Optimizing Thio-NAD Production: A Chemical Route for Commercial Scale-up and Supply Reliability
Optimizing Thio-NAD Production: A Chemical Route for Commercial Scale-up and Supply Reliability
The landscape of biochemical reagent manufacturing is undergoing a significant transformation, driven by the need for more robust and cost-effective synthetic pathways for complex coenzyme analogs. Patent CN102477057A introduces a groundbreaking full-chemical preparation method for Thionicotinamide adenine dinucleotide (Thio-NAD), a critical component in diagnostic reagent kits for bile acid determination. This innovation marks a decisive shift away from traditional biosynthetic methods that rely on unstable and expensive enzymatic catalysts, offering a pathway that is inherently more suitable for industrial scale-up. By establishing a purely chemical route, the process mitigates the risks associated with biological variability and enzyme procurement, ensuring a more predictable supply chain for high-purity Thio-NAD. This technical advancement is particularly relevant for procurement managers and R&D directors seeking to secure reliable diagnostic reagent supplier partnerships that can guarantee long-term continuity and cost efficiency. The ability to synthesize this complex molecule without biological steps represents a substantial leap forward in process chemistry, enabling manufacturers to achieve stringent purity specifications while drastically simplifying the production workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Thionicotinamide adenine dinucleotide has been heavily reliant on biosynthetic means or semi-synthetic routes that incorporate enzymatic steps, which present significant bottlenecks for commercial scalability. Traditional methods often utilize Nicotinamide-NAD as a starting material, employing specific glucosyltransferases from pig brains to modify the vitamin PP moiety, a process that is fraught with logistical and economic challenges. The reliance on biological enzymes necessitates specialized equipment and controlled environments that are difficult to maintain in large-scale chemical plants, leading to higher operational expenditures and complex regulatory hurdles. Furthermore, the post-treatment of enzymatic reactions carries a high risk of product enzymolysis if processing conditions are not meticulously managed, resulting in inconsistent yields and compromised product quality. The cost of raw materials, particularly the specialized enzymes and the NAD precursor, is prohibitively high, making the final product expensive and limiting its accessibility for widespread diagnostic applications. These inherent limitations of the conventional biosynthetic approach create a fragile supply chain that is vulnerable to disruptions and incapable of meeting the growing demand for cost reduction in biochemical manufacturing.
The Novel Approach
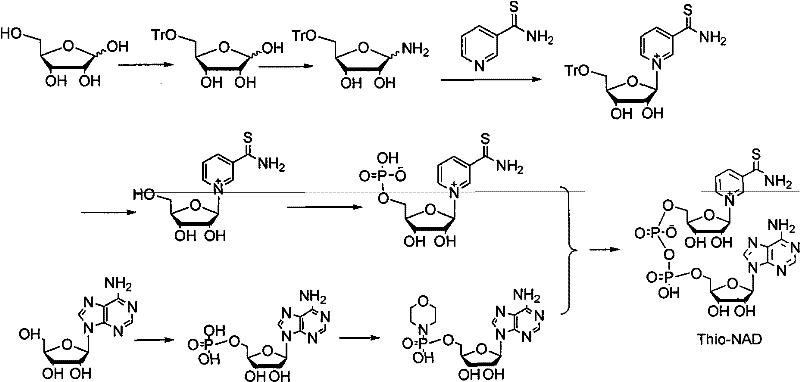
In stark contrast to the fragile biosynthetic legacy, the novel chemical synthesis route detailed in the patent data employs a convergent strategy that fundamentally restructures the production logic for Thio-NAD. This method bypasses the need for any biological enzymes entirely, utilizing readily available chemical reagents that can be sourced from major reagent companies without the lead time constraints associated with specialized biological materials. The core of this innovation lies in the design of a convergent synthetic route that constructs the final dinucleotide from two distinct, stable intermediates, thereby avoiding the risks inherent in long linear synthetic sequences. By decoupling the synthesis of the thio-nicotinamide moiety and the adenosine moiety, the process allows for independent optimization of each branch, leading to better impurity control and higher overall efficiency. This approach not only simplifies the purification process but also enhances the robustness of the reaction conditions, making it far more amenable to commercial scale-up of complex coenzyme analogs. The transition to this all-chemical methodology represents a strategic advantage for supply chain heads, as it ensures that production is not held hostage by the availability of proprietary enzymes or biological feedstocks.
Mechanistic Insights into Convergent Chemical Synthesis
The mechanistic elegance of this synthesis lies in the strategic formation and utilization of two key stable intermediates, which serve as the building blocks for the final dinucleotide assembly. The first critical intermediate, Thio-nicotinamide-α-D-nucleoside monophosphate, is constructed through a series of protection, amination, and phosphorylation steps that ensure the stereochemical integrity of the ribose sugar. The use of trityl or p-methoxyphenyl methyl groups as protecting bases for the 5-position of D-ribose allows for precise control over the reaction sites, preventing unwanted side reactions during the subsequent coupling with thionicotinamide. This level of molecular precision is essential for achieving the high-purity Thio-NAD required for sensitive diagnostic applications, where even minor impurities can interfere with enzymatic assays. The phosphorylation step, utilizing POCl3 and trimethyl phosphite, is carefully controlled at low temperatures to maximize the formation of the monophosphate species without degrading the sensitive nucleoside structure. This meticulous attention to reaction conditions underscores the feasibility of the process for industrial implementation, where reproducibility is paramount.
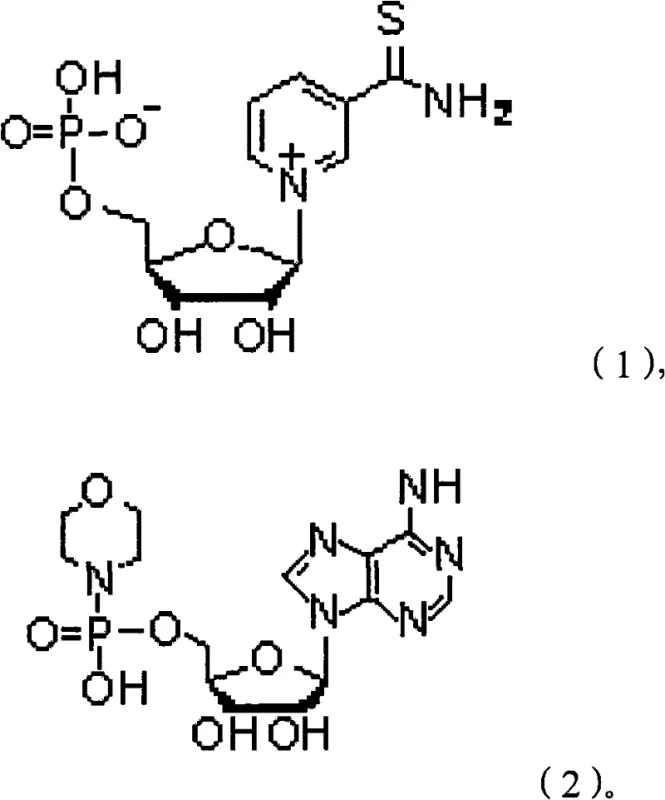
Parallel to the thio-nicotinamide branch, the synthesis of the second intermediate, Morpholine adenosine monophosphate, follows a similarly rigorous chemical pathway designed to activate the adenosine moiety for coupling. The activation of adenosine using POCl3 and trimethyl phosphite generates a reactive phosphate species that is subsequently stabilized by the introduction of morpholine, creating a robust intermediate that can withstand the conditions of the final coupling reaction. The use of DCC (dicyclohexylcarbodiimide) as a coupling agent in the presence of morpholine facilitates the formation of the phosphoramidate bond with high efficiency, minimizing the formation of byproducts that would complicate downstream purification. This dual-intermediate strategy effectively compartmentalizes the synthesis, allowing for the isolation and characterization of each half of the molecule before the final assembly, which significantly reduces the complexity of impurity profiling. For R&D directors, this mechanistic clarity provides confidence in the process's ability to consistently deliver material that meets stringent purity specifications, as the source of any potential impurities can be traced back to specific, controllable steps in the synthesis of the intermediates.
How to Synthesize Thionicotinamide Adenine Dinucleotide Efficiently
The operational execution of this synthesis route is designed to be straightforward yet precise, leveraging standard chemical engineering unit operations that are familiar to manufacturing teams. The process begins with the protection of D-ribose, followed by amination and coupling with thionicotinamide to form the nucleoside, which is then phosphorylated to yield the first key intermediate. Simultaneously, adenosine is phosphorylated and activated with morpholine to create the second intermediate, after which the two halves are coupled in the presence of manganese chloride and magnesium sulfate to form the final Thio-NAD molecule. The detailed standardized synthesis steps see the guide below, which outlines the specific reagent ratios, temperature controls, and purification techniques required to achieve the reported yields and purity levels. This structured approach ensures that the transition from laboratory scale to commercial production is seamless, with clear parameters for quality control at every stage of the manufacturing process.
- Preparation of Thio-nicotinamide-α-D-nucleoside monophosphate via trityl protection, amination, and phosphorylation.
- Synthesis of Morpholine adenosine monophosphate from adenosine using POCl3 and morpholine activation.
- Coupling of the two phosphorylated intermediates using manganese chloride and magnesium sulfate to form the final dinucleotide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this full-chemical synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring supply continuity. The elimination of biological enzymes from the process removes a significant variable cost driver, as chemical reagents are generally more stable, have longer shelf lives, and are available from multiple global suppliers, fostering a competitive pricing environment. This shift fundamentally alters the cost structure of Thio-NAD production, enabling substantial cost savings that can be passed down to the end-user or reinvested into further process optimization. Furthermore, the use of stable intermediates allows for a modular production strategy, where intermediates can be stockpiled to buffer against fluctuations in raw material availability, thereby enhancing supply chain reliability. The robustness of the chemical reactions also means that the process is less sensitive to minor variations in operating conditions, reducing the rate of batch failures and ensuring a consistent output of high-quality product. These factors combined create a resilient supply chain model that is capable of scaling to meet market demand without the bottlenecks associated with biological manufacturing.
- Cost Reduction in Manufacturing: The transition to a fully chemical process eliminates the need for expensive, specialized enzymes that are often proprietary and subject to supply constraints, leading to a drastic simplification of the raw material portfolio. By utilizing commodity chemicals such as D-ribose, adenosine, and standard phosphorylating agents, the manufacturing cost base is significantly lowered, allowing for more competitive pricing in the diagnostic reagent market. The removal of enzymatic steps also reduces the need for specialized containment and purification equipment required to handle biological materials, further decreasing capital and operational expenditures. This economic efficiency is achieved without compromising the quality of the final product, as the chemical route is capable of delivering purity levels that meet or exceed those of biosynthetic methods. Consequently, this represents a strategic opportunity for cost reduction in biochemical manufacturing that aligns with the financial goals of large-scale diagnostic producers.
- Enhanced Supply Chain Reliability: The reliance on commercially available chemical reagents ensures that the supply chain is not vulnerable to the disruptions that often affect the sourcing of biological enzymes, which may have limited suppliers or long lead times. The ability to source raw materials from multiple vendors creates a competitive procurement environment that mitigates the risk of supply shortages and price volatility. Additionally, the stability of the chemical intermediates allows for greater flexibility in production scheduling, as batches can be prepared in advance and stored without the degradation risks associated with biological precursors. This flexibility is crucial for maintaining reducing lead time for high-purity coenzyme analogs, ensuring that customers receive their orders promptly even during periods of high demand. The overall result is a more dependable supply chain that can support the continuous operation of diagnostic manufacturing facilities.
- Scalability and Environmental Compliance: The chemical synthesis route is inherently more scalable than biosynthetic methods, as it utilizes standard reactor configurations and purification techniques that are easily expanded from pilot to commercial scale. The process avoids the generation of biological waste streams that require complex treatment, simplifying environmental compliance and reducing the burden on waste management systems. The use of standard organic solvents and reagents allows for established recycling and recovery protocols, further enhancing the sustainability profile of the manufacturing process. This scalability ensures that the production capacity can be ramped up to meet growing market needs without the need for significant re-engineering of the process, supporting the commercial scale-up of complex coenzyme analogs. The combination of scalability and environmental efficiency makes this route an attractive option for manufacturers looking to expand their production capabilities responsibly.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial inquiries regarding the synthesis and application of Thionicotinamide adenine dinucleotide, based on the detailed patent specifications. These insights are intended to clarify the process advantages and assist decision-makers in evaluating the feasibility of adopting this chemical route for their supply chains. The responses are derived directly from the technical data provided in the patent documentation, ensuring accuracy and relevance for professional stakeholders. Understanding these details is critical for aligning procurement strategies with the technical capabilities of the manufacturing process.
Q: What are the advantages of the chemical synthesis method over enzymatic methods for Thio-NAD?
A: The chemical method avoids expensive biological enzymes and eliminates the risk of product enzymolysis during post-treatment, significantly reducing raw material costs and improving process stability for industrial production.
Q: What are the key stable intermediates in this synthesis route?
A: The process utilizes two stable intermediates: Thio-nicotinamide-α-D-nucleoside monophosphate and Morpholine adenosine monophosphate, which allow for a convergent synthesis strategy that improves overall yield and purity control.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the method uses commercially available chemical reagents instead of specialized enzymes, making it highly scalable and suitable for cost reduction in biochemical manufacturing with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thio-NAD Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging deep technical expertise to bring complex synthetic routes like the one described in CN102477057A to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial supply is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Thio-NAD meets the exacting standards required for diagnostic applications. Our capability to handle convergent synthesis strategies allows us to optimize production efficiency and deliver high-purity Thio-NAD with consistent quality, supporting the needs of global diagnostic reagent manufacturers. By partnering with us, clients gain access to a supply chain that is both robust and responsive, capable of adapting to changing market demands while maintaining the highest levels of product integrity.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific production requirements. We encourage you to request a Customized Cost-Saving Analysis that details the potential economic impact of switching to this chemical method for your supply chain. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver reliable diagnostic reagent supplier solutions. By collaborating with NINGBO INNO PHARMCHEM, you secure a partner dedicated to driving value through technical excellence and supply chain reliability, ensuring your diagnostic products remain competitive in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
