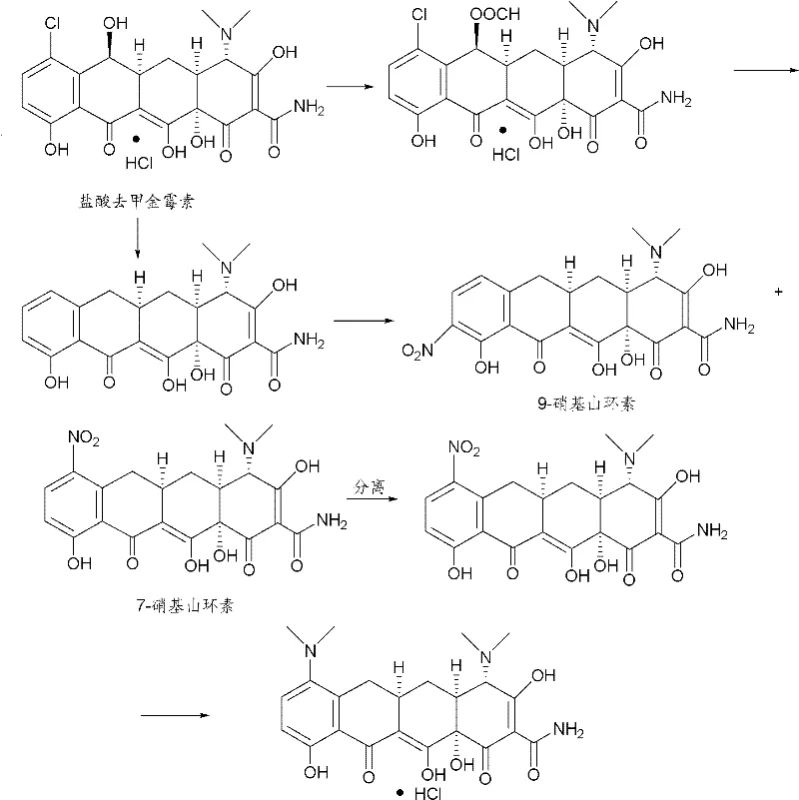
Historically, the industrial synthesis of tigecycline has relied heavily on minocycline as the starting material. As detailed in prior art such as Bioorg. Med. Chem. Lett. and US patents, this approach involves nitration, reduction, and acylation at the 9-position of minocycline. However, the production of minocycline itself is fraught with challenges. The conventional synthesis of minocycline from sancycline involves a nitration step that produces a troublesome mixture of 7-nitro and 9-nitro isomers. Separating these isomers is technically demanding, requires extensive chromatography or recrystallization, and results in significant yield losses. Furthermore, the 4-position epimerization during these harsh chemical transformations introduces difficult-to-remove impurities that compromise the final API quality.
Additionally, relying on minocycline as a feedstock ties the tigecycline supply chain to the volatility of the minocycline market. Any disruption in minocycline manufacturing directly impacts tigecycline availability. The multi-step nature of converting minocycline to tigecycline also accumulates costs, as each isolation and purification stage adds operational expenditure and waste generation. For a procurement manager, these inefficiencies translate into higher unit costs and longer lead times for critical antibiotic intermediates.
The Novel Approach
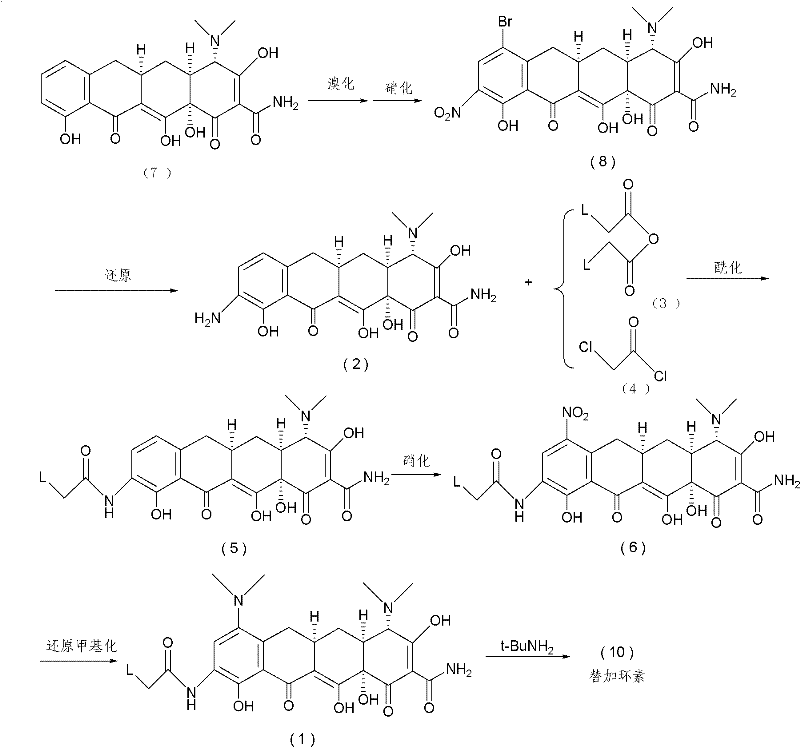
The methodology described in CN101955443B circumvents these issues by reversing the order of functionalization. Instead of starting with minocycline, the process begins with 9-amino-6-demethyl-6-deoxytetracycline, which is derived from the more abundant sancycline. The key innovation lies in protecting the 9-amino group via acylation with a haloacetic anhydride or halide before attempting nitration. By blocking the 9-position with a haloacetamide group, the subsequent nitration reaction is forced to occur exclusively at the 7-position. This regioselective control completely eliminates the formation of the 9-nitro impurity, thereby removing the need for difficult isomer separation.
Following the selective nitration, the process employs a one-pot reductive methylation using Pd/C, formaldehyde, and hydrogen. This single step simultaneously reduces the 7-nitro group to an amine and methylates it to form the 7-dimethylamino group characteristic of the minocycline core. Consequently, the desired 9-haloacetamido minocycline intermediate is generated without ever isolating free minocycline. This telescoped approach not only simplifies the equipment requirements but also drastically improves the overall yield and purity profile of the intermediate.
Mechanistic Insights into Regioselective Nitration and Reductive Methylation
The success of this synthetic strategy hinges on the precise manipulation of electronic effects within the tetracycline ring system. In the acylation step (Step A), the 9-amino group reacts with reagents like chloroacetyl chloride or bromoacetic anhydride in the presence of organic bases such as triethylamine. This reaction is conducted at low temperatures, typically between -15°C and -25°C, to prevent degradation of the sensitive tetracycline backbone. The resulting 9-haloacetamide group acts as a robust protecting group that withstands the harsh acidic conditions of the subsequent nitration step.
In Step B, the nitration is performed using a mixture of nitric acid and sulfuric acid, or nitrates in sulfuric acid, at temperatures ranging from -40°C to -10°C. The presence of the electron-withdrawing haloacetamide group at the 9-position deactivates that site towards electrophilic aromatic substitution. Consequently, the nitronium ion attacks the activated 7-position with high specificity. The addition of proton transfer reagents like DMF further enhances the efficiency of this transformation. This mechanistic control ensures that the crude product contains minimal regioisomeric impurities, simplifying downstream processing significantly.
The final transformation (Step C) utilizes a classic Eschweiler-Clarke type reductive methylation mechanism adapted for heterogeneous catalysis. Under hydrogen pressure of 1 to 4 atm and temperatures of 5 to 40°C, the Pd/C catalyst facilitates the reduction of the 7-nitro group to a primary amine. In the presence of excess formaldehyde, this amine is rapidly converted to the secondary and then tertiary dimethylamine. This cascade reaction is highly efficient, converting the nitro precursor directly into the 7-dimethylamino derivative found in minocycline structures, all while maintaining the integrity of the 9-side chain.
How to Synthesize 9-Haloacetamido Minocycline Efficiently
The synthesis of this critical intermediate involves a carefully orchestrated sequence of protection, functionalization, and reduction. Operators must maintain strict temperature control during the acylation and nitration phases to ensure safety and selectivity. The use of solvents like dichloromethane for acylation and sulfuric acid for nitration requires appropriate corrosion-resistant equipment. Following the reaction, the product is typically isolated by precipitation into non-solvents like methyl tert-butyl ether or hexane/isopropanol mixtures, yielding a high-purity solid suitable for the final coupling with tert-butylamine.
- Acylation of 9-amino-6-demethyl-6-deoxytetracycline with haloacetic anhydride or halide at low temperatures (-15 to -25°C) to protect the 9-position.
- Regioselective nitration of the protected intermediate using HNO3/H2SO4 at -40 to -10°C to introduce the nitro group exclusively at the 7-position.
- Catalytic reductive methylation using Pd/C, formaldehyde, and hydrogen pressure (1-4 atm) to convert the 7-nitro group into the 7-dimethylamino group.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this patented process offers substantial strategic benefits beyond mere technical elegance. By decoupling tigecycline production from the minocycline supply chain, manufacturers can mitigate risks associated with raw material shortages. The ability to source 6-demethyl-6-deoxytetracycline (sancycline) directly from fermentation streams provides a more stable and cost-effective foundation for production. This vertical integration potential allows for better margin control and insulation from market fluctuations affecting semi-synthetic tetracyclines.
- Cost Reduction in Manufacturing: The elimination of the minocycline isolation step removes a significant cost center from the production ledger. Traditional methods require the purification of minocycline, which involves solvent-intensive crystallization processes to remove 9-nitro impurities. By bypassing this entirely, the new route reduces solvent consumption, energy usage for drying and distillation, and labor hours associated with quality control testing of the intermediate. Furthermore, the higher yields reported in each step of this novel pathway mean that less starting material is required to produce the same amount of final API, directly lowering the cost of goods sold.
- Enhanced Supply Chain Reliability: Relying on sancycline, a precursor closer to the fermentation origin, shortens the chemical supply chain. This reduces the number of external vendors involved and minimizes the logistical complexity of transporting hazardous intermediates between different facilities. The robustness of the nitration step, which no longer produces difficult-to-separate isomers, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as low-temperature stirring, filtration, and hydrogenation. The avoidance of complex chromatographic separations makes the process easier to scale from pilot plant to commercial tonnage. Additionally, the improved atom economy and reduced solvent waste contribute to a lower environmental footprint. This aligns with increasingly stringent environmental regulations and supports sustainability goals, making the manufacturing process more resilient to regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations for potential partners.
Q: Why does this new route avoid the formation of ortho/para nitration mixtures?
A: By acylating the 9-amino group prior to nitration, the 9-position is sterically and electronically blocked. This forces the nitration reaction to occur selectively at the 7-position, eliminating the difficult separation of 7-nitro and 9-nitro isomers common in traditional minocycline synthesis.
Q: What are the cost advantages of starting from 6-demethyl-6-deoxytetracycline instead of minocycline?
A: 6-Demethyl-6-deoxytetracycline (Sancycline) is a more upstream and readily available fermentation derivative compared to minocycline. Bypassing the isolation and purification of minocycline significantly reduces raw material costs and processing time.
Q: Is the reductive methylation step scalable for industrial production?
A: Yes, the process utilizes standard heterogeneous catalysis (Pd/C) under moderate hydrogen pressure (1-4 atm) and temperature (5-40°C), making it highly suitable for large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tigecycline Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the technology disclosed in CN101955443B and possesses the technical capability to implement this advanced route for the commercial production of tigecycline intermediates. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade antibiotic intermediates.
We invite you to collaborate with us to leverage this innovative chemistry for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and reduce total landed costs.
