Advanced Nickel-Catalyzed Strategy for High-Purity 1,2-cis-Glycoside Pharmaceutical Intermediates
The construction of 1,2-cis-glycosidic linkages represents one of the most persistent and challenging frontiers in carbohydrate chemistry, particularly for the synthesis of biologically active oligosaccharides and glycoconjugates found in pharmaceutical applications. Traditional approaches often struggle with poor stereocontrol, requiring complex protecting group strategies or toxic reagents that hinder scalability. Addressing these critical bottlenecks, the patent CN112375108A discloses a groundbreaking method for selectively synthesizing 1,2-cis-glycoside compounds using a 1,2-unprotected glycosyl donor under mild conditions. This innovation leverages a Ni(II) or Fe(III) catalyst system combined with a non-nucleophilic organic base to achieve high stereoselectivity without the need for cumbersome chiral auxiliaries. By enabling the direct coupling of unprotected donors with trifluoromethanesulfonyl-substituted acceptors, this technology offers a streamlined pathway for producing high-purity pharmaceutical intermediates.
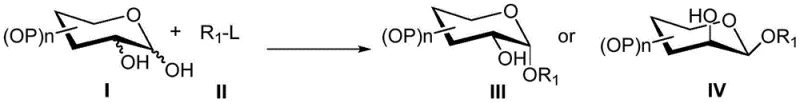
The significance of this development lies in its ability to bypass the thermodynamic preference for 1,2-trans products, which typically dominates glycosylation reactions due to the anomeric effect. By utilizing a coordination chemistry approach, the catalyst preferentially activates the C-1 oxygen atom of the upright bond, directing the nucleophilic attack of the acceptor to form the desired cis-configuration. This mechanistic insight not only solves a long-standing chemical problem but also provides a robust platform for the commercial scale-up of complex glycosides required in modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2-cis-glycosides, such as beta-mannosides, has relied on methodologies that are inherently inefficient and environmentally burdensome. For instance, the chiral prosthetic group process introduced by the Boons group requires the installation of bulky S-(phenylthiomethyl) benzyl ether groups at the C-2 position, leading to longer synthetic routes and poor atom economy. Similarly, aglycone transfer methods, while conceptually elegant, often suffer from the instability of acetal intermediates in acidic environments, resulting in low yields and difficult purification processes. Furthermore, traditional methods frequently employ stoichiometric amounts of toxic tin reagents, as seen in the anomeric carbyloxyalkyl process, which generates hazardous waste streams that are costly to treat and dispose of in a regulated manufacturing environment. These legacy techniques also often require harsh reaction conditions or cryogenic temperatures, limiting their applicability to sensitive substrates and increasing energy consumption during production.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalytic amount of inexpensive nickel or iron salts to drive the reaction with exceptional efficiency. As illustrated in the specific example below, the reaction proceeds smoothly at room temperature in common solvents like 1,2-dichloroethane (DCE), eliminating the need for energy-intensive heating or cooling cycles. The use of 1,2-unprotected donors drastically reduces the number of synthetic steps required to prepare the starting materials, thereby improving the overall throughput of the manufacturing process. This method demonstrates broad substrate tolerance, successfully coupling various glucose, galactose, and mannose-based donors with different acceptors to yield target compounds with high fidelity.
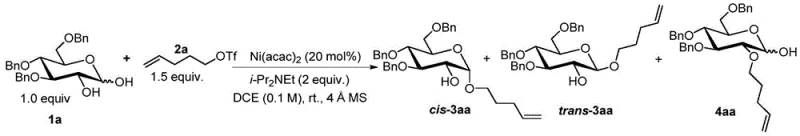
The operational simplicity of this new protocol allows for easier handling and safer processing, as it avoids the use of pyrophoric or highly toxic reagents. The high stereoselectivity observed, such as the 87% yield of cis-3aa in Example 1, ensures that downstream purification is simplified, reducing solvent usage and waste generation. This shift from stoichiometric toxic activators to catalytic green chemistry represents a paradigm shift in how complex carbohydrate intermediates can be manufactured reliably and sustainably.
Mechanistic Insights into Ni(II)-Catalyzed Glycosylation
The core of this technological breakthrough lies in the unique interaction between the transition metal catalyst and the glycosyl donor. Unlike traditional Lewis acid promoters that activate the leaving group indiscriminately, the Ni(II) catalyst, such as nickel diacetone, engages in a specific coordination with the oxygen atoms of the sugar ring. The electronic and steric properties of the ligand environment around the nickel center play a pivotal role in distinguishing between the axial and equatorial oxygen atoms at the anomeric center. The patent elucidates that the catalyst preferentially activates the C-1 oxygen atom of the upright bond (axial position), creating a transient electrophilic species that is sterically poised for backside attack by the acceptor. This specific activation mode overrides the inherent anomeric effect that usually favors the formation of 1,2-trans linkages, effectively flipping the stereochemical outcome to favor the 1,2-cis product.
Furthermore, the presence of the non-nucleophilic organic base, such as N,N-diisopropylethylamine (DIPEA), is crucial for scavenging the triflic acid generated during the activation of the triflate acceptor, preventing acid-catalyzed decomposition of the sensitive glycosidic bond. This dual-activation strategy ensures that the reaction proceeds through a clean SN2-like pathway, minimizing the formation of orthoester byproducts or hydrolysis impurities. The result is a reaction profile characterized by high regioselectivity and minimal side reactions, which is essential for meeting the stringent purity specifications required for active pharmaceutical ingredients (APIs). By controlling the coordination sphere of the metal, chemists can fine-tune the reactivity to accommodate a wide range of electron-rich and electron-deficient substrates without compromising selectivity.
How to Synthesize cis-3aa Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the rigorous drying of the glycosyl donor to prevent hydrolysis, followed by the sequential addition of the catalyst, base, and acceptor under an inert atmosphere. The reaction monitoring is easily performed via thin-layer chromatography (TLC), allowing for precise endpoint determination to maximize yield. For a comprehensive understanding of the operational parameters, including exact molar ratios and workup procedures, please refer to the standardized synthesis guide below.
- Azeotropically dry the glycosyl donor (1a) with toluene three times to remove moisture, then add Ni(acac)2 catalyst and 4 Å molecular sieves under argon protection.
- Add 1,2-dichloroethane (DCE) solvent and stir at room temperature, followed by the addition of N,N-diisopropylethylamine (i-Pr2NEt) base.
- Introduce the glycosyl acceptor (2a) to the mixture and stir at room temperature for 24 hours until TLC indicates completion.
- Filter the reaction mixture, concentrate under reduced pressure, and purify the crude product via column chromatography using ethyl acetate/petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this nickel-catalyzed methodology offers substantial advantages that directly impact the bottom line and supply chain resilience. By replacing expensive and toxic stoichiometric reagents with catalytic amounts of commodity nickel salts, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The elimination of tin waste not only lowers disposal costs but also simplifies regulatory compliance regarding heavy metal residues in the final product. Additionally, the shortened synthetic route for the sugar modules means fewer unit operations, less solvent consumption, and reduced labor hours, all of which contribute to a more lean and efficient production model.
- Cost Reduction in Manufacturing: The transition from stoichiometric activators to a catalytic system fundamentally alters the cost structure of glycoside production. Nickel acetylacetonate is a widely available industrial chemical with a fraction of the cost of specialized chiral auxiliaries or organotin compounds. This substitution eliminates the need for expensive reagent recovery systems and reduces the raw material bill of materials significantly. Furthermore, the high atom economy of the reaction minimizes waste generation, leading to lower environmental compliance costs and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents such as DIPEA, DCE, and simple nickel salts ensures a robust supply chain that is less susceptible to market volatility. Unlike specialized reagents that may have single-source suppliers or long lead times, the components of this catalytic system are commoditized and globally sourced. This availability guarantees continuity of supply for critical API intermediates, reducing the risk of production delays due to raw material shortages. The mild reaction conditions also allow for processing in a wider range of manufacturing facilities, increasing flexibility in sourcing partners.
- Scalability and Environmental Compliance: The mild operating conditions, typically ranging from room temperature to 60°C, make this process highly scalable from gram to multi-ton quantities without requiring specialized high-pressure or cryogenic equipment. The absence of toxic tin byproducts simplifies the wastewater treatment process, aligning with increasingly strict global environmental regulations. This environmental compatibility facilitates faster regulatory approval for new drug candidates and ensures long-term sustainability of the manufacturing process, making it an attractive option for green chemistry initiatives within large pharmaceutical organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and reliability for potential licensees or manufacturing partners.
Q: What catalysts are suitable for this 1,2-cis-glycoside synthesis?
A: The patent specifies the use of Ni(II) catalysts such as nickel diacetone (Ni(acac)2) or Fe(III) catalysts like iron triacetylacetone. These are preferred over stoichiometric toxic tin reagents used in older methods.
Q: Does this method require protecting groups at the 1,2-positions of the donor?
A: No, a key advantage of this invention is the ability to use 1,2-unprotected glycosyl donors. This significantly shortens the synthetic route of the sugar module compared to traditional chiral auxiliary methods.
Q: What kind of stereoselectivity can be expected?
A: The method achieves high regioselectivity and stereoselectivity for 1,2-cis-glycosidic bonds. For example, Example 1 reports an 87% yield of the cis-product with excellent selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-cis-Glycoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed glycosylation technology for the next generation of carbohydrate-based therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2-cis-glycoside intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific pipeline candidates. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-purity glycoside intermediates for your critical projects.
