Advanced Synthesis of Pemetrexed Intermediate via Grignard Reaction for Commercial Scale-up
Advanced Synthesis of Pemetrexed Intermediate via Grignard Reaction for Commercial Scale-up
The global demand for high-quality oncology therapeutics continues to drive innovation in the synthesis of critical active pharmaceutical ingredients (APIs) and their precursors. Pemetrexed disodium, a potent antifolate antineoplastic agent marketed as Alimta, remains a cornerstone treatment for malignant pleural mesothelioma and non-small cell lung cancer. However, the economic viability and supply security of this life-saving medication are intrinsically linked to the efficiency of its synthetic pathway. Patent CN103265428A, published in August 2013, introduces a transformative methodology for preparing 4-[(3-halo-4-oxo) butyl] benzoic acid, a pivotal intermediate in the Pemetrexed value chain. This technical disclosure addresses long-standing bottlenecks in traditional manufacturing by replacing complex transition-metal catalysis with a robust Grignard-based strategy. By leveraging this intellectual property, manufacturers can achieve a reliable pharmaceutical intermediate supplier status, ensuring consistent quality and availability for downstream drug production.
![Chemical structure of Pemetrexed showing the core pyrrolo[2,3-d]pyrimidine scaffold](/insights/img/pemetrexed-intermediate-grignard-synthesis-pharma-supplier-20260305045527-01.png)
The structural complexity of Pemetrexed necessitates a highly precise synthetic approach to maintain stereochemical integrity and minimize genotoxic impurities. The molecule features a glutamic acid moiety linked to a substituted pyrrolo[2,3-d]pyrimidine core, which is further connected to a benzoyl group. The synthesis of the key bridge, 4-[2-(2-amino-4,7-dihydro-4-oxo-1H-pyrrolo[2,3-d]pyrimidine-5-yl)ethyl]benzoic acid (often referred to as Pemetrexed acid or Intermediate VI), is the critical juncture where process efficiency is determined. Traditional routes have struggled with yield losses and purification difficulties at this stage. The innovation detailed in CN103265428A offers a streamlined path to the precursor 4-[(3-halo-4-oxo) butyl] benzoic acid (Intermediate I), which serves as the electrophilic partner for constructing the heterocyclic core. This advancement is crucial for any organization seeking cost reduction in pharmaceutical intermediates manufacturing while adhering to rigorous regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing the Pemetrexed side chain have predominantly relied on Heck coupling reactions or multi-step Friedel-Crafts acylations, both of which present significant operational drawbacks for large-scale production. For instance, prior art such as United States Patent US6066732 describes a route utilizing 3-butene-1-ol and iodomethyl benzoate. While conceptually direct, this method suffers from the high cost and scarcity of 3-butene-1-ol, alongside the necessity for expensive palladium catalysts and phase-transfer agents. The reliance on precious metals not only inflates the bill of materials but also introduces severe challenges in meeting strict residual metal specifications required for oncology drugs. Furthermore, alternative routes involving nitro-condensation, as seen in other patents, often exhibit poor regioselectivity, leading to the formation of difficult-to-remove positional isomers that compromise the overall purity profile.
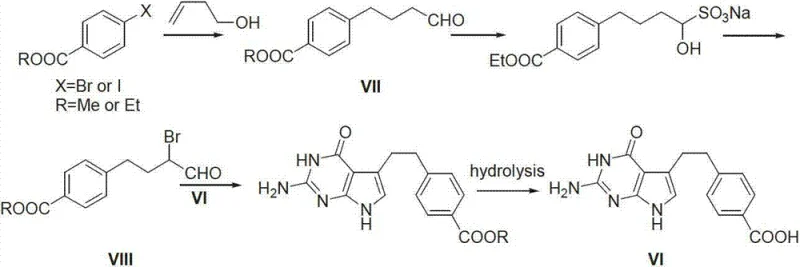
Another common historical pathway involves the use of organolithium reagents, such as n-Butyl Lithium, for alkylating p-toluic acid derivatives. While this avoids palladium, it introduces severe safety hazards associated with pyrophoric reagents and requires cryogenic conditions that are energy-intensive and difficult to manage in bulk reactors. Additionally, routes starting from benzene and maleic anhydride involve excessive step counts, including multiple acylations, reductions, and oxidations. Each additional unit operation in a synthetic sequence inherently compounds yield losses and increases the accumulation of by-products. These cumulative inefficiencies result in a low overall throughput and a substantial environmental footprint due to increased solvent and reagent consumption, rendering many of these legacy processes economically unviable for modern, competitive commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology disclosed in CN103265428A employs a highly efficient Grignard addition strategy that fundamentally simplifies the carbon skeleton construction. The core innovation lies in the reaction between 4-(2-halogenated ethyl) benzoic acid and glyoxal dialkyl acetal. This approach bypasses the need for transition metal catalysts entirely, eliminating the risk of heavy metal contamination and the associated costly purification steps like scavenging or recrystallization specifically for metal removal. The use of glyoxal acetals provides a masked aldehyde functionality that allows for controlled chain extension without the volatility and handling issues associated with free aldehydes. Following the Grignard addition, a tandem halogenation and deprotection sequence efficiently unveils the reactive alpha-halo ketone motif required for the subsequent cyclization.
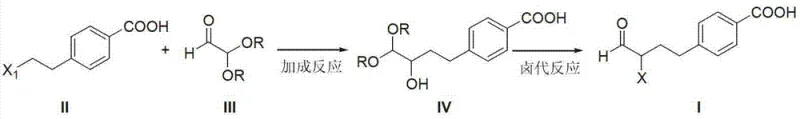
This novel route offers distinct advantages in terms of atom economy and operational simplicity. By utilizing readily available starting materials such as 4-(2-bromoethyl)benzoic acid and glyoxal dimethyl acetal, the process ensures a stable and predictable supply chain, mitigating the risks associated with sourcing specialized reagents. The reaction conditions are relatively mild, typically proceeding in common solvents like tetrahydrofuran (THF) or methyl tert-butyl ether (MTBE) at temperatures ranging from 0°C to 45°C. This thermal flexibility reduces energy consumption compared to cryogenic organolithium processes. Furthermore, the workup procedures described, involving standard aqueous quenches and organic extractions, are easily adaptable to existing multipurpose reactor infrastructure. This adaptability makes the process an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond rapidly to market demands without extensive capital investment in new equipment.
Mechanistic Insights into Grignard-Mediated Chain Extension
The mechanistic elegance of this synthesis rests on the nucleophilic attack of the Grignard reagent on the protected glyoxal species. Initially, the magnesium insertion into the carbon-halogen bond of the benzoic acid derivative generates a highly reactive organomagnesium species. To prevent premature protonation or side reactions with the carboxylic acid proton, the reaction is carefully managed, often utilizing the carboxylate salt form or protecting group strategies implicitly handled by the excess Grignard reagent. The nucleophilic carbon then attacks the electrophilic carbonyl carbon of the glyoxal acetal. This addition forms a new carbon-carbon bond, extending the side chain by two carbons while maintaining the oxidation state necessary for the subsequent ketone formation. The acetal group serves as a crucial protecting group, stabilizing the intermediate alcohol and preventing polymerization or self-condensation that free aldehydes might undergo under basic conditions.
Following the formation of the hydroxy-acetal intermediate, the process transitions to a halogenation and deprotection phase. The use of phosphorus tribromide (PBr3) serves a dual purpose: it converts the secondary alcohol into a leaving group (bromide) and facilitates the hydrolysis of the acetal back to the ketone under acidic workup conditions. This tandem transformation is kinetically favorable and minimizes the isolation of unstable intermediates. The resulting 4-[(3-halo-4-oxo) butyl] benzoic acid possesses an alpha-halo ketone functionality that is perfectly poised for nucleophilic substitution by the amine groups of 2,4-diamino-6-hydroxypyrimidine. This specific arrangement ensures high regioselectivity during the cyclization step, effectively suppressing the formation of unwanted isomers that plague nitro-based routes. The control over impurity profiles at this molecular level is paramount for R&D directors focused on high-purity pharmaceutical intermediates and regulatory compliance.
How to Synthesize 4-[(3-halo-4-oxo) butyl] benzoic acid Efficiently
The practical execution of this synthesis involves a sequence of well-defined unit operations that balance reaction kinetics with safety and yield optimization. The process begins with the generation of the Grignard reagent under an inert atmosphere, followed by the controlled addition of the acetal electrophile. Subsequent steps involve careful pH management during the halogenation and hydrolysis phases to ensure complete conversion while minimizing degradation. The final condensation with the pyrimidine base requires precise temperature control to drive the cyclization to completion. For a comprehensive understanding of the specific molar ratios, solvent volumes, and temperature ramps validated in the patent examples, please refer to the standardized protocol below.
- Perform a Grignard reaction between 4-(2-halogenated ethyl) benzoic acid and glyoxal dialkyl acetal in THF/MTBE solvent to generate the hydroxy-acetal intermediate.
- Subject the intermediate to halogenation using phosphorus tribromide followed by acid-catalyzed deprotection to yield 4-[(3-halo-4-oxo) butyl] benzoic acid.
- Condense the resulting keto-acid with 2,4-diamino-6-hydroxypyrimidine under basic conditions to form the key Pemetrexed precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN103265428A represents a strategic opportunity to optimize the cost structure and resilience of the Pemetrexed supply chain. The elimination of palladium catalysts is not merely a technical improvement but a significant financial lever. Precious metal catalysts are subject to volatile market pricing and require complex recovery or disposal protocols to meet environmental regulations. By shifting to a base-metal-free Grignard process, manufacturers can achieve substantial cost savings in raw material procurement and waste management. Furthermore, the simplified workflow reduces the number of isolation and purification steps, which directly correlates to lower labor costs, reduced solvent consumption, and shorter cycle times. These efficiencies compound to offer a more competitive pricing model for the final API.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the avoidance of expensive transition metal catalysts and the utilization of commodity chemicals. Glyoxal acetals and halo-ethyl benzoic acids are produced on a massive industrial scale for various applications, ensuring stable pricing and abundant availability. Unlike specialized allyl alcohols or nitro-compounds which may have limited suppliers, these feedstocks can be sourced from multiple global vendors, fostering a competitive bidding environment that drives down input costs. Additionally, the higher selectivity of the reaction reduces the burden on downstream purification, meaning less product is lost to mother liquors or chromatography, thereby improving the effective yield per batch and maximizing asset utilization.
- Enhanced Supply Chain Reliability: Supply chain continuity is often jeopardized by reliance on single-source or geographically concentrated raw materials. The starting materials for this Grignard route are chemically simple and widely manufactured, significantly mitigating the risk of supply disruptions. The robustness of the chemistry also means that the process is less sensitive to minor fluctuations in reagent quality, providing a buffer against supply variability. This reliability is critical for maintaining uninterrupted production schedules for essential oncology medications. By securing a manufacturing process based on ubiquitous chemical building blocks, companies can insulate themselves from the geopolitical and logistical fragilities that often impact the supply of complex, specialized reagents used in older synthetic methods.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a cleaner profile. The absence of heavy metals simplifies wastewater treatment and reduces the hazard classification of the final product regarding residual impurities. The solvents employed, such as THF and dichloromethane, are standard in the industry with well-established recovery and recycling infrastructures. The reduction in step count inherently lowers the total E-factor (mass of waste per mass of product), aligning with green chemistry principles and increasingly stringent environmental regulations. This compliance ease facilitates faster regulatory approvals and smoother audits, ensuring that the manufacturing facility can operate at full capacity without environmental bottlenecks, thus supporting seamless commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims within patent CN103265428A, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does this Grignard-based route compare to traditional Heck reaction methods?
A: Unlike conventional Heck coupling methods that require expensive palladium catalysts and difficult-to-source allyl alcohols, this novel route utilizes readily available glyoxal acetals and avoids precious metals, significantly reducing raw material costs and simplifying purification.
Q: What are the purity advantages of this synthesis method?
A: The process described in CN103265428A minimizes the formation of positional isomers often seen in nitro-condensation routes, resulting in a key intermediate with superior stability and high purity suitable for stringent pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the methodology features concise reaction steps, mild conditions, and the use of common solvents like THF and dichloromethane, making it highly adaptable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-[(3-halo-4-oxo) butyl] benzoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry outlined in CN103265428A can be seamlessly translated into your supply chain. Our facilities are equipped with state-of-the-art reactors capable of handling Grignard reactions and moisture-sensitive chemistry under strict inert atmospheres. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for oncology drug synthesis, providing you with a secure and high-quality source of critical materials.
We invite you to collaborate with us to unlock the full potential of this advanced synthetic route. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to our process optimization expertise and reliable logistics network. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can support your goal of delivering affordable and accessible cancer therapies to patients worldwide.
