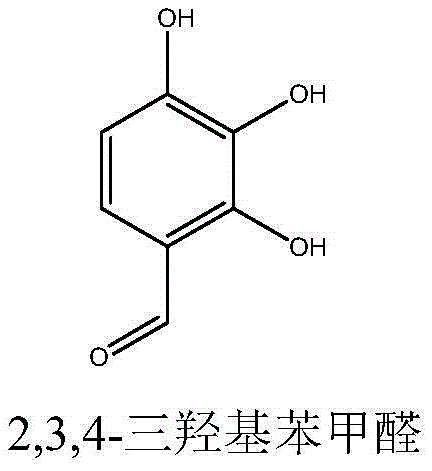
Advanced Synthesis of 2,3,4-Trihydroxybenzaldehyde for Commercial API Manufacturing
The pharmaceutical industry's demand for high-purity intermediates for antiparkinson medications has driven significant innovation in synthetic methodology, particularly for 2,3,4-trihydroxybenzaldehyde, a critical precursor to Benserazide. As detailed in the recent patent CN112830872A, a robust three-step synthesis route has been developed that overcomes the historical limitations of toxicity and low yield associated with traditional methods. This technical breakthrough leverages a strategic phenolic hydroxyl protection mechanism followed by regioselective formylation and mild deprotection. For R&D directors and procurement specialists, this represents a paradigm shift towards safer, more scalable manufacturing processes that align with modern green chemistry principles while ensuring the stringent purity specifications required for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,4-trihydroxybenzaldehyde has been plagued by significant safety hazards and inefficiencies that hinder large-scale commercial viability. The classic Gattermann reaction, while theoretically low-cost, relies on hydrocyanic acid or zinc cyanide, introducing extreme toxicity risks that have led to its near-abandonment in modern regulated facilities. Alternatively, the Reimer-Tiemann reaction using chloroform and sodium hydroxide typically results in yields below 50% with challenging ortho-to-para isomer ratios that complicate downstream purification. Furthermore, direct Friedel-Crafts approaches using triethyl orthoformate generate excessive wastewater, often reaching 40 to 50 times the volume of the raw material input, creating an unsustainable environmental burden for chemical manufacturers.
The Novel Approach
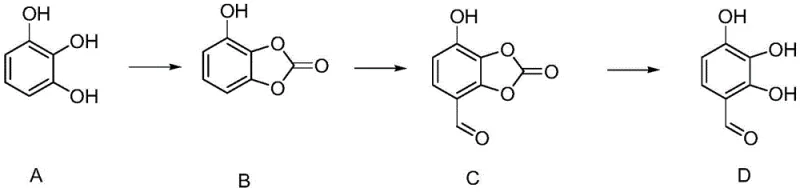
The methodology disclosed in patent CN112830872A introduces a sophisticated protection-formylation-deprotection sequence that fundamentally resolves these legacy issues. By initially converting pyrogallol into a cyclic carbonate derivative (Compound B), the synthesis effectively masks specific hydroxyl groups, thereby directing the subsequent formylation exclusively to the desired position. This strategic intervention not only eliminates the formation of difficult-to-separate isomers but also facilitates a much cleaner reaction profile. The result is a streamlined process where the final product achieves a purity exceeding 99% directly after operation, bypassing the need for multiple recrystallization steps that typically erode overall yield in conventional direct formylation protocols.
Mechanistic Insights into Regioselective Formylation via Carbonate Protection
The core innovation of this synthesis lies in the precise manipulation of electronic effects through temporary protecting groups. In the first step, pyrogallol reacts with diphenyl carbonate in the presence of triethylamine to form a five-membered cyclic carbonate ring. This structural modification drastically alters the electron density distribution of the aromatic system, deactivating certain positions while leaving the target carbon available for electrophilic attack. When Compound B is subjected to formylation conditions using dichloromethyl ethyl ether and aluminum trichloride at controlled low temperatures of 0-5°C, the Lewis acid catalyst activates the formylating agent to attack the specific activated site on the ring. This regiocontrol is the mechanistic key that prevents the formation of unwanted byproducts, ensuring that the aldehyde group is installed exactly where needed for the final API structure.
Following formylation, the deprotection step utilizes a remarkably simple hydrolysis mechanism that underscores the process's elegance. Unlike harsh acidic or basic conditions that might degrade sensitive functional groups, the cyclic carbonate in Compound C is cleaved merely by heating in water under reflux. This hydrolytic instability of the carbonate linkage allows for the regeneration of the free phenolic hydroxyl groups without requiring exotic reagents or generating hazardous waste streams. The simplicity of this final transformation contributes significantly to the high isolated yield of 93-95% in the last step, demonstrating how thoughtful molecular design can simplify downstream processing and reduce the complexity of impurity profiles in the final crude product.
How to Synthesize 2,3,4-Trihydroxybenzaldehyde Efficiently
The operational protocol for this synthesis is designed for reproducibility and scalability, utilizing common industrial solvents like methyl tert-butyl ether (MTBE). The process begins with the protection of pyrogallol, followed by the critical low-temperature formylation which requires careful thermal management to maintain selectivity. The final hydrolysis step is operationally trivial, requiring only water and heat. For process chemists looking to implement this route, the detailed standardized synthesis steps are outlined in the guide below, ensuring consistent quality and yield across different batch sizes.
- Step 1: Protection of pyrogallol using diphenyl carbonate and triethylamine in methyl tert-butyl ether to form the cyclic carbonate intermediate (Compound B).
- Step 2: Regioselective formylation of Compound B using dichloromethyl ethyl ether and aluminum trichloride at low temperatures (0-5°C) to yield Compound C.
- Step 3: Hydrolytic deprotection of Compound C by refluxing in water to remove the carbonate group and isolate the final 2,3,4-trihydroxybenzaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this patented route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The ability to recycle solvents and aqueous layers directly translates to reduced raw material consumption and lower waste disposal costs, which are critical factors in the total cost of ownership for fine chemical intermediates. Furthermore, the elimination of highly toxic cyanide reagents simplifies regulatory compliance and reduces the need for specialized safety infrastructure, making the manufacturing process more accessible and resilient against regulatory tightening.
- Cost Reduction in Manufacturing: The implementation of this protective group strategy significantly lowers production costs by minimizing the loss of valuable starting materials during purification. Because the reaction achieves high regioselectivity, the need for expensive chromatographic separations or multiple recrystallizations is virtually eliminated, preserving the mass balance throughout the synthesis. Additionally, the recovery and reuse of methyl tert-butyl ether and aqueous wash solutions create a closed-loop system that drastically cuts down on solvent procurement expenses and waste treatment fees.
- Enhanced Supply Chain Reliability: By avoiding the use of restricted and highly hazardous substances like hydrocyanic acid, manufacturers can secure a more stable supply of raw materials without facing the logistical bottlenecks associated with controlled chemicals. The robustness of the reaction conditions, particularly the tolerance for standard industrial equipment and the mild nature of the deprotection step, ensures that production schedules are less likely to be disrupted by equipment corrosion or safety incidents, guaranteeing consistent delivery timelines for downstream API producers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with the exothermic formylation step being manageable at 0-5°C using standard cooling jackets available in most multipurpose reactors. The substantial reduction in wastewater generation—avoiding the 40-50 fold volume increase seen in older methods—aligns perfectly with increasingly strict environmental regulations, allowing facilities to operate at higher capacities without exceeding discharge limits. This environmental efficiency future-proofs the supply chain against potential regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety protocols, and purity outcomes for stakeholders evaluating this technology for adoption.
Q: Why is the cyclic carbonate protection method superior to direct formylation for 2,3,4-trihydroxybenzaldehyde?
A: Direct formylation methods like Reimer-Tiemann often suffer from low yields (<50%) and difficult ortho/para separation. The protection strategy described in patent CN112830872A ensures regioselectivity, preventing side reactions and achieving a total yield of 82-86% with >99% purity without complex purification.
Q: What are the safety advantages of this synthesis route compared to the Gattermann reaction?
A: Traditional Gattermann reactions utilize highly toxic hydrocyanic acid or zinc cyanide, posing severe operational risks. This novel method eliminates the need for cyanide reagents entirely, utilizing safer formylation agents like dichloromethyl ethyl ether, significantly enhancing workplace safety and environmental compliance.
Q: How does this process impact solvent consumption and waste generation?
A: The process utilizes methyl tert-butyl ether (MTBE) which can be repeatedly recycled after concentration. Additionally, aqueous layers from the workup can be reused multiple times after pH adjustment. This contrasts sharply with direct Friedel-Crafts methods that generate sewage volumes 40-50 times the feed amount, offering substantial waste reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4-Trihydroxybenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in patent literature are faithfully reproduced on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of 2,3,4-trihydroxybenzaldehyde meets the exacting standards required for Parkinson's disease medication synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your project's efficiency and profitability.
